

铜催化芳基重氮乙酸酯与双[(频哪醇)硼基]甲烷的偶联反应
收稿日期: 2023-01-31
修回日期: 2023-03-12
网络出版日期: 2023-03-24
基金资助
国家自然科学基金(21871010)
Copper-Catalyzed Cross-Coupling of Aryldiazoacetates with Bis[(pinacolato)boryl]methane
Received date: 2023-01-31
Revised date: 2023-03-12
Online published: 2023-03-24
Supported by
National Natural Science Foundation of China(21871010)
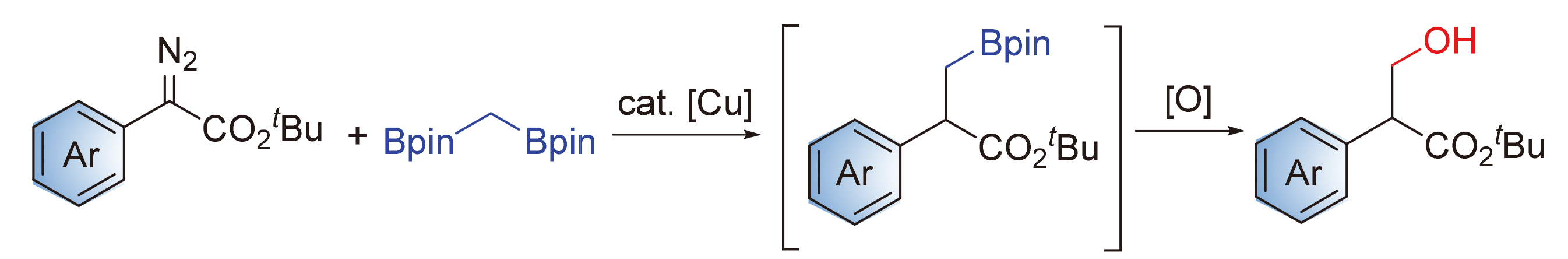
报道铜催化下重氮苯乙酸酯作为卡宾前体与双[(频哪醇)硼基]甲烷的交叉偶联反应, 实现sp3-杂化碳碳键的构筑. 条件筛选表明亚磷酰胺配体monophos对该反应的成功尤为关键. 原位氧化使得硼酯产物以醇的形式被分离, 反应具有高效率以及良好的官能团兼容性. 该两步反应是芳基重氮酯类化合物形式上的羟甲基化.
关键词: 铜催化; sp3-杂化碳碳键构筑; 重氮化合物; 双[(频哪醇)硼基]甲烷
鲍志成 , 李慕尧 , 王剑波 . 铜催化芳基重氮乙酸酯与双[(频哪醇)硼基]甲烷的偶联反应[J]. 有机化学, 2023 , 43(5) : 1808 -1814 . DOI: 10.6023/cjoc202301027
The copper-catalyzed cross-coupling reaction of 1,1-bis[(pinacolato)boryl]methane with α-diazophenylacetate as carbene precursor was reported, leading to the construction of C(sp3)—C(sp3) bond. Reaction optimization experiments showed that Monophos ligand was critical for the success of this reaction. In situ oxidation afforded the corresponding primary alcohols instead of the relatively unstable boronates. The reaction has high efficiency and good functional group compatibility. The two-step transformation represents a formal hydroxymethylation of diazoacetates.

| [1] | For selected reviews, see: (a) Liu,Z.; Zhang, Y.; Wang, J. Chin. J. Org. Chem. 2013, 33, 687. (in Chinese) |
| [1] | (刘振兴, 张艳, 王剑波, 有机化学, 2013, 33, 687.) |
| [1] | (b) Xia, Y.; Qiu, D.; Wang, J. Chem. Rev. 2017, 117, 13810. |
| [1] | (c) Liu, Z.; Sivaguru, P.; Zanoni, G.; Bi, X. Acc. Chem. Res. 2022, 55, 1763. |
| [1] | For selected recent examples, see: |
| [1] | (d) Xu, Z.-W.; Zhang, W.; Lin, J.-H.; Jin, C.-M.; Xiao, J.-C. Chin. J. Chem. 2020, 38, 1647. |
| [1] | (e) Liu, B.; Xu, M.-H. Chin. J. Chem. 2021, 39, 1911. |
| [1] | (f) Li, F.; Lu, B.; Liu, Y.; Wang, X. Chin. J. Org. Chem. 2022, 42, 3390. (in Chinese) |
| [1] | (李芳洁, 卢斌, 刘阳, 王晓明, 有机化学, 2022, 42, 3390.) |
| [1] | (g) Li, S.; Zhou, L. Chin. J. Org. Chem. 2022, 42, 3944. |
| [1] | (h) Shang, M.; Zhang, L.; Chen, M.; Hu, W.; He, X.; Lu, H. Chin. J. Org. Chem. 2022, 42, 3816. (in Chinese) |
| [1] | (商铭洲, 张兰兰, 陈淼淼, 胡汪成, 何心伟, 陆红健, 有机化学, 2022, 42, 3816.) |
| [1] | (i) Xia, J.; Gu, Z. Chin. J. Chem. 2020, 38, 1081. |
| [2] | For selected examples of alkynes, see: (a) Xiao,Q.; Xia, Y.; Li, H.; Zhang, Y.; Wang, J. Angew. Chem., Int. Ed. 2011, 50, 1114. |
| [2] | (b) Ye, F.; Ma, X.; Xiao, Q.; Li, H.; Zhang, Y.; Wang, J. J. Am. Chem. Soc. 2012, 134, 5742. |
| [2] | (c) Hossain, M. L.; Ye, F.; Zhang, Y.; Wang, J. J. Org. Chem. 2013, 78, 1236. |
| [2] | (d) Ye, F.; Hossain, M. L.; Xu, Y.; Ma, X.; Xiao, Q.; Zhang, Y.; Wang, J. Chem.-Asian J. 2013, 8, 1404. |
