

可见光诱导下喹喔啉酮与吡咯衍生物的氧化偶联
收稿日期: 2022-07-25
修回日期: 2022-09-15
网络出版日期: 2022-10-25
基金资助
浙江省自然科学基金(LZ22B020003); 国家自然科学基金(22071171)
Visible-Light-Induced Regio-selective Oxidative Coupling of Quinoxalinones with Pyrrole Derivatives
Received date: 2022-07-25
Revised date: 2022-09-15
Online published: 2022-10-25
Supported by
Natural Science Foundation of Zhejiang Province(LZ22B020003); National Natural Science Foundation of China(22071171)
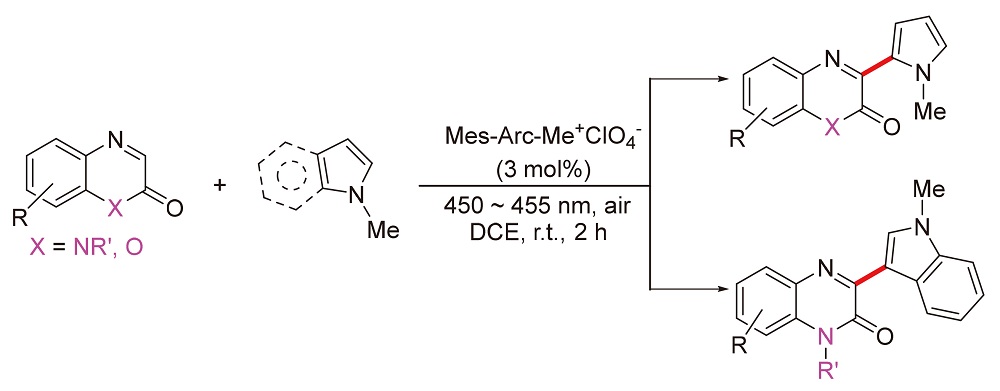
发展了一种区域选择性的喹喔啉酮3-位氧化偶联富电子吡咯衍生物的方法. 以$\text{Mes-Acr-M}{{\text{e}}^{\text{+}}}\text{ClO}_{\text{4}}^{-}$(3 mol%)为光敏剂, 空气氧为氧化剂, 高收率得到喹喔酮与吡咯衍生物的氧化偶联产物. 该方法底物的官能团适用范围广, 反应高效绿色, 可快速构建基于喹喔啉酮-吡咯衍生物的药物分子库. 相同测试条件下的Stern-Volmer荧光淬灭实验表明, 缺电子芳烃1-甲基喹喔啉酮对光敏剂的淬灭速率常数Kq=1.2×109 L•mol-1•s-1, 而富电子芳烃1-甲基吡咯/1-甲基吲哚的淬灭常数约为1.06~1.07×1010 L•mol-1•s-1, 后者的荧光淬灭速率几乎是前者的10倍.
沈梦涵 , 李来强 , 周泉 , 王洁慧 , 王磊 . 可见光诱导下喹喔啉酮与吡咯衍生物的氧化偶联[J]. 有机化学, 2023 , 43(2) : 697 -704 . DOI: 10.6023/cjoc202207031
Herein, one feasible methodology has been developed for oxidative cross-coupling between quinoxalinones and electron-rich aromatic rings (pyrroles or indoles). Utilizing the commerial available photocatalyst $\text{Mes-Acr-M}{{\text{e}}^{\text{+}}}\text{ClO}_{\text{4}}^{-}$ (3 mol%) as catalyst and air as external oxidant, the oxidative coupling products of quinolones and pyrrole derivatives were obtained in high yields. The protocol provides effective acceess to such quinoxalinones-pyrrole derivatives with profiles of broad functional group, efficient conversion and easy-handling. Furthermore, Stern-Volmer fluorescent quenching experiments under the same condition disclosed that quenching rate constant of electron-rich aromatic ring (pyrrole or indole, kq 1.06×1010~1.07×1010 L•mol-1•s-1) was nearly one order of magnitude larger than that of electro-deficient quinoxalinones (kq=1.2×109 L•mol-1•s-1).

