

电化学条件下喹喔啉-2(1H)-酮C(3)—H功能化的研究进展
收稿日期: 2021-07-18
修回日期: 2021-09-04
网络出版日期: 2021-09-15
基金资助
国家自然科学基金(21901219); 河南教育厅重点项目(20B150024); 2021年度河南省高等教育青年骨干教师培养计划(2021GGJS146); 河南省药品质量控制与评价重点实验室(郑州大学)开放课题科研基金和许昌学院杰出青年骨干资助项目
Recent Advances in Electrochemical C(3)—H Functionalization of Quinoxalin-2(1H)-ones
Received date: 2021-07-18
Revised date: 2021-09-04
Online published: 2021-09-15
Supported by
National Natural Science Foundation of China(21901219); Key Project of Education Department of Henan Province(20B150024); Young Key Teacher Training Plan of Henan Provincial Higher Education in 2021(2021GGJS146); Open Subject Research Fund of the Key Laboratory of Drug Quality Control and Evaluation in Henan Province and the Young Backbone Teachers Project of Xuchang University
喹喔啉酮及其衍生物是一类重要的氮杂环化合物, 广泛存在于具有各种生物活性的天然产物、药物及功能材料中, 因此, 喹喔啉酮的C—H功能化引起了化学工作者的广泛关注. 电化学合成无需外加氧化还原试剂, 直接利用电子这一“清洁试剂”参与氧化还原反应, 具有反应条件温和及原子经济性好等特点, 十分符合绿色化学符合绿色化学和可持续发展的要求. 随着对电化学合成反应机理的深入研究以及反应设备的标准化, 该方法已经成为喹喔啉酮类化合物功能化的强有力工具. 综述了有机电化学在喹喔啉-2(1H)-酮C(3)—H功能化领域所取得的研究进展, 对反应的条件和机理进行了系统的总结, 并对该领域所面临的挑战及发展方向进行了展望与探讨.
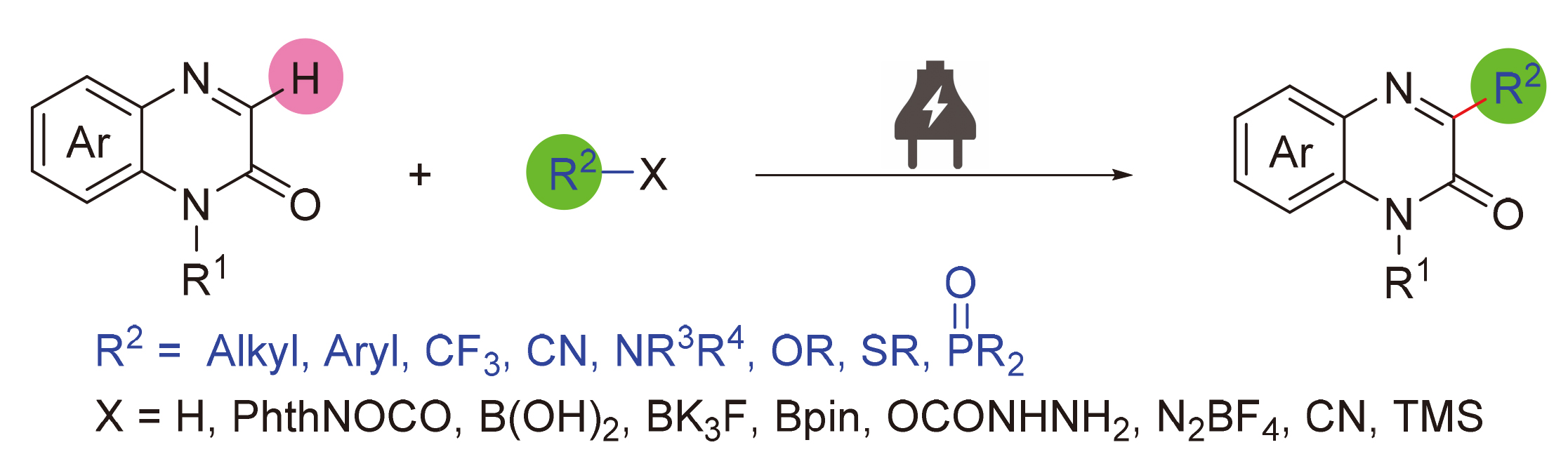
关键词: 电化学; 喹喔啉-2(1H)-酮; C(3)—H功能化
李丹丹 , 王晓辰 , 李闪闪 , 付晨雨 , 李倩倩 , 许东涛 , 马莹莹 . 电化学条件下喹喔啉-2(1H)-酮C(3)—H功能化的研究进展[J]. 有机化学, 2021 , 41(12) : 4610 -4622 . DOI: 10.6023/cjoc202107042
Quinoxalinone and its derivatives are a class of important nitrogen heterocyclic compounds, which widely exist in natural products, drugs and functional materials. Therefore, the C—H functionalization of quinoxalinone has attracted extensive attention of chemical workers. In recent years, electrochemical synthesis directly uses electron as a “clean reagent” to participate in redox reaction without additional redox reagent, which has the characteristics of mild reaction conditions and good atom economy. This method meets the requirements of green chemistry and sustainable development. With the in-depth study on the mechanism of electrochemical synthesis and the standardization of reaction equipment, this method has become a powerful tool for functionalization of quinoxalineones. The recent advances in the electrochemical C(3)—H functionalization of quinoxaline-2(1H)-one are summarized. The reaction transformation conditions and mechanisms are systematically discussed, and the challenges and future directions of this field are included.