| [2] | (e) Ye, F.; Wang, C.; Ma, X.; Hossain, M. L.; Xia, Y.; Zhang, Y.; Wang, J. J. Org. Chem. 2015, 80, 647. |
| [2] | (f) Poh, J.-S.; Tran, D. N.; Battilocchio, C.; Hawkins, J. M.; Ley, S. V. Angew. Chem., Int. Ed. 2015, 54, 7920. |
| [2] | (g) Tang, Y.; Chen, Q.; Liu, X.; Wang, G.; Lin, L.; Feng, X. Angew. Chem., Int. Ed. 2015, 54, 9512. |
| [2] | (h) Chu, W.-D.; Zhang, L.; Zhang, Z.; Zhou, Q.; Mo, F.; Zhang, Y.; Wang, J. J. Am. Chem. Soc. 2016, 138, 14558. |
| [2] | For selected examples of arenes, see: (i) Zhao, X.; Wu, G.; Zhang, Y.; Wang, J. J. Am. Chem. Soc. 2011, 133, 3296. |
| [2] | (j) Xiao, Q.; Ling, L.; Ye, F.; Tan, R.; Tian, L.; Zhang, Y.; Li, Y.; Wang, J. J. Org. Chem. 2013, 78, 3879. |
| [2] | (k) Xu, S.; Wu, G.; Ye, F.; Wang, X.; Li, H.; Zhao, X.; Zhang, Y.; Wang, J. Angew. Chem., Int. Ed. 2015, 54, 4669. |
| [3] | For selected examples, see: (a) Hu,M.; Ni, C.; Hu, J. J. Am. Chem. Soc. 2012, 134, 15257. |
| [3] | (b) Hu, M.; He, Z.; Gao, B.; Li, L.; Ni, C.; Hu, J. J. Am. Chem. Soc. 2013, 135, 17302. |
| [3] | (c) Wang, X.; Zhou, Y.; Ji, G.; Wu, G.; Li, M.; Zhang, Y.; Wang, J. Eur. J. Org. Chem. 2014, 3093. |
| [3] | (d) Das, A.; Wang, D.; Belhomme, M. C.; Szabo?, K. J. Org. Lett. 2015, 17, 4754. |
| [3] | (e) Xu, S.; Gao, Y.; Chen, R.; Wang, K.; Zhang, Y.; Wang, J. Chem. Commun. 2016, 52, 4478. |
| [3] | (f) Zhang, H.; Wu, G.; Yi, H.; Sun, T.; Wang, B.; Zhang, Y.; Dong, G.; Wang, J. Angew. Chem., Int. Ed. 2017, 56, 3945. |
| [4] | For selected reviews, see: (a) Paul,S.; Das, K. K.; Aich, D.; Manna, S.; Panda, S. Org. Chem. Front. 2022, 9, 838. |
| [4] | (b) Lee, Y.; Han, S.; Cho, S. H. Acc. Chem. Res. 2021, 54, 3917. |
| [4] | (c) Nallagonda, R.; Padala, K.; Masarwa, A. Org. Biomol. Chem. 2018, 16, 1050. |
| [4] | (d) Miralles, N.; Maza, R. J.; Fernández, E. Adv. Synth. Catal. 2018, 360, 1306. |
| [4] | (e) Wu, C.; Wang, J. Tetrahedron Lett. 2018, 59, 2128. |
| [4] | (f) Jo, W.; Lee, J. H.; Cho, S. H. Chem. Commun. 2021, 57, 4346. |
| [4] | (g) Xu, L.; Zhang, S.; Li, P. Chem. Soc. Rev. 2015, 44, 8848. |
| [5] | Li, H.; Shangguan, X.; Zhang, Z.; Huang, S.; Zhang, Y.; Wang, J. Org. Lett. 2014, 16, 448. |
| [6] | For selected examples, see: (a) Wu,C.; Bao, Z.; Dou, B.; Wang, J. Chem.-Eur. J. 2021, 27, 2294. |
| [6] | (b) Bao, Z.; Wu, C.; Wang, J. Eur. J. Org. Chem. 2022, e202201264. |
| [6] | (c) Bao, Z.; Huang, M.; Xu, Y.; Zhang, X.; Wu, Y.-D.; Wang, J. Angew. Chem., Int. Ed. 2023, 62, e202216356. |
| [7] | Walton, J. C.; McCarroll, A. J.; Chen, Q.; Carboni, B.; Nziengui, R. J. Am. Chem. Soc. 2000, 122, 5455. |
| [8] | Huang, Z.; Hartwig, J. F. Angew. Chem., Int. Ed. 2012, 51, 1028. |
/
| 〈 |
|
〉 |