| [1] | (a) Han, W. B.; Lu, Y. H.; Zhang, A. H.; Zhang, G. F.; Mei, Y. N.; Jiang, N.; Lei, X.; Song, Y. C.; Ng, S. W.; Tan, R. X. Org. Lett. 2014, 16, 5366. |
| [1] | (b) Michael, J. P. Nat. Pro. Rep. 2007, 24, 191. |
| [1] | (c) Zhang, J.; Morris-Natschke, S. L.; Ma, D.; Shang, X.-F.; Yang, C.-J.; Liu, Y.-Q.; Lee, K.-H. Med. Res. Rev. 2021, 41, 928. |
| [2] | Liang, F.; Eda, K.; Okazoe, T.; Wada, A.; Mori, N.; Konishi, K.; Tsuda, A. J. Org. Chem. 2021, 86, 6504. |
| [3] | Tucker, J. W.; Narayanam, J. M. R.; Krabbe, S. W.; Stephenson, C. R. J. Org. Lett. 2010, 12, 368. |
| [4] | Ghosh, I.; Ghosh, T.; Bardagi, J. I.; K?nig, B. Science 2014, 346, 725. |
| [5] | (a) Kim, H.; Kim, H.; Lambert, T. H.; Lin, S. J. Am. Chem. Soc. 2020, 142, 2087. |
| [5] | (b) Cowper, N. G. W. C.; Chernowsky, P.; Williams, O. P.; Wickens, Z. K. J. Am. Chem. Soc. 2020, 142, 2093. |
| [5] | (c) Neumeier, M.; Sampedro, D.; Májek, M.; de la Pe?a O'Shea, V. A.; Jacobi von Wangelin, A.; Pérez-Ruiz, R. Chem.-Eur. J. 2018, 24, 105. |
| [5] | (d) Caby, S.; Bouchet, L. M.; Argüello, J. E.; Rossi, R. A.; Bardagi, J. I. ChemCatChem 2021, 13, 3001. |
| [5] | (e) Ghosh, I.; Shaikh, R. S.; K?nig, B. Angew. Chem., Int. Ed. 2017, 56, 8544. |
| [5] | (f) Ghosh, I.; K?nig, B. Angew. Chem., Int. Ed. 2016, 55, 7676. |
| [5] | (g) Chernowsky, C. P.; Chmiel, A. F.; Wickens, Z. K. Angew. Chem., Int. Ed. 2021, 60, 21418. |
| [5] | (h) Ding, Y.-X.; Zhang, R.-Q.; Ma, R.-C.; Ma, Y.-M. Adv. Synth. Catal. 2022, 364, 355. |
| [6] | Li, Z.-J.; Li, S.; Hofman, E.; Hunter Davis, A.; Leem, G.; Zheng, W. Green Chem. 2020, 22, 1911. |
| [7] | (a) Carta, A.; Piras, S.; Loriga, G.; Paglietti, G. Mini-Rev. Med. Chem. 2006, 6, 1179. |
| [7] | (b) Liu, R.; Huang, Z.; Murray, M. G.; Guo, X.; Liu, G. J. Med. Chem. 2011, 54, 5747. |
| [8] | Carr?r, A.; Brion, J.-D.; Messaoudi, S.; Alami, M. Org. Lett. 2013, 15, 5606. |
| [9] | Ramesh, B.; Reddy, C. R.; Kumar, G. R.; Reddy, B. V. S. Tetrahedron Lett. 2018, 59, 628. |
| [10] | (a) Hong, Y.-Y.; Peng, Z.; Ma, H.; Zhu, Q.; Xu, X.-Q.; Yang, L.-H.; Xie, L.-Y. Tetrahedron Lett. 2022, 89, 153595. |
| [10] | (b) Kishor, G.; Ramesh, V.; Rao, V. R.; Pabbaraja, S.; Adiyala, P. R. RSC Adv. 2022, 12, 12235. |
| [10] | (c) Ni, H.; Li, Y.; Deng, J.; Shi, X.; Pan, Q. New J. Chem. 2021, 45, 22432. |
