

铁催化碳-碳偶联反应研究进展
收稿日期: 2016-08-16
修回日期: 2016-09-22
网络出版日期: 2016-10-18
基金资助
国家自然科学基金(Nos.21372031,21502075)、江西理工大学博士启动经费资助项目.
Recent Progress in Iron Catalyzed C-C Coupling Reactions
Received date: 2016-08-16
Revised date: 2016-09-22
Online published: 2016-10-18
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 21372031, 21502075) and the Doctoral Scientific Research Foundation of Jiangxi University of Science and Technology.
李娟华 , 刘昆明 , 段新方 , 刘晋彪 . 铁催化碳-碳偶联反应研究进展[J]. 有机化学, 2017 , 37(2) : 314 -334 . DOI: 10.6023/cjoc201608009
Transition-metal-catalyzed coupling reactions play an important role in construct C-C bond. As one of the most abundant metal in the earth's crust, along with its inexpensive price, relatively low toxicity and multiple redox states, Fe is an ideal catalyst for coupling reactions. Recently, green, highly effective and selective reactions have attracted much attention, which accelerated the research of iron catalyzed coupling. Particularly, many new catalyst systems and various reaction types such as iron catalyzed oxidative coupling, reductive coupling and C-H direct functionalization were explored during the past decade. The present review surveys the recent progress in Fe catalyzed C-C coupling concerning their reaction types, including mechanism and application. Furthermore, the prospects of this reaction are also discussed.
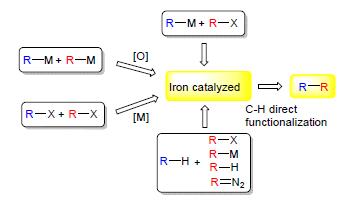
Key words: iron catalyzed; C-C coupling reaction; organic synthesis
[1] (a) De Mejiere, A.; Diederich, F. Metal-Catalyzed Cross-Coupling Reactions, Wiley-VCH, Weinheim, 2004.
(b) Hartwig, J. F. Organotransition Metal Chemistry, University Science Books, Sausalito, California, 2010.
(c) Liu, C.; Zhang, H.; Shi, W.; Lei, A. W. Chem. Rev. 2011, 111, 1780.
[2] (a) Graening, T.; Schmalz, H.-G. Angew. Chem., Int. Ed. 2003, 42, 2580.
(b) Nicolaou, K. C.; Bulger, P. G.; Sarlah, D. Angew. Chem., Int. Ed. 2005, 44, 4490.
(c) Magano, J.; Dunetz, J. R. Chem. Rev. 2011, 111, 2177.
[3] (a) Wu, X.-F.; Anbarasan, P.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2010, 49, 9047.
(b) Chen, X.; Engle, K. M.; Wang, D.-H.; Yu, J.-Q. Angew. Chem., Int. Ed. 2009, 48, 5094.
[4] Van Leeuwen, P. N. M.; Kamer, P. C. J.; Claver, C.; Pamies, O.; Dieguez, M. Chem. Rev. 2011, 111, 2077.
[5] For recent reviews, see:
(a) Song, Y.; Tang, X. S.; Hou, X. M.; Bai, Y. J. Chin. J. Org. Chem. 2013, 33, 76 (in Chinese).(宋阳, 唐雪松, 侯晓萌, 白银娟, 有机化学, 2013, 33, 76.)
(b) Riener, K.; Haslinger, S.; Raba, A.; Högerl, M. P.; Cokoja, M.; Herrmann, W. A.; Kühn. F. E. Chem. Rev. 2014, 114, 5215.
[6] (a) Bolm, C.; Legros, J.; Paih, J. L.; Zani, L. Chem. Rev. 2004, 104, 6217.
(b) Gopalaiah. K. Chem. Rev. 2013, 113, 3248.
(c) Bauer, I.; Knölker, H.-J. Chem. Rev. 2015, 115, 3170.
(d) Guo, N.; Zhu, S. F. Chin. J. Org. Chem. 2015, 35, 1383 (in Chinese).(郭娜, 朱守非, 有机化学2015, 35, 1383.)
[7] (a) Bolm, C.; Legros, J.; Le Paih, J.; Zani, L. Chem. Rev. 2004, 104, 6217.
(b) Plietker, B. Iron Catalysis in Organic Chemistry: Reactions and Applications, Wiley-VCH, Weinheim, 2008.