| [1] | (a) Waring, M. J.; Ben-Hadda, T.; Kotchevar, A. T.; Ramdani, A.; Touzani, R.; Elkadiri, S.; Hakkou, A.; Bouakka, M.; Ellis, T. Molecules 2002, 7, 641. |
| [1] | (b) Horton, D. A.; Bourne, G. T.; Smythe, M. L. Chem. Rev. 2003, 103, 893. |
| [1] | (c) Refaat, H. M.; Moneer, A. A.; Khalil, O. M. Arch. Pharm. Res. 2004, 27, 1093. |
| [1] | (d) Carta, A.; Piras, S.; Loriga, G.; Paglietti, G. Mini-Rev. Med. Chem. 2006, 6, 1179. |
| [1] | (e) El-Hawash, S. A. M.; Habib, N. S.; Kassem, M. A. Arch. Pharm. 2006, 339, 564. |
| [1] | (f) Meyer, E.; Joussef, A. C.; de Souza, L. D. B. P. Synth. Commun. 2006, 36, 729. |
| [1] | (g) Moarbess, G.; Deleuze-Masquefa, C.; Bonnard, V.; Gayraud- Paniagua, S.; Vidal, J.-R.; Bressolle, F.; Pinguet, F.; Bonnet, P.-A. Bioorg. Med. Chem. 2008, 16, 6601. |
| [1] | (h) Ramli, Y.; Benzeid, H.; Bouhfid, R.; Rodi, Y. K.; Ferfra, S.; Essassi, E. M. Stud. Cercet. Stiint.: Chim. Ing. Chim., Biotehnol., nd. Aliment. 2010, 11, 67. |
| [1] | (i) Liu, R.; Huang, Z.; Murray, M. G.; Guo, X.; Liu, G. J. Med. Chem. 2011, 54, 5747. |
| [1] | (j) Galal, S. A.; Khairat, S. H. M.; Ragab, F. A. F.; Abdelsamie, A. S.; Ali, M. M.; Soliman, S. M.; Mortier, J.; Wolber, G.; El Diwani, H. I. Eur. J. Med. Chem. 2014, 86, 122. |
| [1] | (k) Hussain, S.; Parveen, S.; Hao, X.; Zhang, S.; Wang, W.; Qin, X.; Yang, Y.; Chen, X.; Zhu, S.; Zhu, C.; Ma, B. Eur. J. Med. Chem. 2014, 80, 383. |
| [1] | (l) Khattab, S. N.; Abdel Moneim, S. A. H.; Bekhit, A. A.; El Massry, A. M.; Hassan, S. Y.; El-Faham, A.; Ali Ahmed, H. E.; Amer, A. Eur. J. Med. Chem. 2015, 93, 308. |
| [1] | (m) Qin, X.; Hao, X.; Han, H.; Zhu, S.; Yang, Y.; Wu, B.; Hussain, S.; Parveen, S.; Jing, C.; Ma, B.; Zhu, C. J. Med. Chem. 2015, 58, 1254. |
| [1] | (n) Cil, O.; Phuan, P.-W.; Lee, S.; Tan, J.; Haggie, P. M.; Levin, M. H.; Sun, L.; Thiagarajah, J. R.; Ma, T.; Verkman, A. S. Cell Mol. Gastroenterol. Hepatol. 2016, 2, 317. |
| [1] | (o) Shi, L.; Li, X.; Hu, W.; Wu, J.; Zhou, H.; Zhou, H. Mini-Rev. Med. Chem. 2018, 18, 392. |
| [2] | Weïwer, M.; Spoonamore, J.; Wei, J.; Guichard, B.; Ross, N. T.; Masson, K.; Silkworth, W.; Dandapani, S.; Palmer, M.; Scherer, C. A.; Stern, A. M.; Schreiber, S. L.; Munoz, B. ACS Med. Chem. Lett. 2012, 3, 1034. |
| [3] | (a) Smits, R. A.; Lim, H. D.; Hanzer, A.; Zuiderveld, O. P.; Guaita, E.; Adami, M.; Coruzzi, G.; Leurs, R.; de Esch, I. J. P. J. Med. Chem. 2008, 51, 2457. |
