

新型白藜芦醇-查尔酮酰胺衍生物的合成及其细胞毒活性
收稿日期: 2017-08-17
修回日期: 2017-10-25
网络出版日期: 2017-11-17
基金资助
国家自然科学基金(Nos.81560620,81460624)、云南省应用基础研究(No.2014FZ078)和云南省科学技术厅-云南中医学院应用基础研究联合专项(No.2017FF117(-023))资助项目.
Synthesis and Cytotoxic Activity of Novel Resveratrol-Chalcone Amide Derivatives
Received date: 2017-08-17
Revised date: 2017-10-25
Online published: 2017-11-17
Supported by
Project supported by the National Natural Science Foundation of China (Nos. 81560620, 81460624), the Application Basic Research Program of Yunnan Province (No. 2014FZ078) and the Yunnan Provincial Science and Technology Department-Applied Basic Research Joint Special Funds of Yunnan University of Traditional Chinese Medicine (No. 2017FF117(-023)).
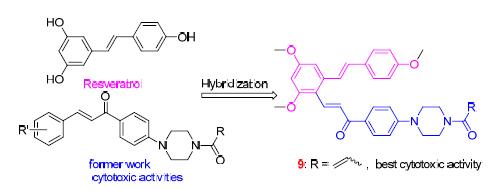
白藜芦醇又名芪三酚,是一种生物活性很强的茋类化合物.在前期研究基础上,采用活性亚结构拼接的方法,将白藜芦醇与查耳酮片段连接起来,以寻找活性较高的新型抗肿瘤分子.白藜芦醇经Vilsmeier甲酰化、Aldol缩合、哌嗪取代及酰胺化反应,设计合成了13个未见文献报道的白藜芦醇-查尔酮衍生物,其结构经IR、1H NMR、13C NMR和HRMS确证.采用噻唑蓝(MTT)法对目标化合物的体外细胞毒活性进行测试,结果表明,该类化合物具有较强的体外细胞毒活性,特别是(E)-3-(2,4-二甲氧基-6-((E)-4-甲氧基苯乙烯基)苯基-1-(4-(N-丙烯酰基)哌嗪-1-基)苯基丙烷-2-烯-1-酮(9)对肿瘤细胞株(A549和Hela)均表现出良好的体外细胞毒活性(IC50值分别为0.26和7.35 μmol·L-1).并且,流式细胞技术分析表明化合物9能够显著诱导肿瘤细胞A549的凋亡.
高慧 , 郑喜 , 祁燕 , 王斯 , 万春平 , 饶高雄 , 毛泽伟 . 新型白藜芦醇-查尔酮酰胺衍生物的合成及其细胞毒活性[J]. 有机化学, 2018 , 38(3) : 648 -655 . DOI: 10.6023/cjoc201708031
Resveratrol is a type of natural phenol with a broad range of good biological activities. Based on former work, in order to look for new anticancer agents, a series of novel amide compounds between resveratrol and chalcone possessing piperazine moiety have been synthesized by connecting principle of active biological groups. The structures of compounds were characterized by IR, 1H NMR, 13C NMR and HRMS, and in vitro cytotoxic activity was evaluated against a panel of human tumor cell lines (Hela, A549 and SGC7901) by the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. The results demonstrated that amide compounds contributed good cytotoxic activity. Especially, (E)-3-(2,4-dimethoxy-6-((E)-4-methoxystyryl)phenyl)-1-(4-(N-acryloyl)piperazin-1-yl)phenylprop-2-en-1-one (9) showed the best cytotoxic activity against A549 and Hela (IC50=0.26 and 7.35 μmol·L-1, respectively), and fluorescence-activated cell sorter (FACs) analysis showed that compound 9 significantly induced apoptosis in A549 cell.

Key words: resveratrol-chalcone; amide derivatives; synthesis; cytotoxic activity
[1] Xue, S. T.; Guo, H. F.; Liu, M. J.; Jin, J.; Ju, D. H.; Liu, Z. Y.; Li, Z. R. Eur. J. Med. Chem. 2015, 96, 151.