(c) Sherry, B. D.; Fürstner, A. Acc. Chem. Res. 2008, 41, 1500.
(d) Fürstner, A. Angew. Chem., Int. Ed. 2009, 48, 1364.
(e) Cahiez, G.; Moyeux, A. Chem. Rev. 2010, 110, 1435.
[8] (a) Wang, C.; Fu, Y.; Liu, L.; Guo, Q. X. Chin. J. Org. Chem. 2007, 27, 703 (in Chinese).(王晨, 傅尧, 刘磊, 郭庆祥, 有机化学, 2007, 27, 703.)
(b) Nakamura, E.; Yoshikai, N. J. Org. Chem. 2010, 75, 6061.
[9] Kharasch, M. S.; Fields, E. K. J. Am. Chem. Soc. 1941, 63, 2316.
[10] (a) Kochi, J. K. J. Am. Chem. Soc. 1971, 93, 1487.
(b) Kochi, J. K. J. Org. Chem. 1976, 41, 599.
[11] Walborsky, H. M. J. Org. Chem. 1981, 46, 5074.
[12] Molander, G. A. Tetrahedron Lett. 1983, 24, 5449.
[13] (a) Cahiez, G. Synthesis 1998, 1199.
(b) Cahiez, G. Synlett 2001, 1901.
(c) Cahiez, G.; Avedissian, H. Tetrahedron Lett. 1998, 39, 6159.
(d) Cahiez, G.; Marquais, S. Pure Appl. Chem. 1996, 68, 53.
[14] (a) Fürstner, A.; Leitner, A. Angew. Chem., Int. Ed. 2002, 41, 609.
(b) Fürstner, A.; Leitner, A.; Méndez, M.; Krause, H. J. Am. Chem. Soc. 2002, 124, 13856.
[15] (a) Fürstner, A.; Leitner, A. Angew. Chem., Int. Ed. 2003, 42, 308.
(b) Fürstner, A.; Souza, D. D.; Rapado, L. P.; Jensen, J. T. Angew. Chem., Int. Ed. 2003, 42, 5358.
(c) Fürstner, A.; Turet, L. Angew. Chem., Int. Ed. 2005, 44, 3462.
[16] Scheiper, B.; Bonnekessel, M.; Krause, H.; Fürstner, A. J. Org. Chem. 2004, 69, 3949.
[17] Seidel, G.; Laurich, D.; Fürstner, A. J. Org. Chem. 2004, 69, 3950.
[18] (a) Jonas, K.; Schieferstein, L.; KrMger, C.; Tsay, Y.-H. Angew. Chem., Int. Ed. Engl. 1979, 18, 550.
(b) Jonas, K.; Schieferstein, L. Angew. Chem., Int. Ed. Engl. 1979, 18, 549.
[19] Hocek, M.; Dvo?ákova, H. J. Org. Chem. 2003, 68, 5773.
[20] Ottesen, L. K.; Fredrik, E. K.; Olsson, R. Org. Lett. 2006, 8, 1771.
[21] Nakamura, M.; Matsuo, K.; Ito, S.; Nakamura, E. J. Am. Chem. Soc. 2004, 126, 3686.
[22] Martin, R.; Fürstner, A. Angew. Chem., Int. Ed. 2004, 43, 3955.
[23] Cahiez, G.; Habiak, V.; Duplais, C.; Moyeux, A. Angew. Chem., Int. Ed. 2007, 46, 4364.
[24] Cahiez, G.; Duplais, C.; Moyeux, A. Org. Lett. 2007, 9. 325.
[25] Gurinot, A.; Reymond, S.; Cossy, J. Angew. Chem., Int. Ed. 2007, 46, 6521.
[26] Gurinot, A.; Lepesqueux, G.; Sable, S.; Reymond, S.; Cossy, J. J. Org. Chem. 2010, 75, 5151.
[27] (a) Bedford, R. B.; Bruce, D. W.; Frost, R. M.; Goodbyb, J. W.; Hirdb, M. Chem. Commun. 2004, 2822.
(b) Bedford, R. B.; Bruce, D. W.; Frost, R. M.; Hirdb, M. Chem. Commun. 2005, 4161.
[28] Chowdhury, R. R.; Crane, A. K.; Fowler, C.; Kwong, P.; Kozak, C. M. Chem. Commun. 2008, 94.