| [3] | (b) Abu-Hashem, A. A.; Gouda, M. A.; Badria, F. A. Eur. J. Med. Chem. 2010, 45, 1976. |
| [3] | (c) Mangold, S. L.; Prost, L. R.; Kiessling, L. L. Chem. Sci. 2012, 3, 772. |
| [4] | Quinn, J.; Guo, C.; Ko, L.; Sun, B.; He, Y.; Li, Y. RSC Adv. 2016, 6, 22043. |
| [5] | (a) Xie, L.-Y.; Bai, Y.-S.; Xu, X.-Q.; Peng, X.; Tang, H.-S.; Huang, Y.; Lin, Y.-W.; Cao, Z.; He, W.-M. Green Chem. 2020, 22, 1720. |
| [5] | (b) Wang, L.; Bao, P.; Liu, W.; Liu, S.; Hu, C.; Yue, H.; Yang, D.; Wei, W. Chin. J. Org. Chem. 2018, 38, 3189. (in Chinese) |
| [5] | ( 王雷雷, 鲍鹏丽, 刘维伟, 刘思彤, 胡昌松, 岳会兰, 杨道山, 魏伟, 有机化学, 2018, 38, 3189.) |
| [5] | (c) Wu, Y.; Chen, J.-Y.; Ning, J.; Jiang, X.; Deng, J.; Deng, Y.; Xu, R.; He, W.-M. Green Chem. 2021, 23, 3950. |
| [5] | (d) Shi, J.; Wei, W. Chin. J. Org. Chem. 2020, 40, 2170. (in Chinese) |
| [5] | ( 时建伟, 魏伟, 有机化学, 2020, 40, 2170.) |
| [5] | (e) Mao, P.; Zhu, J.; Yuan, J.; Yang, L.; Xiao, Y.; Zhang, C. Chin. J. Org. Chem. 2019, 39, 1529. (in Chinese) |
| [5] | ( 毛璞, 朱军亮, 袁金伟, 杨亮茹, 肖咏梅, 张长森, 有机化学, 2019, 39, 1529.) |
| [5] | (f) Xie, L.-Y.; Peng, S.; Yang, L.-H.; Peng, C.; Lin, Y.-W.; Yu, X.; Cao, Z.; Peng, Y.-Y.; He, W.-M. Green Chem. 2021, 23, 374. |
| [5] | (g) Yi, R.; He, W. Chin. J. Org. Chem. 2021, 41, 1267. (in Chinese) |
| [5] | ( 易荣楠, 何卫民, 有机化学, 2021, 41, 1267.) |
| [5] | (h) Xie, L.-Y.; Liu, Y.-S.; Ding, H.-R.; Gong, S.-F.; Tan, J.-X.; He, J.-Y.; Cao, Z.; He, W.-M. Chin. J. Catal. 2020, 41, 1168. |
| [5] | (i) Chen, J.-Y.; Wu, H.-Y.; Gui, Q.-W.; Yan, S.-S.; Deng, J.; Lin, Y.-W.; Cao, Z.; He, W.-M. Chin. J. Catal. 2021, 42, 1445. |
| [5] | (j) Ke, Q.; Yan, G.; Yu, J.; Wu, X. Org. Biomol. Chem. 2019, 17, 5863. |
| [5] | (k) Rostoll-Berenguer, J.; Blay, G.; Pedro, J. R.; Vila, C. Eur. J. Org. Chem. 2020, 2020, 6148. |
| [5] | (l) Monika, M.; Selvakumar, S. Synthesis 2019, 51, 4113. |
| [5] | (m) Ghosh, P.; Das, S. Synth. Commun. 2020, 50, 2266. |
| [6] | (a) Frontana-Uribe, B. A.; Little, R. D.; Ibanez, J. G.; Palma, A.; Vasquez-Medrano, R. Green Chem. 2010, 12, 2099. |
| [6] | (b) Jiang, Y.; Xu, K.; Zeng, C. Chem. Rev. 2018, 118, 4485. |
| [6] | (c) Jiao, K.-J.; Xing, Y.-K.; Yang, Q.-L.; Qiu, H.; Mei, T.-S. Acc. Chem. Res. 2020, 53, 300. |
| [6] | (d) Karkas, M. D. Chem. Soc. Rev. 2018, 47, 5786. |
| [6] | (e) Ma, C.; Fang, P.; Mei, T.-S. ACS Catal. 2018, 8, 7179. |
| [6] | (f) Maeda, H.; Ohmori, H. Acc. Chem. Res. 1999, 32, 72. |
| [6] | (g) Minteer, S. D.; Baran, P. Acc. Chem. Res. 2020, 53, 545. |
