

烯烃自由基胺硒化: β-氨基硒醚的简易合成
收稿日期: 2021-12-21
修回日期: 2022-01-28
网络出版日期: 2022-02-10
基金资助
国家自然科学基金(21801007)
Radical Aminoselenation of Styrenes: Facile Access to β-Amido-selenides
Received date: 2021-12-21
Revised date: 2022-01-28
Online published: 2022-02-10
Supported by
National Natural Science Foundation of China(21801007)
殷一樊 , 李晨 , 孙凯 , 刘颖杰 , 王薪 . 烯烃自由基胺硒化: β-氨基硒醚的简易合成[J]. 有机化学, 2022 , 42(5) : 1431 -1437 . DOI: 10.6023/cjoc202112028
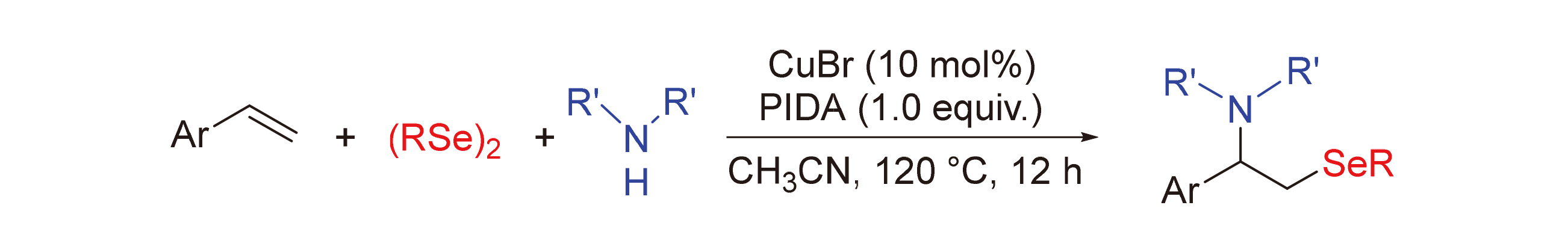
An efficient protocol for the intermolecular amidoselenation of alkenes with diphenyl diselenides and alkyl amines has been developed, affording a series of β-amido-selenides in high yields under copper salt and (diacetoxyiodo)benzene (PIDA) conditions. The preliminary experimental results supported the involvement of active selenide radical species, and a radical pathway was therefore proposed for the reaction.
Key words: amidoselenation; β-amido-selenide; active selenide; radical
| [1] | (a) Mugesh, G.; Du Mont, W. W.; Sies, H. Chem. Rev. 2001, 101, 2125. |
| [1] | (b) Nogueira, C. W.; Zeni, G.; Rocha, J. B. T. Chem. Rev. 2004, 104, 6255. |
| [1] | (c) Perin, G.; Lenardão, E. J.; Jacob, R. G.; Panatieri, R. B. Chem. Rev. 2009, 109, 1277. |
| [1] | (d) Bhabak, K. P.; Mugesh, G. Acc. Chem. Res. 2010, 43, 1408. |
| [1] | (e) Mukjerjee, A. J.; Zade, S. S.; Singh, H. B.; Sunoj, R. B. Chem. Rev. 2010, 110, 4357. |
| [1] | (f) Debnath, S.; Chithiravel, S.; Sharma, S.; Bedi, A.; Krishnamoorthy, K.; Zade, S. S. ACS Appl. Mater. Interfaces 2016, 8, 18222. |
| [1] | (g) Sun, K.; Wang, X.; Li, C.; Wang, H.; Li, L. Org. Chem. Front. 2020, 7, 3100. |
| [2] | (a) Kumar, A.; Rao, G. K.; Saleem, F.; Singh, A. K. Dalton Trans. 2012, 41, 11949. |
| [2] | (b) Ortgies, S.; Breder, A. ACS Catal. 2017, 7, 5828. |
| [3] | (a) Wang, X.-W.; Miao, Z.-H.; Ma, Y.; Chen, H.-J.; Qian, H.-S.; Zha, Z.-B. Nanoscale 2017, 9, 14512. |
| [3] | (b) Singh, F. V.; Wirth, T. Catal. Sci. Technol. 2019, 9, 1073. |
| [3] | (c) Shao, L. -, X.; Li, Y.; Lu, J.; Jiang, X. Org. Chem. Front. 2019, 6, 2999. |
| [3] | (d) Arora, A.; Oswal, P.; Kumar Rao, G.; Kumar, S.; Kumar, A. Dalton Trans. 2021, 50, 8628. |
| [4] | Selected works for asymmetric selenides synthesis with elemental selenium, diselenides, selenols, please see: (a) Kreft, A.; Ehlers, S.; Jones, P. G.; Werz, D. B. Org. Lett. 2015, 17, 2649. |