[29] Yamaguchi, Y.; Ando, H.; Nagaya, M.; Hinago, H.; Ito, T.; Asami, M. Chem. Lett. 2011, 40, 983.
[30] Bedford, R. B.; Betham, M.; Bruce, D. W.; Danopoulos, A. A.; Robert, M. F.; Michael, H. J. Org. Chem. 2006, 71, 1104.
[31] Bedford, R. B.; Huwea, M.; Wilkinson, M. C. Chem. Commun. 2009, 600.
[32] Hatakeyama, T.; Kondo, Y.; Fujiwara, Y.-I.; Takaya, H.; Ito, S.; Nakamura, E.; Nakamura, M. Chem. Commun. 2009, 1216.
[33] (a) Tamura, M.; Kochi, J. J. Organomet. Chem. 1971, 31, 289.
(b) Tamura, M.; Kochi, J. K. Bull. Chem. Soc. Jpn. 1971, 44, 3063.
[34] Adams, C. J.; Bedford, R. B.; Carter, E.; Gower, N. J.; Haddow, M. F.; Harvey, J. N.; Huwe, M.; Cartes, M. Á.; Mansell, S. M.; Mendoza, C.; Murphy, D. M.; Neeve, E. C.; Nunn, J. J. Am. Chem. Soc. 2012, 134, 10333.
[35] Bedford, R. B.; Carter, E.; Cogswell, P. M.; Gower, N. J.; Haddow, M. F.; Harvey, J. N.; Murphy, D. M.; Neeve, E. C.; Nunn, J. Angew. Chem., Int. Ed. 2013, 52, 1285.
[36] Jana, R.; Pathak, T. P.; Sigman, M. S. Chem. Rev. 2012, 111, 1417.
[37] (a) Hatakeyama, T.; Hashimoto, T.; Kondo, Y.; Fujiwara, Y.; Seike, H.; Takaya, H.; Tamada, Y.; Ono, T.; Nakamura, M. J. Am. Chem. Soc. 2010, 132, 10674.
(b) Hashimoto, T.; Hatakeyama, T.; Nakamura, M. J. Org. Chem. 2012, 77, 1168.
(c) Hatakeyama, T.; Hashimoto, T.; Kathriarachchi, K. K. A. D. S.; Zenmyo, T.; Seike, H.; Nakamura, M. Angew. Chem., Int. Ed. 2012, 124, 1.
(d) Nakagawa, N.; Hatakeyama, T.; Nakamura, M. Chem. Lett. 2015, 44, 486.
[38] Bedford, R. B.; Brenner, P. B.; Carter, E.; Clifton, J.; Cogswell, P. M.; Gower, N. J.; Haddow, M. F.; Harvey, J. N.; Kehl, J. A.; Murphy, D. M.; Neeve, E. C.; Neidig, M. L.; Nunn, J.; Snyder, B. E. R.; Taylor, J. Organometallics 2014, 33, 5767.
[39] Nakamura, M.; Hirai, A.; Nakamura, E. J. Am. Chem. Soc. 2000, 122, 978.
[40] Jin, M.; Adak, L.; Nakamura, M. J. Am. Chem. Soc. 2015, 137, 7128.
[41] Jin, M.; Nakamura, M. Chem. Lett. 2011, 40, 1012.
[42] Cahiez, G.; Marquais, S. Pure Appl. Chem. 1996, 68, 53..
[43] (a) Sapountzis, I.; Lin, W. W.; Kofink, C. C.; Despotopoulou, C.; Knochel, P. Angew. Chem., Int. Ed. 2005, 44, 1654.
(b) Kofink, C. C.; Blank, B.; Pagano, S.; Gótz, N.; Knochel, P. Chem. Commun. 2007, 1954.
[44] (a) Hatakeyama, T.; Nakagawa, N.; Nakamura, M. Org. Lett. 2009, 11, 4496.
(b) Ito, S.; Fujiwara, Y.-I.; Nakamura, E.; Nakamura, M. Org. Lett. 2009, 11, 4306.
[45] Cahiez, G.; Habiak, V.; Gager, O. Org. Lett. 2008, 10, 2389.
[46] Li, B.-J.; Xu, L.; Wu, Z.-H.; Guan, B.-T.; Sun, C.-L.; Wang, B.-Q.; Shi, Z.-J. J. Am. Chem. Soc. 2009, 131, 14656.
