

含直链烷烃的胍基腙衍生物的合成及其抗菌活性评价
收稿日期: 2022-10-24
修回日期: 2022-12-05
网络出版日期: 2023-01-05
基金资助
国家自然科学基金(81560561); 江西省自然科学基金(20224ACB206044)
Synthesis and Antibacterial Activity Evaluation of Guanidine Hydrazone Derivatives Containing Linear Alkanes
Received date: 2022-10-24
Revised date: 2022-12-05
Online published: 2023-01-05
Supported by
National Natural Science Foundation of China(81560561); Natural Science Foundation of Jiangxi Province(20224ACB206044)
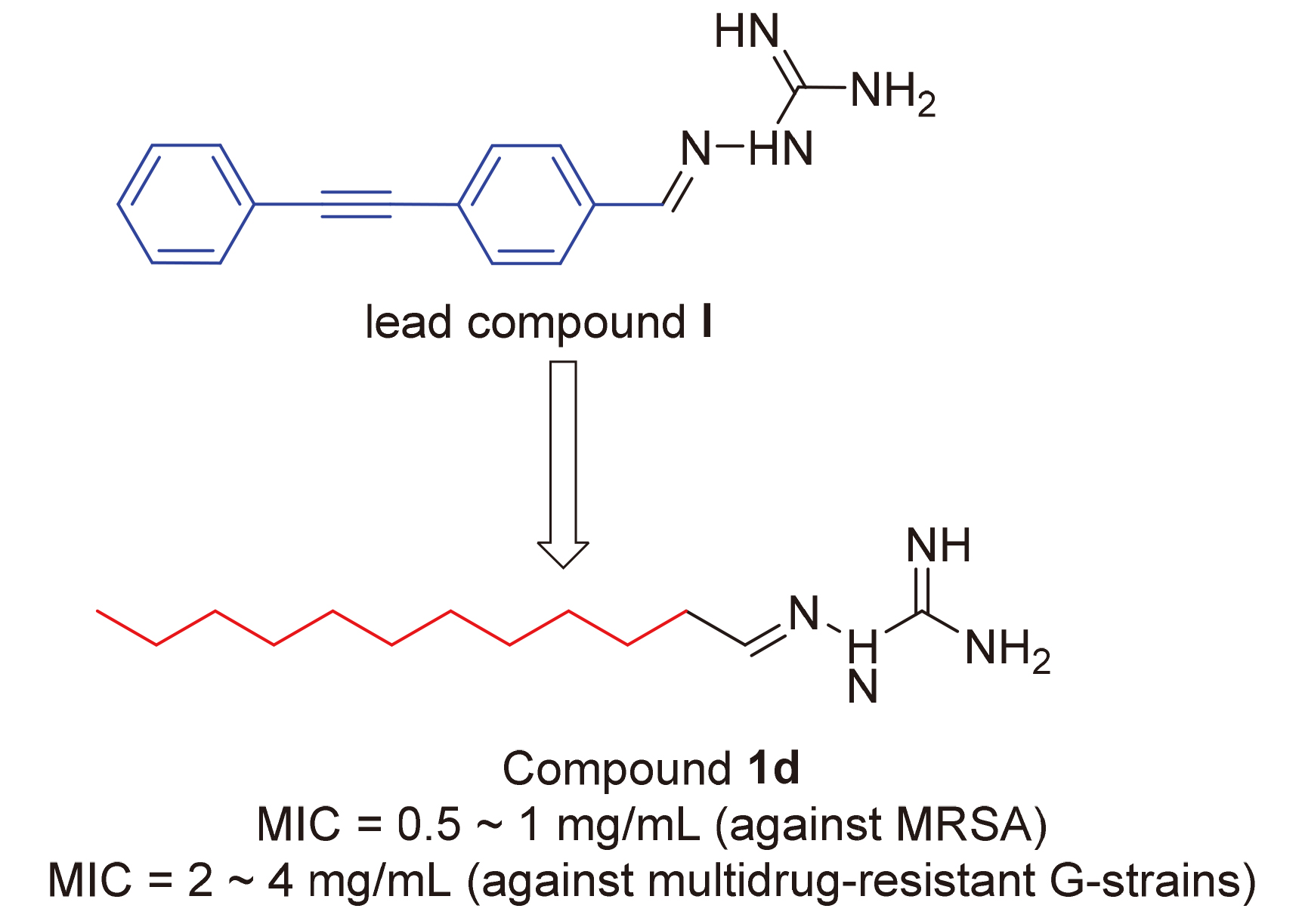
抗生素耐药性的上升和新抗生素发现的减少造成了全球健康危机. 为了获得有效的抗菌剂和缓解细菌耐药性,设计合成了一系列新型的含直链烷烃的胍基腙, 评价了目标化合物对五株革兰氏阳性菌株、四株革兰氏阴性菌株和四株多药耐药菌株的抗菌活性. 结果显示, 除(E)-2-己基亚肼-1-碳酰亚胺(1a)之外, 其他所有化合物均表现出不同程度的抗菌性能, 最低抑菌浓度(MIC)为0.5~64 μg/mL. 其中(E)-2-十二烷基亚肼-1-碳酰亚胺(1d)、(E)-2-十四烷基亚肼-1-碳酰亚胺(1e)、(E)-2-(4-(庚氧基)亚苄基)肼-1-碳酰亚胺(2d)和(E)-2-(4-(辛氧基)亚苄基)肼-1-碳酰亚胺(2e)对菌株33693、29212、63501、10104和43300的抗菌活性与青霉素和诺氟沙星相当或更好. 本研究证实化合物2d还具有优良的杀菌性能, 并且未发现细菌对化合物2d产生耐药性趋势. 细胞毒性评价显示该系列化合物具有一定细胞毒性, 但抗菌活性浓度下未见毒性反应. 此外, 还进行了2d与LpxC的分子对接, 结果证明化合物2d与LpxC结合良好, 这可能是其产生优良抗菌活性的分子机制之一. 这些发现说明胍腙与直链烷烃偶联可以作为一种潜在骨架, 用于开发新型抗菌药物.
宋明霞 , 朱洋女 , 王世帅 , 黄玉萍 , 邓先清 , 黄玉珊 . 含直链烷烃的胍基腙衍生物的合成及其抗菌活性评价[J]. 有机化学, 2023 , 43(6) : 2163 -2170 . DOI: 10.6023/cjoc202210029
The rise of antibiotic resistance and the declining discovery of new antibiotics have created a global health crisis. To obtain effective antibacterial agents and relieve bacterial resistance, a series of novel guanidine