| [10] | (d) Sun, K.; Shi, A.; Liu, Y.; Chen, X.; Xiang, P.; Wang, X.; Qu, L.; Yu, B. Chem. Sci. 2022, 13, 5659. |
| [10] | (e) Xie, L.-Y.; Bai, Y.-S.; Xu, X.-Q.; Peng, X.; Tang, H.-S.; Huang, Y.; Lin, Y.-W.; Cao, Z.; He, W.-M. Green Chem. 2020, 22, 1720. |
| [11] | (a) Yuan, J.; Liu, S.; Qu, L. Adv. Syn. Catal. 2017, 359, 4197. |
| [11] | (b) Yuan, J.-W.; Fu, J.-H.; Liu, S.-N.; Xiao, Y.-M.; Mao, P.; Qu, L.-B. Org. Biomol. Chem. 2018, 16, 3203. |
| [11] | (c) Xie, L.-Y.; Peng, S.; Fan, T.-G.; Liu, Y.-F.; Sun, M.; Jiang, L.-L.; Wang, X.-X.; Cao, Z.; He, W.-M. Sci. China: Chem. 2019, 62, 460. |
| [11] | (d) Meng, N.; Wang, L.; Liu, Q.; Li, Q.; Lv, Y.; Yue, H.; Wang, X.; Wei, W. J. Org. Chem. 2020, 85, 6888. |
| [12] | (a) Niu, K.; Ding, L.; Zhou, P.; Hao, Y.; Liu, Y.; Song, H.; Wang, Q. Green Chem. 2021, 23, 3246. |
| [12] | (b) Niu, K.; Song, L.; Hao, Y.; Liu, Y.; Wang, Q. Chem. Commun. 2020, 56, 11673. |
| [12] | (c) Gao, Y.; Wu, Z.; Yu, L.; Wang, Y.; Pan, Y. Angew. Chem., Int. Ed. 2020, 59, 10859. |
| [12] | (d) Lin, D.-Z.; Huang, J.-M. Org. Lett. 2019, 21, 5862. |
| [12] | (e) Lei, N.; Shen, Y.; Li, Y.; Tao, P.; Yang, L.; Su, Z.; Zheng, K. Org. Lett. 2020, 22, 9184. |
| [13] | (a) Clinton, C. D.; Prasad, C. D.; Thombal, R. S.; Lee, Y. R. Adv. Syn. Catal. 2021, 363, 776. |
| [13] | (b) Ni, H.; Li, Y.; Shi, X.; Pang, Y.; Jin, C.; Zhao, F. Tetrahedron Lett. 2021, 68, 152915. |
| [13] | (c) Xu, J.; Yang, H.; He, L.; Huang, L.; Shen, J.; Li, W.; Zhang, P. Org. Lett. 2021, 23, 195. |
| [14] | (a) Tan, Y.; Liu, B.; Han, Y.-P.; Zhang, Y.; Zhang, H.-Y.; Zhao, J. Chem.-Asian J. 2020, 15, 3365. |
| [14] | (b) Wei, W.; Wang, L.; Bao, P.; Shao, Y.; Yue, H.; Yang, D.; Yang, X.; Zhao, X.; Wang, H. Org. Lett. 2018, 20, 7125. |
| [14] | (c) Yuan, J.-W.; Zhu, J.-L.; Li, B.; Yang, L.-Y.; Mao, P.; Zhang, S.-R.; Li, Y.-C.; Qu, L.-B. Org. Biomol. Chem. 2019, 17, 10178. |
| [15] | (a) Jiang, X.; Yang, L.; Ye, Z.; Du, X.; Fang, L.; Zhu, Y.; Chen, K.; Li, J.; Yu, C. Eur. J. Org. Chem. 2020, 2020, 1687. |
| [15] | (b) Xu, J.; Yang, H.; Cai, H.; Bao, H.; Li, W.; Zhang, P. Org. Lett. 2019, 21, 4698. |
| [15] | (c) Zhao, L.; Wang, L.; Gao, Y.; Wang, Z.; Li, P. Adv. Syn. Catal. 2019, 361, 5363. |