[47] Agrawal, T.; Cook, S. P. Org. Lett. 2013, 15, 96.
[48] Itami, K.; Higashi, S.; Mineno, M.; Yoshida, J.-I. Org. Lett. 2005, 7, 1219.
[49] Denmark, S. E.; Cresswell, A. J. J. Org. Chem. 2013, 78, 12593.
[50] Chandra, M.; Volla, R.; Vogel, P. Angew. Chem., Int. Ed. 2008, 47, 1305.
[51] Dongol, K. G.; Koh, H.; Sau, M. and Chai, C. L. L. Adv. Synth. Catal. 2007, 349, 1015.
[52] Hatakeyama, T.; Hashimoto, T.; Kathriarachchi, K. K. A. D. S.; Zenmyo, T.; Seike, H.; Nakamura, M. Angew. Chem., Int. Ed. 2012, 51, 8834.
[53] Hatakeyama, T.; Yoshimoto, Y.; Gabriel, T.; Nakamura, M. Org. Lett. 2008, 10, 5341.
[54] Xie, X.; Xu, X. B.; Li, H. F.; Xu, X. L.; Yang, J. Y.; Lia, Y. Z. Adv. Synth. Catal. 2009, 351, 1263.
[55] Carril, M.; Correa, A.; Bolm, C. Angew. Chem., Int. Ed. 2008, 47, 4862.
[56] Mao, J. C.; Xie, G. L.; Wu, M. Y.; Guo, J.; Ji, S. J. Adv. Synth. Catal. 2008, 350, 2477.
[57] Chandra, M.; Volla, R.; Vogel, P. Tetrahedron Lett. 2008, 49, 5961.
[58] Hatakeyama, T.; Okada, Y.; Yoshimoto, Y.; Nakamura M. Angew. Chem., Int. Ed. 2011, 50, 10973.
[59] Nakagawa, N.; Hatakeyama, T.; Nakamura, M. Chem. Lett. 2015, 44, 486.
[60] Cheung, C. W.; Ren, P.; Hu, X. L. Org. Lett. 2014, 16, 2566.
[61] Quintin, J.; Franck, X.; Hocquemiller, R.; Figadère, B. Tetrahe-dron Lett. 2002, 43, 3547.
[62] Ludovic, B.; Mirca, D.; Alain, T.; Nelly, P. J. Heterocycl. Chem. 2005, 42, 1423.
[63] Kuzmina, O. M.; Steib, A. K.; Flubacher, D.; Knochel, P. Org. Lett. 2012, 14, 4818.
[64] Kuzmina, O. M.; Steib, A. K.; Markiewicz, J. T.; Flubacher, D.; Knochel, P. Angew. Chem., Int. Ed. 2013, 52, 4945.
[65] Hatakeyama, T.; Nakamura, M. J. Am. Chem. Soc. 2007, 129, 9844.
[66] Hatakeyama, T.; Hashimoto, S.; Ishizuka, K.; Nakamura, M. J. Am. Chem. Soc. 2009, 131, 11949.
[67] Agrawal, T.; Cook, S. P. Org. Lett. 2014, 16, 5080.
[68] (a) Liu, C.; Zhang, H.; Shi, W.; Lei, A. W. Chem. Rev. 2011, 1780.
(b) Shi, W.; Liu, C.; Lei, A. W. Chem. Soc. Rev. 2011, 40, 2761.
[69] Selected reviews:
(a) Littke, A. F.; Fu, G. C. Angew. Chem., Int. Ed. 2002, 41, 4176.
(b) Fu, G. C. Acc. Chem. Res. 2008, 41, 1555.
(c) Martin, R.; Buchwald, S. L. Acc. Chem. Res. 2008, 41, 1461.
(d) Marion, N.; Nolan, S. P. Acc. Chem. Res. 2008, 41, 1440.
(e) Fortman, G. C.; Nolan, S. P. Chem. Soc. Rev. 2011, 40, 5151.
[70] Selected reviews:
(a) Mulvey, R. E.; Mongin, F.; Uchiyama, M.; Kondo, Y. Angew. Chem., Int. Ed. 2007, 46, 3802.
(b) Haag, B.; Mosrin, M.; Ila, H.; Malakhov, V.; Knochel, P. Angew. Chem., Int. Ed. 2011, 50, 9794.
(c) Mongin, F.; Harrison-Marchand, A. Chem. Rev. 2013, 113, 7563.