| [6] | (h) Möhle, S.; Zirbes, M.; Rodrigo, E.; Gieshoff, T.; Wiebe, A.; Waldvogel, S. R. Angew. Chem., nt. Ed. 2018, 57, 6018. |
| [6] | (i) Novaes, L. F. T.; Liu, J.; Shen, Y.; Lu, L.; Meinhardt, J. M.; Lin, S. Chem. Soc. Rev. 2021, 50, 7941. |
| [6] | (j) Röckl, J. L.; Pollok, D.; Franke, R.; Waldvogel, S. R. Acc. Chem. Res. 2020, 53, 45. |
| [6] | (k) Sauermann, N.; Meyer, T. H.; Qiu, Y.; Ackermann, L. ACS Catal. 2018, 8, 7086. |
| [6] | (l) Waldvogel, S. R.; Lips, S.; Selt, M.; Riehl, B.; Kampf, C. J. Chem. Rev. 2018, 118, 6706. |
| [6] | (m) Wang, H.; Gao, X.; Lv, Z.; Abdelilah, T.; Lei, A. Chem. Rev. 2019, 119, 6769. |
| [6] | (n) Yamamoto, K.; Kuriyama, M.; Onomura, O. Acc. Chem. Res. 2020, 53, 105. |
| [6] | (o) Yan, M.; Kawamata, Y.; Baran, P. S. Chem. Rev. 2017, 117, 13230. |
| [6] | (p) Yi, H.; Zhang, G.; Wang, H.; Huang, Z.; Wang, J.; Singh, A. K.; Lei, A. Chem. Rev. 2017, 117, 9016. |
| [6] | (q) Yuan, Y.; Lei, A. Acc. Chem. Res. 2019, 52, 3309. |
| [7] | Tan, Y.; Wang, J.; Zhang, H.-Y.; Zhang, Y.; Zhao, J. Front. Chem. 2020, 8, 582. |
| [8] | Lian, F.; Xu, K.; Meng, W.; Zhang, H.; Tan, Z.; Zeng, C. Chem. Commun. 2019, 55, 14685. |
| [9] | Niu, K.; Song, L.; Hao, Y.; Liu, Y.; Wang, Q. Chem. Commun. 2020, 56, 11673. |
| [10] | Traister, K. M.; Molander, G. A. Synthesis and Application of Organoboron Compounds, In Topics in Organometallic Chemistry, Vol. 49, Eds.: Fernández, E.; Whiting, A., Springer, Switzerland, 2015, pp. 117-151. |
| [11] | Niu, K.; Hao, Y.; Song, L.; Liu, Y.; Wang, Q. Green Chem. 2021, 23, 302. |
| [12] | Gao, Y.; Wu, Z.; Yu, L.; Wang, Y.; Pan, Y. Angew. Chem., nt. Ed. 2020, 59, 10859. |
| [13] | Mo, F.; Qiu, D.; Zhang, L.; Wang, J. Chem. Rev. 2021, 121, 5741. |
| [14] | Jiang, Y.-Y.; Dou, G.-Y.; Zhang, L.-S.; Xu, K.; Little, R. D.; Zeng, C.-C. Adv. Synth. Catal. 2019, 361, 5170. |
| [15] | Hu, C.; Hong, G.; Zhou, C.; Tang, Z.-C.; Han, J.-W.; Wang, L.-M. Asian J. Org. Chem. 2019, 8, 2092. |
| [16] | (a) Lehnherr, D.; Lam, Y.-H.; Nicastri, M. C.; Liu, J.; Newman, J. A.; Regalado, E. L.; DiRocco, D. A.; Rovis, T. J. Am. Chem. Soc. 2020, 142, 468. |
| [16] | (b) Nicastri, M. C.; Lehnherr, D.; Lam, Y.-H.; DiRocco, D. A.; Rovis, T. J. Am. Chem. Soc. 2020, 142, 987. |
| [16] | (c) Zhang, S.; Li, L.; Li, X.; Zhang, J.; Xu, K.; Li, G.; Findlater, M. Org. Lett. 2020, 22, 3570. |
| [16] | (d) Zhang, X.; Yang, C.; Gao, H.; Wang, L.; Guo, L.; Xia, W. Org. Lett. 2021, 23, 3472. |