[2] Hayashi, S.; Ueno, N.; Murase, A.; Nakagawa, Y.; Takada, J. Eur. J. Med. Chem. 2012, 50, 179.
[3] Baur, J. A.; Sinclair, D. A. Nat. Rev. Drug Discovery 2006, 5, 493.
[4] Nivelle, L.; Hubert, J.; Courot, E.; Jeandet, P.; Aziz, A.; Nuzillard, J. M.; Renault, J. H.; Clément, C.; Martiny, L.; Delmas, D.; Tarpin, M. Molecules 2017, 22, 474.
[5] Du, C.; Ren, Y. J.; Wang, Q. W.; Jin, L. Chin. J. Org. Chem. 2013, 33, 1279(in Chinese). (杜成, 任玉杰, 王庆伟, 金鹭, 有机化学, 2013, 33, 1279.)
[6] Ferraz da Costa, D. C.; Fialho, E.; Silva, J. L. Molecules 2017, 22, 1014.
[7] Duan, Y. C.; Guan, Y. Y.; Zhai, X. Y.; Ding, L. N.; Qin, W. P.; Shen, D. D.; Liu, X. Q.; Sun, X. D.; Zheng, Y. C.; Liu, H. M. Eur. J. Med. Chem. 2017, 126, 246.
[8] Chen, E.; Ge, X.; Xu, F.; Zhang, Y.; Li, Z.; Pan, J.; Song, J.; Dai, Y.; Zhou, J.; Feng, J.; Liang, G. Bioorg. Med. Chem. Lett. 2015, 25, 2998.
[9] Huang, X. F.; Ruan, B. F.; Wang, X. T.; Xu, C.; Ge, H. M.; Zhu, H. L.; Tan, R. X. Eur. J. Med. Chem. 2007, 42, 263.
[10] Zhang, E.; Wang, M. M.; Xu, S. M.; Wang, S.; Zhao, D.; Bai, P. Y.; Cui, D. Y.; Hua, Y. G.; Wang, Y. N.; Qin, S. S.; Liu, H. M. Chin. J. Org. Chem. 2017, 37, 959(in Chinese). (张恩, 王铭铭, 徐帅民, 王上, 赵娣, 白鹏燕, 催得运, 化永刚, 王亚娜, 秦上尚, 刘宏民, 有机化学, 2017, 37, 959.)
[11] Kamal, A.; Ramakrishna, G.; Raju, P.; Viswanath, A.; Ramaiah, M. J.; Balakishan, G.; Pal-Bhadra, M. Bioorg. Med. Chem. Lett. 2010, 20, 4865.
[12] Wei, Z. Y.; Chi, K. Q.; Yu, Z. K.; Liu, H. Y.; Sun, L. P.; Zheng, C. J.; Piao, H. R. Bioorg. Med. Chem. Lett. 2016, 26, 5920.
[13] Mao, Z. W.; Zheng, X.; Lin, Y. P.; Qi, Y.; Hu, C. Y.; Wan, C. P.; Rao, G. X. Heterocycles 2016, 92, 1102.
[14] Mao, Z. W.; Zheng, X.; Qi, Y.; Zhang, M. D.; Huang, Y.; Wan, C. P.; Rao, G. X. RSC Adv. 2016, 6, 7723.
[15] Ma, Y.; Zheng, X.; Gao, H.; Wan, C.; Rao, G.; Mao, Z. Molecules 2016, 21, 1684.
[16] Lin, Y. P.; Hu, C. Y.; Zheng, X.; Wang, X. L.; Wan, C. P.; Mao, Z. W. Chin. J. Org. Chem. 2017, 37, 237(in Chinese). (林玉萍, 虎春艳, 郑喜, 王秀丽, 万春平, 毛泽伟, 有机化学, 2017, 37, 237.)
[17] Ruan, B. F.; Cheng, H. J.; Ren, J.; Li, H. L.; Guo, L. L.; Zhang, X. X.; Liao, C. Z. Eur. J. Med. Chem. 2015, 103, 185.
/
| 〈 |
|
〉 |