(d) Klatt, T.; Markiewicz, J. T. Sämann, C.; Knochel, P. J. Org. Chem. 2014, 79, 4253.
[71] Nagano, T.; Hayashi, T. Org. Lett. 2005, 7, 491.
[72] Cahiez, G.; Chaboche, C.; Betzer, F. M.; Ahr, M. Org. Lett. 2005, 7, 1943.
[73] Cahiez, G.; Moyeux, A.; Buendia, J.; Duplais, C. J. Am. Chem. Soc. 2007, 129, 13788.
[74] Liu, W.; Lei, A. W. Tetrahedron Lett. 2008, 49, 610.
[75] Truong, T.; Alvarado, J.; Tran, L. D.; Daugulis, O. Org. Lett. 2010, 12, 1200.
[76] Cahiez, G.; Foulgoc, L.;Moyeux, A. Angew. Chem., Int. Ed. 2009, 48, 2969.
[77] Liu, K. M.; Liao, L. Y.; Duan, X. F. Chem. Commun. 2015, 51, 1124.
[78] Liu, K. M.; Wei, J.; Duan, X. F. Chem. Commun. 2015, 51, 4655.
[79] Xu, X. L.; Cheng, D. P.; Pei, W. J. Org. Chem. 2006, 71, 6637.
[80] Sridevi, V. S.; Leong, W. -K. Tetrahedron Lett. 2007, 48, 6669.
[81] Czaplik, W. M.; Mayer, M.; von Wangelin, A. J. ChemCatChem 2011, 3, 135
[82] For selected literatues on Pd catalyzed C-H activation:
(a) Jia, C.; Piao, D.; Oyamada, J.; Lu, W.; Kitamura, T.; Fujiwara, Y. Science 2000, 287, 1992.
(b) Lyons, T. W.; Sanford, M. S. Chem. Rev. 2010, 110, 1147.
(c) Chen, X.; Engle, K. M.; Wang, D.-H.; Yu, J.-Q. Angew. Chem., Int. Ed. 2009, 48, 5094. Ir catalyzed C-H activation:
(d) Cho, J.-Y.; Tse, M. K.; Holmes, D. R.; Maleczka, E., Jr.; Smith, M. R., Ⅲ Science 2002, 295, 305.
(e) Mkhalid, I. A. I.; Barnard, J. H.; Marder, T. B.; Murphy, J. M.; Hartwig, J. F. Chem. Rev. 2010, 110, 890. Ru catalyzed C-H activation:
(f) Ritleng, V.; Sirlin, C.; Pfeffer, M. Chem. Rev. 2002, 102, 1731.
(g) Colby, D. A.; Bergman, R. G.; Ellman, J. A. Chem. Rev. 2010, 110, 624. Pt catalyzed C-H activation:
(h) Lersch, M.; Tilset, M. Chem. Rev. 2005, 105, 2471.
(i) Yang, S.; Li, Z.; Jian, X.; He, C. Angew. Chem., Int. Ed. 2009, 48, 3999.
[83] Jones, W. D.; Foster, G. P.; Putinas, J. M. J. Am. Chem. Soc. 1987, 109, 5047.
[84] Wen, J.; Zhang, J.; Chen, S.-Y.; Li, J.; Yu, X.-Q. Angew. Chem., Int. Ed. 2008, 47, 8897.
[85] Wen, J.; Qin, S.; Ma, L.-F.; Dong, L.; Zhang, J.; Liu, S.-S.; Duan, Y.-S.; Chen, S.-Y.; Hu, C.-W.; Yu X.-Q. Org. Lett. 2010, 12, 2694.
[86] Deb, A.; Manna, S.; Maji, A.; Dutta, U.; Maiti, D. Eur. J. Org. Chem. 2013, 5251.
[87] Liu, W.; Cao, H.; Lei, A. W. Angew. Chem., Int. Ed. 2010, 49, 2004.
[88] Vallée, F.; Mousseau, J. J.; Charette, A. J. Am. Chem. Soc. 2010, 132, 1514.
[89] Norinder, J.; Matsumoto, A.; Yoshikai, N.; Nakamura, E. J. Am. Chem. Soc. 2008, 130, 5858.
[90] Yoshikai, N.; Matsumoto, A.; Norinder, J.; Nakamura, E. Angew. Chem., Int. Ed. 2009, 48, 2925.