| [17] | Wen, J.; Yang, X.; Yan, K.; Qin, H.; Ma, J.; Sun, X.; Yang, J.; Wang, H. Org. Lett. 2021, 23, 1081. |
| [18] | (a) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320. |
| [18] | (b) Nie, J.; Guo, H.-C.; Cahard, D.; Ma, J.-A. Chem. Rev. 2011, 111, 455. |
| [18] | (c) Tomashenko, O. A.; Grushin, V. V. Chem. Rev. 2011, 111, 4475. |
| [18] | (d) Liu, T.; Shen, Q. Eur. J. Org. Chem. 2012, 2012, 6679. |
| [19] | Dou, G.-Y.; Jiang, Y.-Y.; Xu, K.; Zeng, C.-C. Org. Chem. Front. 2019, 6, 2392. |
| [20] | (a) Scheuer, P. J. Acc. Chem. Res. 1992, 25, 433. |
| [20] | (b) Fleming, F. Nat. Prod. Rep. 1999, 16, 597. |
| [20] | (c) Fleming, F. F.; Yao, L.; Ravikumar, P. C.; Funk, L.; Shook, B. C. J. Med. Chem. 2010, 53, 7902. |
| [21] | (a) Larock, R. C. Comprehensive Organic Transformations, Wiley-VCH, New York, 1989, p. 819. |
| [21] | (b) Caboni, P.; Sammelson, R. E.; Casida, J. E. J. Agric. Food Chem. 2003, 51, 7055. |
| [21] | (c) Kuhn, P.; Thomas, A.; Antonietti, M. Macromolecules 2009, 42, 319. |
| [22] | (a) Liskey, C. W.; Liao, X.; Hartwig, J. F. J. Am. Chem. Soc. 2010, 132, 11389. |
| [22] | (b) Wade, J. R. Organic Chemistry, 8 th ed., Pearson Education Inc, Glenview, Illinois, USA, 2013. |
| [22] | (c) Zhang, S.; del Pozo, J.; Romiti, F.; Mu, Y.; Torker, S.; Hoveyda, A. H. Science 2019, 364, 45. |
| [23] | Zhan, Y.; Li, Y.; Tong, J.; Liu, P.; Sun, P. Eur. J. Org. Chem. 2021, 2021, 2193. |
| [24] | (a) Vitaku, E.; Smith, D. T.; Njardarson, J. T. J. Med. Chem. 2014, 57, 10257. |
| [24] | (b) Cernak, T.; Dykstra, K. D.; Tyagarajan, S.; Vachal, P.; Krska, S. W. Chem. Soc. Rev. 2016, 45, 546. |
| [25] | Li, K.-J.; Xu, K.; Liu, Y.-G.; Zeng, C.-C.; Sun, B.-G. Adv. Synth. Catal. 2019, 361, 1033. |
| [26] | Jiang, X.; Yang, L.; Ye, Z.; Du, X.; Fang, L.; Zhu, Y.; Chen, K.; Li, J.; Yu, C. Eur. J. Org. Chem. 2020, 2020, 1687. |
| [27] | Song, C.; Liu, K.; Dong, X.; Chiang, C.-W.; Lei, A. Synlett 2019, 30, 1149. |
| [28] | Zhou, J.; Li, Z.; Sun, Z.; Ren, Q.; Zhang, Q.; Li, H.; Li, J. J. Org. Chem. 2020, 85, 4365. |
| [29] | (a) Demmer, C. S.; Krogsgaard-Larsen, N.; Bunch, L. Chem. Rev. 2011, 111, 7981. |
| [29] | (b) Montchamp, J.-L. Acc. Chem. Res. 2014, 47, 77. |
| [29] | (c) Mady, M. F.; Kelland, M. A. Energy Fuels 2017, 31, 4603. |
| [29] | (d) Ntatsopoulos, V.; Macegoniuk, K.; Mucha, A.; Vassiliou, S.; Berlicki, Ł. Eur. J. Med. Chem. 2018, 159, 307. |
| [30] | Li, K.-J.; Jiang, Y.-Y.; Xu, K.; Zeng, C.-C.; Sun, B.-G. Green Chem. 2019, 21, 4412. |
/
| 〈 |
|
〉 |