

基于光驱动水氧化的有机底物氧化
收稿日期: 2017-09-05
修回日期: 2017-10-12
网络出版日期: 2017-11-03
基金资助
国家自然科学基金(No.21603046)资助项目.
Oxygenation of Organic Substrates Based on Light-Driven Water Oxidation
Received date: 2017-09-05
Revised date: 2017-10-12
Online published: 2017-11-03
Supported by
Project supported by the National Natural Science Foundation of China (No. 21603046).
苗思文 , 那永 . 基于光驱动水氧化的有机底物氧化[J]. 有机化学, 2018 , 38(3) : 575 -584 . DOI: 10.6023/cjoc201709006
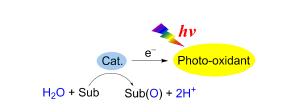
Hydrogen production by artificial photosynthetic water splitting is an efficient approach to convert solar ennergy into chemical bonds. Oxygenation of an organic substrate based on light-driven water oxidation is innovative way to mimic the oxygen evolving center (OEC) in Photosystem Ⅱ. The metal catalyst will accomplish H2O activation to generate high valent metal-oxo intermediate, which can transfer the oxygen atom to an organic substrate, during which the H atoms in H2O molecule could be released. This review is a perspective of the recent advances in oxygenation of organic substrates with water as oxygen source. In the meanwhile, research prospect on photocatalytic hydrogen production coupled with the photocatalytic oxygenation of an organic substrate for a new water splitting system has been proposed.
[1] Lewis, N. S.; Nocera, D. G. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 15729.
[2] Barber, J. Chem. Soc. Rev. 2009, 38, 185.
[3] Duan, L. L.; Wang, L.; Li, F. S.; Li, F.; Sun, L. Acc. Chem. Res. 2015, 48, 2084.
[4] Shen, J. R. Annu. Rev. Plant Biol. 2015, 66, 23.
[5] Cox, N.; Retegan, M.; Neese, F.; Pantazis, D. A.; Boussac, A.; Lubitz, W. Science 2014, 345, 804.
[6] Groves, J. T. Nat. Chem. 2014, 6, 89.
[7] Oloo, W. N.; Que Jr., L. Acc. Chem. Res. 2015, 48, 2612.
[8] Nam, W.; Lee, Y. M.; Fukuzumi, S. Acc. Chem. Res. 2014, 47, 1146.
[9] Fukuzumi, S.; Ohkubo, K.; Lee, Y. M.; Nam, W. Chem.-Eur. J. 2015, 21, 17548.
[10] Moyer, B. A.; Meyer, T. J. J. Am. Chem. Soc. 1978, 100, 3601.
[11] Wasylenko, D. J.; Ganesamoorthy, C.; Henderson, M. A.; Berlinguette, C. P. Inorg. Chem. 2011, 50, 3662.
[12] Duan, L.; Tong, L.; Xu, Y.; Sun, L. Energy Environ. Sci. 2011, 4, 3296.
[13] Funyu, S.; Isobe, T.; Takagi, S.; Tryk, D. A.; Inoue, H. J. Am. Chem. Soc. 2003, 125,5734.
[14] Funyu, S.; Kinai, M.; Masui, D.; Takagi, S.; Shimada, T.; Tachibanaa, H.; Inoue, H. Photochem. Photobiol. Sci. 2010, 9, 931.
[15] Kurimoto, K.; Yamazaki, T.; Suzuri, Y.; Nabetani, Y.; Onuki, S.; Takagi, S.; Shimada, T.; Tachibana, H.; Inoue, H. Photochem. Photobiol. Sci. 2014, 13, 154.
[16] Fukuzumi, S.; Kishi, T.; Kotani, H.; Lee, Y. M.; Nam, W. Nat. Chem. 2011, 3, 38.
[17] Fukuzumi, S.; Mizuno, T.; Ojiri, T. Chem.-Eur. J. 2012, 18, 15794.
[18] Treadway, J. A.; Moss, J. A.; Meyer, T. J. Inorg. Chem. 1999, 38, 4386.
[19] Hirai, Y.; Kojima, T.; Mizutani, Y.; Shiota, Y.; Yoshizawa, K.; Fukuzumi, S. Angew. Chem., Int. Ed. 2008, 47, 5772.