[91] Yoshikai, N.; Matsumoto, A.; Norinder, J.; Nakamura, E. Synlett 2010, 313.
[92] Yoshikai, N.; Asako, S.; Yamakawa, T.; Ilies, L.; Nakamura, E. Chem. Asian J. 2011, 6, 3059.
[93] Ilies, L.; Kobayashi, M.; Matsumoto, A.; Yoshikai, N.; Nakamura, E. Adv. Synth. Catal. 2012, 354, 593.
[94] Shang, R.; Ilies, L.; Matsumoto, A.; Nakamura, E. J. Am. Chem. Soc. 2013, 135, 6030.
[95] Shang, R.; Ilies, L.; Nakamura, E. J. Am. Chem. Soc. 2015, 137, 7660.
[96] Shang, R.; Ilies, L.; Nakamura, E. J. Am. Chem. Soc. 2016, 138, 10132.
[97] Li, Z. P.; Cao, L.; Li, C.-J. Angew. Chem., Int. Ed. 2007, 46, 6505.
[98] Li, Y.-Z;. Li, B.-J.; Lu, X.-Y.; Lin, S.; Shi, Z.-J. Angew. Chem., Int. Ed. 2009, 48, 1.
[99] Zhang, Y.; Li, C.-J. Eur. J. Org. Chem. 2007, 4654.
[100] Li, Z. P.; Yu, R.; Li, H. J. Angew. Chem., Int. Ed. 2008, 47, 7497.
[101] Li, H.; He, Z.; Guo, X.; Li, W.; Zhao, X.; Li, Z. Org. Lett. 2009, 11, 4176.
[102] Liu, P.; Wang, Z. M.; Lin, J.; Hu, X. M. Eur. J. Org. Chem. 2012, 1583.
[103] For Selected literatures on the synthesis of amino acid derivatives:
(a) Beak, P.; Zajdel, W. J.; Reitz, D. B. Chem. Rev. 1984, 84, 471.
(b) Knowles, H. S.; Hunt, K.; Parsons, A. F. Tetrahedron Lett. 2000, 41, 7121.
(c) Burger, K.; Geith, K.; Gaa, K. Angew. Chem., Int. Ed. Engl. 1988, 27, 848.
(d) Ireland, R. E.; Mueller, R. H. Willard, A. K. J. Am. Chem. Soc. 1976, 98, 2868;
[104] Yoshikai, N.; Mieczkowski, A.; Matsumoto, A.; Ilies, L.; Nakamura, E. J. Am. Chem. Soc. 2010, 132, 5568.
[105] Qian, B.; Xie, P.; Xie, Y. J.; Huang, H. M. Org. Lett. 2011, 13, 2580.
[106] Volla, C. M. R.; Vogel, P. Org. Lett. 2009, 11, 1701.
[107] Chandrasekharam, M.; Chiranjeevi, B. P.; Gupta, K. S. V.; Sridhar, B. J. Org. Chem. 2011, 76, 10229.
[108] Guo, X.; Yu, R.; Li, H.; Li, Z. J. Am. Chem. Soc. 2009, 131,17387.
[109] For recent reviews, see:
(a) Davies, H. M. L.; Manning, J. R. Nature 2008, 451, 417.
(b) Giri, R.; Shi, B. F.; Engle, K. M.; Maugel, N.; Yu, J.-Q. Chem. Soc. Rev. 2009, 38, 3242.
(c) Doyle, M. P.; Duffy, R.; Ratnikov, M.; Zhou, L. Chem. Rev. 2010, 110, 704.
[110] Zhu, S.-F.; Zhou, Q.-L. Natl. Sci. Rev. 2014, 1, 580.
[111] Li, Y.; Huang, J.-S.; Zhou, Z.-Y.; Che, C.-M.; You, X.-Z. J. Am. Chem. Soc. 2002, 124,13185.
[112] Mbuvi, H. M.; Woo, L. K. Organometallics 2008, 27, 637.
[113] Cai, Y.; Zhou, S.-F.; Wang, G.-P.; Zhou, Q.-L. Adv. Synth. Catal. 2011, 353, 2939.
[114] Yang, J.-M.; Cai, Y.; Zhu, S.-F.; Zhou, Q.-L. Org. Biomol. Chem. 2016, 14, 5516.
/
| 〈 |
|
〉 |