hydrazones containing linear alkanes were designed and synthesized. The antimicrobial activities of these compounds were evaluated against five gram-positive strains, four gram-negative strains and four multidrug-resistant strains. All of the targets except (E)-2-hexyli- denehydrazine-1-carboximidamide (1a) exhibited a different degree of antibacterial properties with minimum inhibitory concentration (MIC) in 0.5~64 μg/mL. Among them, (E)-2-dodecylidenehydrazine-1-carboximidamide (1d), (E)-2-tetradecyli- denehydrazine-1-carbox-imidamide (1e), (E)-2-(4-(heptyloxy)benzylidene)hydrazine-1-carboximidamide (2d) and (E)-2-(4- (octyloxy)benzylidene)hydrazine-1-carbox-imidamide (2e) showed equivalent or better antibacterial activity than penicillin and norfloxacin against strains 33693, 29212, 63501, 10104 and 43300. Excellent bactericidal properties of 2d and low frequency of bacteria developing resistance toward 2d were established. The cytotoxicity evaluation showed that the target compounds had certain cytotoxicity, but no cytotoxicity was found under the concentration of antibacterial activity. Molecular docking of 2d with LpxC was performed, which demonstrated that 2d has a forceful binding with LpxC. These findings strongly support the assumption that guanidine hydrazone coupled with linear alkane is a potential skeleton to develop new antimicrobial agents.