[20] Ohzu, S.; Ishizuka, T.; Hirai, Y.; Jiang, H.; Sakaguchi, M.; Ogura, T.; Fukuzumi, S.; Kojima, T. Chem. Sci. 2012, 3, 3421.
[21] Kalita, D.; Radaram, B.; Brooks, B.; Kannam, P. P.; Zhao, X. ChemCatChem 2011, 3, 571.
[22] Ohzu, S.; Ishizuka, T.; Hirai, Y.; Fukuzumi, S.; Kojima, T. Chem.-Eur. J. 2013, 19, 1563.
[23] Singh, W. M.; Pegram, D.; Duan, H. F.; Kalita, D.; Simone, P.; Emmert, G. L.; Zhao, X. Angew. Chem., Int. Ed. 2012, 51, 1653.
[24] Giovanni, C. D.; Poater, A.; Benet-Buchholz, J.; Cavallo, L.; Solà, M.; Llobet, A. Chem.-Eur. J. 2014, 20, 3898.
[25] Farràs, P.; Giovanni, C. D.; Clifford, J. N.; Garrido-Barros, P.; Palomares, E.; Llobet, A. Green Chem. 2016, 18, 255.
[26] Li, F.; Yu, M.; Jiang, Y.; Huang, F.; Li, Y. Q.; Zhang, B.; Sun, L. Chem. Commun. 2011, 47, 8949.
[27] Zhou, X.; Li, F.; Li, X.; Li, H.; Wang, Y.; Sun, L. Dalton Trans. 2015, 44, 475.
[28] Bai, L.; Li, F.; Wang, Y.; Li, H.; Jiang, X.; Sun, L. Chem. Commun. 2016, 52, 9711.
[29] Hamelin, O.; Guillo, P.; Loiseau, F.; Boissonnet, M.; Ménage, S. Inorg. Chem. 2011, 50, 7952.
[30] Guillo, P.; Hamelin, O.; Batat, P.; Jonusauskas, G.; McClenaghan, N. D.; Ménage, S. Inorg. Chem. 2012, 51, 2222.
[31] Li, T. T.; Li, F. M.; Zhao, W. L.; Tian, Y. H.; Chen, Y.; Cai, R.; Fu, W. F. Inorg. Chem. 2015, 54, 183.
[32] Phungsripheng, S.; Kozawa, K.; Akita, M.; Inagaki, A. Inorg. Chem. 2016, 55, 3750.
[33] Lee, Y. M.; Dhuri, S. N.; Sawant, S. C.; Cho, J.; Kubo, M.; Ogura, T.; Fukuzumi, S.; Nam, W. Angew. Chem., Int. Ed. 2009, 48, 1803.
[34] Kotani, H.; Suenobu, T.; Lee, Y. M.; Nam, W.; Fukuzumi, S. J. Am. Chem. Soc. 2011, 133, 3249.
[35] Company, A; Sabenya, G.; González-Béjar, M; Gómez, L; Clémancey, M; Blondin, G; Jasniewski, A. J.; Puri, M; Browne, W. R; Latour, J.; Que Jr., L.; Costas, M.; Pérez-Prieto, J.; Lloret-Fillol, J. J. Am. Chem. Soc. 2014, 136, 4624.
[36] Chantarojsiri, T.; Sun, Y.; Long, J. R.; Chang, C. J. Inorg. Chem. 2015, 54, 5879.
[37] Herrero, C.; Quaranta, A.; Sircoglou, M.; Sénéchal-David, K.; Baron, A.; Marín, I. M.; Buron, C.; Baltaze, J.; Leibl, W.; Aukauloo, A.; Banse, F. Chem. Sci. 2015, 6, 2323.
[38] Sawant, S. C.; Wu, X.; Cho, J.; Cho, K.; Kim, S. H.; Seo, M. S.; Lee, Y. M.; Kubo, M.; Ogura, T.; Shaik, S.; Nam, W. Angew. Chem., Int. Ed. 2010, 49, 8190.
[39] Wu, X.; Yang, X.; Lee, Y. M.; Nam, W.; Sun, L. Chem. Commun. 2015, 51, 4013.
[40] Shen, D.; Saracini, C.; Lee, Y. M.; Sun, W.; Fukuzumi, S.; Nam, W. J. Am. Chem. Soc. 2016, 138, 15857.
/
| 〈 |
|
〉 |