| [4] | (b) Iwasaki, M.; Miki, N.; Tsuchiya, Y.; Nakajima, K.; Nishihara, Y. Org. Lett. 2017, 19, 1092. |
| [4] | (c) Ma, W.-B.; Weng, Z.-Y.; Rogge, T.; Gu, L.-H.; Lin, J.-F.; Peng, A.; Luo, X.; Gou, X.-J.; Ackermann, L. Adv. Synth. Catal. 2018, 360, 704. |
| [4] | (d) Senol, E.; Scattolin, T.; Schoenebeck, F. Chem.-Eur. J. 2019, 25, 9419. |
| [4] | (e) Li, F.-H.; Wang, D.; Chen, H.-Y.; He, Z. Zhou, L.-H.; Zeng, Q.-L. Chem. Commun. 2020, 56, 13029. |
| [4] | (f) Cremer, C.; Eltester, M. A.; Bourakhouadar, H.; Atodiresei, I. L.; Patureau, F. W. Org. Lett. 2021, 23, 3243. |
| [5] | Álvarez-Pérez, M.; Ali, W.; Maŕc, M. A.; Handzlik, J.; Domínguez-Álvarez, E. Molecules 2018, 23, 628. |
| [6] | (a) Vieira, A. A.; Azeredo, J. B.; Godoi, M.; Santi, C.; Eufranio, N.; Braga, A. L. J. Org. Chem. 2015, 80, 2120. |
| [6] | (b) Zhang, Y.-K.; Wu, S.-X.; Yan, J. Helv. Chim. Acta. 2016, 99, 654. |
| [7] | (a) Toshimitsu, A.; Aoai, T.; Owada, H.; Uemura, S.; Okano, M. J. Org. Chem. 1981, 46, 4727. |
| [7] | (b) Sun, K.; Wang, X.; Lv, Y.-H.; Li, G.; Jiao, H.-Z.; Dai, C.-W.; Li, Y.-Y.; Zhang, C.; Liu, L. Chem. Commun. 2016, 52, 8471. |
| [7] | (c) Sun, K.; Wang, X.; Zhang, C.; Zhang, S.-F.; Chen, Y.; Jiao, H.-Z.; Du, W.-M. Chem.-Asian J. 2017, 12, 713. |
| [7] | (d) Wang, X.-L.; Li, H.-J.; Zhu, M.; Yan, J. RSC Adv. 2017, 7, 15709. |
| [7] | (e) Tan, Y.-X.; Liu, X.-Y.; Zhao, Y.-S.; Tian, P.; Lin, G.-Q. Org. Lett. 2019, 21, 1297. |
| [7] | (f) Huang, B.-B.; Li, Y.-N.; Yang, C.; Xia, W.-J. Green Chem. 2020, 22, 2804. |
| [7] | (g) Zhu, L.-L.; Tian, L.-F.; Cai, B.; Liu, G.-L.; Zhang, H.; Wang, Y.-H. Chem. Commun. 2020, 56, 2979. |
| [8] | (a) Back, T. G.; Collins, S. J. Org. Chem. 1981, 46, 3249. |
| [8] | (b) Sun, K.; Lv, Y.-H.; Shi, Z.-D.; Fu, F.-F.; Zhang, C.; Zhang, Z.-G. Sci. China: Chem. 2017, 60, 730. |
| [9] | (a) Feng, C.-L.; Zhu, J.; Tang, Q.-J.; Zhou, A.-H. Chin. J. Org. Chem. 2019, 39, 1187. (in Chinese) |
| [9] | (冯春来, 朱杰, 唐秋洁, 周爱华, 有机化学, 2019, 39, 1187.) |
| [9] | (b) Pan, C.; Liu, P.; Wu, A.-G.; Li, M.; Wen, L.-R.; Guo, W.-S. Chin. J. Org. Chem. 2020, 40, 2855. (in Chinese) |
| [9] | (潘超, 刘鹏, 武安国, 李明, 文丽荣, 郭维斯, 有机化学, 2020, 40, 2855.) |
| [9] | (c) Wu, Y.; Chen, J.-Y.; Ning, J.; Jiang, X.; Deng, J.; Deng, Y.; Xu, R.; He, W.-M. Green Chem. 2021, 23, 3950. |
