

借助有机催化去共轭-羟醛缩合反应来获得α-乙烯基-β-炔基取代的烯醛
收稿日期: 2022-11-28
修回日期: 2022-12-24
网络出版日期: 2023-01-12
基金资助
国家自然科学基金(21871160); 国家自然科学基金(21672121); 国家自然科学基金(22071130)
Access to α-Vinyl β-Alkynyl Enals via an Organocatalytic Deconjugation-Aldol Condensation Sequence
Received date: 2022-11-28
Revised date: 2022-12-24
Online published: 2023-01-12
Supported by
National Natural Science Foundation of China(21871160); National Natural Science Foundation of China(21672121); National Natural Science Foundation of China(22071130)
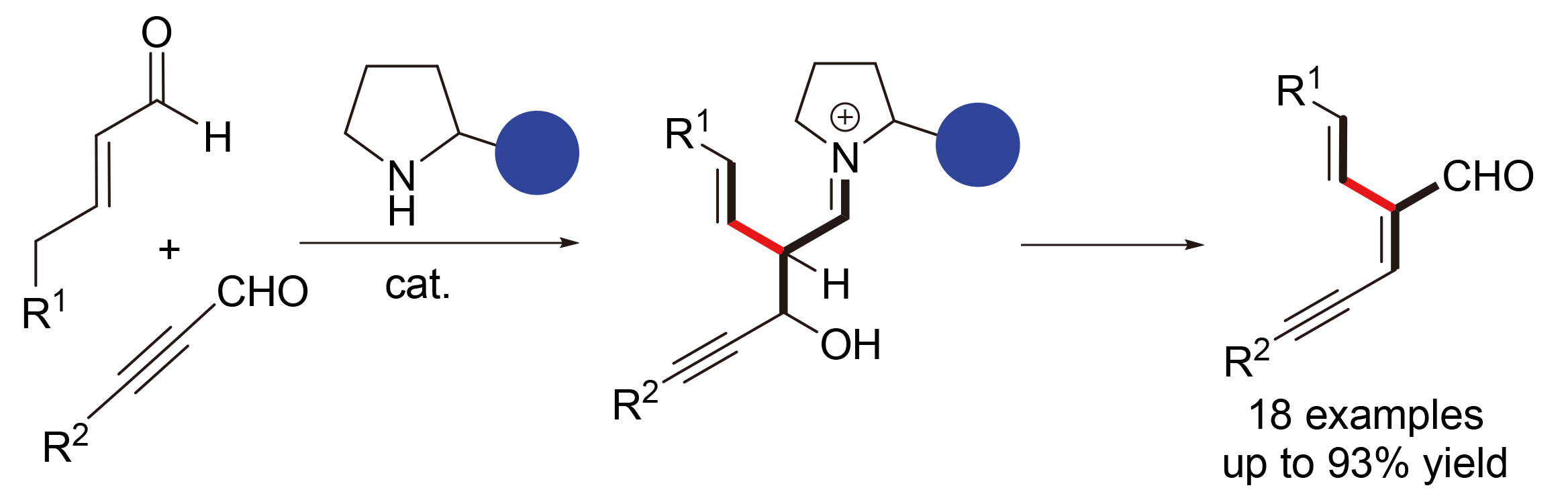
发展了一种从α,β-不饱和醛和炔醛出发来获取α-乙烯基-β-炔基取代的烯醛的新绿色合成路线. 该方法体现出较为广泛的底物适用范围, 且反应条件温和. 此外, 还对反应过程中共轭-羟醛缩合的催化过程进行了详细讨论.
关键词: α-乙烯基-β-炔基取代的烯醛; 共轭-羟醛缩合; 有机催化; 烯胺
全翌雯 , 蒋心惠 , 李文军 , 汪舰 . 借助有机催化去共轭-羟醛缩合反应来获得α-乙烯基-β-炔基取代的烯醛[J]. 有机化学, 2023 , 43(6) : 2120 -2125 . DOI: 10.6023/cjoc202211035
A new green synthetic route to α-vinyl β-alkynyl enals from α,β-unsaturated aldehydes and ynals is developed. This protocol allows a broad substrate scope and mild conditions. Furthermore, a proposed mechanism of a deconjugation-aldol condensation process is discussed in detail.