Key words: guanidine hydrazone; antimicrobial; linear alkane; resistance
| [1] | Hughes, D.; Andersson, D. I. Annu. Rev. Microbiol. 2017, 8, 579. |
| [2] | Mühlberg, E.; Umst?tter, F.; Kleist, C.; Domhan, C.; Mier, W.; Uhl, P. Can. J. Microbiol. 2020, 66, 11. |
| [3] | Chellat, M. F.; Raguz?, L.; Riedl, R. Angew. Chem., nt. Ed. 2016, 55, 6600. |
| [4] | Yang, T.; Wang, J. Y.; Cao, J. Y.; Zhang, X. Y.; Lai, Y.; Li, L. N.; Ye, X. Y.; You, C. Ital. J. Pediatr. 2021, 47, 169. |
| [5] | So, M.; Walti, L. Curr. Infect. Dis. Rep. 2022, 24, 63. |
| [6] | Rai, M.; Zimowska, B.; Gade, A.; Ingle, P. AMB Express 2022, 12, 60. |
| [7] | Prasad, N. K.; Seiple, I. B.; Cirz, R. T.; Rosenberg, O. S. Antimicrob. Agents Chemother. 2022, 66, e0005422. |
| [8] | Alfei, S.; Schito, A. M. Pharmaceuticals (Basel) 2022, 15, 476. |
| [9] | Whittington, D. A.; Rusche, K. M.; Shin, H.; Fierke, C. A.; Christianson, D. W. Proc. Natl. Acad. Sci. U. S. A. 2003, 100, 8146. |
| [10] | Kalinin, D. V.; Holl, R. Curr. Top Med. Chem. 2016, 16, 2379. |
| [11] | Erwin, A. L. Cold Spring Harbor Perspect. Med. 2016, 6, a025304. |
| [12] | Zhou, P.; Hong, J. Acc. Chem. Res. 2021, 54, 1623. |
| [13] | Kang, Y.; Zhao, M.; Zhang, J. Chin. J. Antibiot. 2017, 42, 169. (in Chinese) |
| [13] | (康悦, 赵明, 张菁, 中国抗生素杂志, 2017, 42, 169.) |
| [14] | Sidoryk, K.; ?witalska, M.; Rózga, P.; Wietrzyk, J.; Bujak, I.; ?erek, B.; Kaczmarek, ?.; Cybulski, M. Med. Chem. Res. 2017, 26, 3354. |
| [15] | D?m?t?r, O.; May, N. V.; Gál, G. T.; Spengler, G.; Dobrova, A.; Arion, V. B.; Enyedy, é. A. Molecules 2022, 27, 2044. |
| [16] | Deng, X. Q.; Song, M. X. J. Enzyme Inhib. Med. Chem. 2020, 35, 354. |
| [17] | Yu, H.-H.; Zhou, S.-C.; Guo, T.-T.; Liang, Z.; Chen, H.-B.; Dai, W.-K.; Song, M.-X. Chin. J. Org. Chem. 2019, 39, 1497. (in Chinese) |
| [17] | (余海红, 周胜超, 郭婷婷, 梁焯, 陈华斌, 代卫凯, 宋明霞, 有机化学, 2019, 39, 1497.) |
| [18] | Song, M. X.; Wang, S. B.; Wang, Z. T.; Fu, Z. Y.; Zhou, S. C.; Cheng, H. B.; Liang, Z.; Deng, X. Q. Eur. J. Med. Chem. 2019, 166, 108. |
| [19] | Brown, E. D.; Wright, G. D. Nature 2016, 529, 336. |
| [20] | Dubey, K. K.; Sharma, I. M. Arch. Pharm. (Weinheim, Ger.) 2020, 353, e2000168. |
| [21] | Makovitzki, A.; Avrahami, D.; Shai, Y. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 15997. |
| [22] | Hoque, J.; Akkapeddi, P.; Yadav, V.; Manjunath, G. B.; Uppu, D. S.; Konai, M. M.; Yarlagadda, V.; Sanyal, K.; Haldar, J. ACS Appl. Mater. Interfaces 2015, 7, 1804. |
| [23] | Kosowska-Shick, K.; Clark, C.; Pankuch, G. A.; McGhee, P.; Dewasse, B.; Beachel, L.; Appelbaum, P. C. Antimicrob. Agents Chemother. 2009, 53, 4217. |
| [24] | Qiu, X.; Janson, C. A.; Smith, W. W.; Head, M.; Lonsdale, J.; Konstantinidis, A. K. J. Mol. Biol. 2001, 307, 341. |
/
| 〈 |
|
〉 |