| [15] | (d) Jin, J.; Tong, J.; Yu, W.; Qiao, J.; Shen, C. Catal. Commun. 2020, 141, 106008. |
| [15] | (e) Wang, L.-F.; Li, S.; Xiao, X.-Q.; Xu, W.-M.; Zhang, P.-F.; Ma, Y.-M. Adv. Synth. Catal. 2022, 364, 855. |
| [16] | (a) Teng, Q.-H.; Yao, Y.; Wei, W.-X.; Tang, H.-T.; Li, J.-R.; Pan, Y.-M. Green Chem. 2019, 21, 6241. |
| [16] | (b) Zhou, J.; Li, Z.; Sun, Z.; Ren, Q.; Zhang, Q.; Li, H.; Li, J. J. Org. Chem. 2020, 85, 4365. |
| [16] | (c) Zhou, J.; Zhou, P.; Zhao, T.; Ren, Q.; Li, J. Adv. Syn. Catal. 2019, 361, 5371. |
| [17] | (a) Kim, Y.; Kim, D. Y. Tetrahedron Lett. 2018, 59, 2443. |
| [17] | (b) Li, K.-J.; Jiang, Y.-Y.; Xu, K.; Zeng, C.-C.; Sun, B.-G. Green Chem. 2019, 21, 4412. |
| [17] | (c) Rawat, D.; Kumar, R.; Subbarayappa, A. Green Chem. 2020, 22, 6170. |
| [17] | (d) Zhang, J.-Q.; Hu, D.; Song J.; Ren, H. J. Org. Chem. 2021, 86, 4646. |
| [17] | (e) Zhang, J.-Q.; Liu, J.; Hu, D.; Song, J.; Zhu, G.; Ren, H. Org. Lett. 2022, 24, 786. |
| [18] | Dai, C.; Zhan, Y.; Liu, P.; Sun, P. Green Chem. 2021, 23, 314. |
| [19] | Chupakhin, O. N.; Sidorov, E. O.; Postovskii, I. Y. Khim. Geterotsikl. Soedin. 1975, 10, 1433. |
| [20] | Noikham, M.; Kittikool, T.; Yotphan, S. Synthesis 2018, 50, 2337. |
| [21] | Utepova, I. A.; Trestsova, M. A.; Chupakhin, O. N.; Charushin, V. N.; Rempel, A. A. Green Chem. 2015, 17, 4401. |
| [22] | Romero, N. A.; Nicewicz, D. A. Chem. Rev. 2016, 116, 10075. |
| [23] | Akaba, R.; Ohshima, K.; Kawai, Y.; Obuchi, Y.; Negishi, A.; Sakuragi, H.; Tokumaru, K. Tetrahedron Lett. 1991, 32, 109. |
| [24] | Sadki, S.; Schottland, P.; Brodie, N.; Sabouraud, G. Chem. Soc. Rev. 2000, 29, 283. |
| [25] | Benniston, A. C.; Harriman, A.; Li, P.; Rostron, J. P.; Ramesdonk, H. J.; Groeneveld, M. M.; Zhang, H.; Verhoeven, J. W. J. Am. Chem. Soc. 2005, 127, 16054. |
| [26] | Speckmeier, E.; Fischer, T. G.; Zeitler, K. J. Am. Chem. Soc. 2018, 140, 15353. |
| [27] | (a) Wang, B.; Zou, L.; Wang, L.; Sun, M.; Li, P. Chin. Chem. Lett. 2021, 32, 1229. |
| [27] | (b) Wang, Z.; Wang, L.; Wang, Z.; Li, P.; Zhang, Y. Chin. Chem. Lett. 2021, 32, 429. |
| [27] | (c) Xie, X.; Wang, L.; Zhou, Q.; Ma, Y.; Wang, Z.-M.; Li, P. Chin. Chem. Lett. 2022, 33, 5069. |
| [27] | (d) Zhou, Q.; Yu, H.-Y.; Zhou, Y.; Wei, J.-R.; Wang, L. Org. Biomol. Chem. 2022, 20, 5575. |
/
| 〈 |
|
〉 |