| [1] | Johnson, P. J. M.; Farag, M. H.; Halpin, A.; Morizumi, T.; Prok- horenko, V. I.; Knoester, J.; Jansen, T. L. C.; Ernst, O. P.; Miller, R. J. D. J. Phys. Chem. B 2017, 121, 4040. |
| [2] | Abramson, S. N.; Radic, Z.; Manker, D.; Faulkner, D. J.; Taylor, P. Mol. Pharmacol. 1989, 36, 349. |
| [3] | (a) Amade, P.; Lemée, R. Aquat. Toxicol. 1998, 43, 287. |
| [3] | (b) Hansen, E.; Even, Y.; Genevière, A.-M. Toxicol. Sci. 2004, 81, 190. |
| [4] | (a) Kobayashi, S.; Kudo, K.; Ito, A.; Honjo, T.; Yata, M.; Otani, T.; Kutsumura, N.; Saito, T.; Berrée, F.; Romain, E.; Tripoteau, F.; Carboni, B. Eur. J. Org. Chem. 2015, 2015, 4367. |
| [4] | (b) Kobayashi, S.; Kudo, K.; Ito, A.; Hirama, S.; Otani, T.; Saito, T. Chem. Rev. 2003, 103, 3401. |
| [4] | (c) Benaglia, M.; Puglisi, A.; Cozzi, F. Chem. Rev. 2003, 103, 3401. |
| [5] | (a) Dalko, P. I.; Moisan, L. Angew. Chem., Int. Ed. 2001, 40, 3726. |
| [5] | (b) Benaglia, M.; Puglisi, A.; Cozzi, F. Chem. Rev. 2003, 103, 3401. |
| [5] | (c) Berkessel, A.; Gr?ger, H. In Metal-Free Organic Catalysts in Asymmetric Synthesis, Wiley-VCH, Weinheim, 2004. |
| [5] | (d) Houk, K. N.; List, B. Acc. Chem. Res. 2004, 37, 487. |
| [5] | (e) Dalko, P. I.; Moisan, L. Angew. Chem., Int. Ed. 2004, 43, 5138. |
| [5] | (f) Mohr, J. T.; Krout, M. R.; Stoltz, B. M. Nature 2008, 455, 323. |
| [5] | (g) MacMillan, D. W. C. Nature 2008, 455, 304. |
| [5] | (h) Berkessel, A.; Gr?ger, H. In Asymmetric Organocatalysis, Wiley-VCH, Weinheim, 2005. |
| [5] | (i) Dalko, P. I. In Enantioselective Organocatalysis, Wiley-VCH, Weinheim, 2007. |
| [5] | (j) Reetz, M. T.; List, B.; Jaroch, S.; Weinmann, H. In Organocatalysis, Springer, Berlin, 2007. |
| [5] | (k) List, B. In Asymmetric Organocatalysis, Springer, Heidelberg, 2009. |
| [6] | Mukherjee, S.; Yang, J. W.; Hoffmann, S.; List, B. Chem. Rev. 2007, 107, 5471. |
| [7] | (a) Ramachary, D. B.; Reddy, Y. V. Eur. J. Org. Chem. 2012, 2012, 865. |
| [7] | (b) Nielsen, M.; Worgull, D.; Zweifel, T.; Gschwend, B.; Bertelsen, S.; J?rgensen, K. A. Chem. Commun. 2011, 47, 632. |
| [8] | Jia, Z.-J.; Jiang, H.; Li, J.-L.; Gschwend, B.; Li, Q.-Z.; Yin, X.; Grouleff, J.; Chen, Y.-C.; J?rgensen, K. A. J. Am. Chem. Soc. 2011, 133, 5053. |
| [9] | Zhang, X.; Zhang, S.; Wang, W. Angew. Chem., Int. Ed. 2010, 122, 1523. |
| [10] | Erkkil?, A.; Majander, I.; Pihko, P. M. Chem. Rev. 2007, 107, 5416. |
| [11] | Beeson, T. D.; Mastracchio, A.; Hong, J.; Ashton, K.; MacMillan, D. W. C. Science 2007, 316, 582. |
| [12] | (a) Hong, B.-C.; Wu, M.-F.; Tseng, H.-C.; Liao, J.-H. Org. Lett. 2006, 8, 2217. |