| [9] | (d) Wang, S.-C.; Jiang, B. Chin. J. Org. Chem. 2021, 41, 4531. (in Chinese) |
| [9] | (王世超, 姜波, 有机化学, 2021, 41, 4531.) |
| [9] | (e) Xu, Y.; Li, C.; Meng, J.-P.; Huang, Y.-L.; Fu, J.-Y.; Liu, B.; Liu, Y.-J.; Chen, N. Chin. J. Org. Chem. 2021, 41, 1012. (in Chinese) |
| [9] | (许颖, 李晨, 孟建萍, 黄玉玲, 付纪源, 刘冰, 刘颖杰, 陈宁, 有机化学, 2021, 41, 1012.) |
| [9] | (f) Chen, J.-Y.; Wu, H.-Y.; Gui, Q.-W.; Yan, S.-S.; Deng, Jie.; Lin, Y.-W.; Cao, Z.; He, W.-M. Chin. J. Catal. 2021, 42, 1445. |
| [9] | (g) Wu, Z.-L.; Chen, J.-Y.; Tian, X.-Z.; Ouyang, W.-T.; Zhang, Z.-T.; He, W.-M. Chin. Chem. Lett. 2021, DOI: 10.1016/j.cclet.2021.08.071. |
| [10] | (a) Sun, K.; Shi, Z.-D.; Liu, Z.-H.; Luan, B.-X.; Zhu, J.-L.; Xue, Y.-R. Org. Lett. 2018, 20, 6687. |
| [10] | (b) Sun, K.; Wang, S.-N.; Feng, R.-R.; Zhang, Y.-X.; Wang, X.; Zhang, Z.-G.; Zhang, B. Org. Lett. 2019, 21, 2052. |
| [10] | (c) Wang, X.; Lei, J.; Guo, S.; Zhang, Y.; Ye, Y.; Tang, S.; Sun, K. Chem. Commun. 2022, 58, 1526. |
| [10] | (d) Wang, X.; Wang, Q.-L.; Xue, Y.-R.; Sun, K.; Wu, L.-L.; Zhang, B. Chem. Commun. 2020, 56, 4436. |
| [10] | (e) Wang, X.; Guo, S.; Zhang, Y.; Zhang, Z.-G.; Zhang, G.-S.; Ye, Y.; Sun, K. Adv. Synth. Catal. 2021, 363, 3290. |
| [11] | (a) Wang, A.-F.; Zhu, L.-Y.; Wang, S.-L.; Hao, W.-J.; Li, G.-G.; Tu, S.-J.; Jiang, B. J. Org. Chem. 2016, 81, 1099. |
| [11] | (b) Yang, D.-S.; Li, G.-Q.; Xing, C.-Y.; Cui, W.-W.; Li, K.-X.; Wei, W. Org. Chem. Front. 2018, 5, 2974. |
| [11] | (c) Guo, Y.-H.; Xiang, Y.-F.; Wei, L.; Wan, J.-P. Org. Lett. 2018, 20, 3971. |
| [11] | (d) Liu, Y.; Chen, X.-L.; Sun, K.; Li, X.-Y.; Zeng, F.-L.; Liu, X.-C.; Qu, L.-B.; Zhao, Y.-F.; Yu, B. Org. Lett. 2019, 21, 4019. |
| [11] | (e) Li, G.-Q.; Gan, Z.-Y.; Kong, K.-X.; Dou, X.-M.; Yang, D.-S. Adv. Synth. Catal. 2019, 361, 1808. |
| [11] | (f) Song, S.-Z.; Meng, Y.-N.; Li, Q.; Wei, W.-T. Adv. Synth. Catal. 2020, 362, 2120. |
| [11] | (g) Wang, X.-Y.; Zhong, Y.-F.; Mo, Z.-Y.; Wu, S.-H.; Xu, Y.-L.; Tang, H.-T.; Pan, Y.-M. Adv. Synth. Catal. 2021, 363, 208. |
| [12] | (a) Song, Z.-Q.; Ding, C.-C.; Wang, S.-L.; Dai, Q.; Sheng, Y.-G.; Zheng, Z.-L.; Liang, G. Chem. Commun. 2020, 56, 1847. |
| [12] | (b) Ghosh, P.; Chhetri, G.; Perl, E.; Das, S. Adv. Synth. Catal. 2021, 363, 2148. |
| [12] | (c) Lear, J. M.; Buquoi, J. Q.; Gu, X.; Pan, K.; Mustafa, D. N.; Nagib, D. A. Chem. Commun. 2019, 55, 8820. |
/
| 〈 |
|
〉 |