| [12] | (b) Hong, B.-C.; Wu, M.-F.; Tseng, H.-C.; Huang, G.-F.; Su, C.-F.; Liao, J.-H. J. Org. Chem. 2007, 72, 8459. |
| [12] | (c) Hong, B.-C.; Tseng, H.-C.; Chen, S.-H. Tetrahedron 2007, 63, 2840. |
| [12] | (d) Liu, K.; Chougnet, A.; Woggon, W.-D. Angew. Chem., Int. Ed. 2008, 47, 5827. |
| [12] | (e) Bergonzini, G.; Vera, S.; Melchiorre, P. Angew. Chem., Int. Ed. 2010, 49, 9685. |
| [12] | (f) Han, B.; He, Z.-Q.; Li, J.-L.; Jiang, K.; Liu, T.-Y.; Chen, Y.-C. Angew. Chem., Int. Ed. 2009, 48, 5474. |
| [12] | (g) Bencivenni, G.; Galzerano, P.; Mazzanti, A.; Bartoli, G.; Melchiorre, P. Proc. Natl. Acad. Sci. U. S. A. 2010, 107, 20642. |
| [12] | (h) Han, B.; Li, J.-L.; Ma, C.; Zhang, S.-J.; Chen, Y.-C. Angew. Chem., Int. Ed. 2008, 47, 9971. |
| [12] | (i) Cassani, C.; Melchiorre, P. Org. Lett. 2012, 14, 5590. |
| [12] | (j) Orue, A.; Reyes, E.; Vicario, J. L.; Carrillo, L.; Uria, U. Org. Lett. 2012, 14, 3740. |
| [12] | (k) Stiller, J.; Marques-Lopez, E.; Herrera, R. P.; Frohlich, R.; Strohmann, C.; Christmann, M. Org. Lett. 2011, 13, 70. |
| [12] | (l) Albrecht, ?.; Dickmeiss, G.; Weise, C. F.; Rodríguez-Escrich, C.; J?rgensen, K. A. Angew. Chem., Int. Ed. 2012, 51, 13109. |
| [12] | (m) Albrecht, ?.; Dickmeiss, G.; Cruz-Acosta, F.; Rodríguez- Escrich, C.; Davis, R. L.; J?rgensen, K. A. J. Am. Chem. Soc. 2012, 134, 2543. |
| [12] | (n) Talavera, G.; Reyes, E.; Vicario, J. L.; Carillo, L. Angew. Chem., Int. Ed. 2012, 51, 4104. |
| [13] | Li, W.; Wei, J.; Jia, Q.; Du, Z.; Zhang, K.; Wang, J. Chem.-Eur. J. 2014, 20, 6592. |
| [14] | Li, W.; Du, Z.; Huang, J.; Zhang, K.; Wang, J. Green Chem. 2014, 16, 3003. |
| [15] | (a) Jones, S. B.; Simmons, B.; MacMillan, D. W. C. J. Am. Chem. Soc. 2009, 131, 13606. |
| [15] | (b) Aleman, J.; Fraile, A.; Marzo, L.; Ruano, J. L. G.; Izquierdo, C.; Diaz-Tendero, S. Adv. Synth. Catal. 2012, 354, 1665. |
| [15] | (c) Cai, X.; Wang, C.; Sun, J. Adv. Synth. Catal. 2012, 354, 359. |
| [15] | (d) Dong, L.-J.; Fan, T.-T.; Wang, C.; Sun, J. Org. Lett. 2013, 15, 204. |
| [15] | (e) Zhang, X.-S.; Zhang, S.-L.; Wang, W. Angew. Chem., Int. Ed. 2010, 49, 1481. |
| [15] | (f) Liu, C.; Zhang, X.-S.; Wang, R.; Wang, W. Org. Lett. 2010, 12, 4948. |
| [15] | (g) Zhang, X.-S.; Song, X-X.; Li, H.; Zhang, S.-L.; Chen, X.-B.; Yu, X.-H.; Wang, W. Angew. Chem., Int. Ed. 2012, 51, 7282. |
| [15] | (h) Song, A.; Chen, X.; Song, X.; Zhang, X.; Zhang, S.; Wang, W. Org. Lett. 2013, 15, 2510. |
| [16] | Ryan, W.; Bedard, K.; Baidilov, D.; Tius, M.; Hudlicky, T. Tetrahedron Lett. 2018, 59, 2467. |
| [17] | Riveira, M. J.; Sarotti, A. M. Org. Biomol. Chem. 2018, 16, 1442. |
/
| 〈 |
|
〉 |