

三组分交叉偶联反应高效合成双芳基硫醚
收稿日期: 2024-09-04
修回日期: 2024-10-10
网络出版日期: 2024-11-08
基金资助
国家自然科学基金(22201158); 高等学校学科创新引智计划(111计划); 高等学校学科创新引智计划(D20015); 湖北省自然科学基金(2024AFB212); 湖北省自然科学基金(2024AFB152)
Efficient Synthesis of Diaryl Sulfides via Three Component Cross Coupling
Received date: 2024-09-04
Revised date: 2024-10-10
Online published: 2024-11-08
Supported by
National Natural Science Foundation of China(22201158); Programme of Introducing Talents of Discipline to Universities (111 Project); Programme of Introducing Talents of Discipline to Universities(D20015); Hubei Provincial Natural Science Foundation(2024AFB212); Hubei Provincial Natural Science Foundation(2024AFB152)
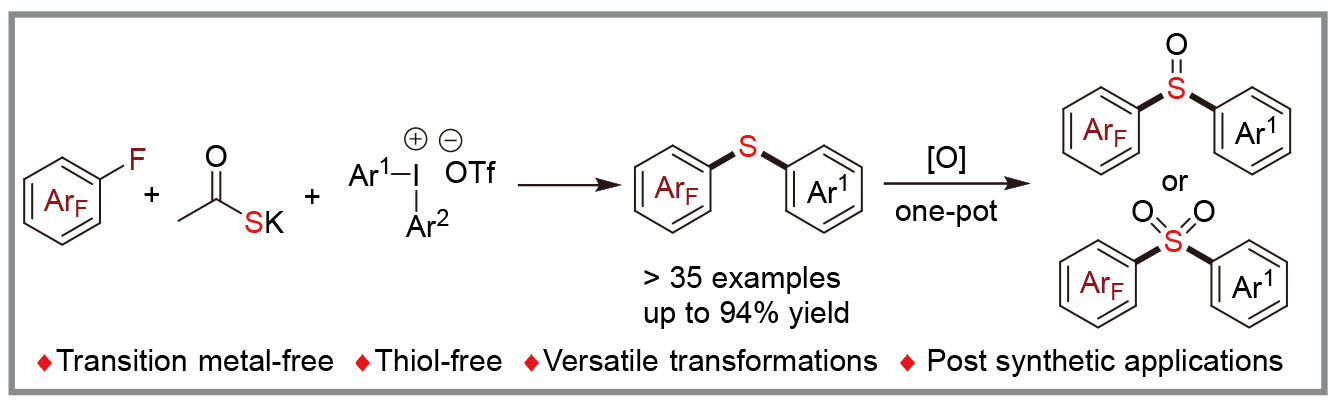
双芳基硫醚常存在于药物和天然产物中, 通常涉及金属交叉偶联反应、高温以及使用稳定性差、气味难闻的硫酚进行合成. 基于此, 研究了在无过渡金属催化条件下, 多氟芳烃、二芳基碘盐及硫代乙酸钾的三组分交叉偶联反应. 该反应通过商业化的硫代乙酸钾与两种缺电子芳烃经连续两次亲电取代反应, 合成了系列非对称的双芳基硫醚, 表现出了宽泛的底物适用范围和官能团兼容性. 此外, 通过一锅法氧化反应可以实现砜和亚砜类化合物的合成, 拓展了该策略的应用范围; 克级规模实验和衍生化实验均表现出优异的结果, 进一步证明了该反应具有潜在的应用价值. 通过对照、自由基抑制及中间体捕获实验阐明了其机制.
刘梦琴 , 陈贻庭 , 张榉元 , 周和烨 , 秦涛 , 刘彬 . 三组分交叉偶联反应高效合成双芳基硫醚[J]. 有机化学, 2025 , 45(4) : 1283 -1296 . DOI: 10.6023/cjoc202407024
Diaryl sulfides are found in drugs and natural products, yet their synthesis traditionally requires metal-catalyzed cross-coupling reactions, high temperatures, and the use of the air-sensitive and extremely odorous thiophenols. A novel transition-metal-free strategy for the synthesis of unsymmetric diaryl sulfides via a three-component cross-coupling of polyfluoro- arenes, diaryliodide salts and potassium thioacetate is reported. This approach enables the formation of a diverse array of unsymmetric diaryl thioethers through two successive electrophilic substitution reactions involving commercially available potassium thioacetate and two electron-deficient aromatic hydrocarbons, demonstrating broad substrate scope and functional group compatibility. Additionally, this strategy facilitates the synthesis of sulfones and sulfoxides via a one-pot oxidation reaction, thereby extending its applicability. Gram-scale reactions and derivatization experiments have shown excellent results, further proving the potential application value of this reaction. Mechanistic insights were elucidated through control, radical inhibition and intermediate capture experiments.

| [1] | (a) Liu, H.; Fujiwara, T.; Nishikawa, T.; Mishima, Y.; Nagai, H.; Shida, T.; Tachibana, K.; Kobayashi, H.; Mangindaan, R. E.; Namikoshi, M. Tetrahedron 2005, 61, 8611. |
| [1] | (b) Nakazawa, T.; Xu, J.; Nishikawa, T.; Oda, T.; Fujita, A.; Ukai, K.; Mangindaan, R. E.; Rotinsulu, H.; Kobayashi, H.; Namikoshi, M. J. Nat. Prod. 2007, 70, 439. |
| [1] | (c) Dunbar, K. L.; Scharf, D. H.; Litomska, A.; Hertweck, C. Chem. Rev. 2017, 117, 5521. |
| [1] | (d) Iino, H.; Usui, T.; Hanna, J. Nat. Commun. 2015, 6, 1. |
| [1] | (e) Roy, K. M. Ullmann?s Encycl. Ind. Chem. 2000, 36, 625. |
| [2] | Mao, Y.; Jiang, L.; Chen, T.; He, H.; Liu, G.; Wang, H. Synthesis 2015, 47, 1387. |
| [3] | Chekal, B. P.; Guinness, S. M.; Lillie, B. M.; McLaughlin, R. W.; Palmer, C. W.; Post, R. J.; Sieser, J. E.; Singer, R. A.; Sluggett, G. W.; Vaidyanathan, R. Org. Process Res. Dev. 2014, 18, 266. |
| [4] | Moreno, E.; Calvo, A.; Schwartz, J.; Navarro-Blasco, I.; González-Pe?as, E.; Sanmartín, C.; Irache, J. M.; Espuelas, S. Pharmaceutics 2019, 11, 607. |
| [5] | Scott, K. A.; Njardarson, J. T. Top. Curr. Chem. 2018, 376, 1. |
| [6] | (a) Shen, C.; Zhang, P.; Sun, Q.; Bai, S.; Hor, T. S. A.; Liu, X. Chem. Soc. Rev. 2015, 44, 291. |
| [6] | (b) Zhang, B.; Kang, Y.; Shi, R. Chin. J. Org. Chem. 2016, 36, 1814 (in Chinese). |
| [6] | (张变香, 亢永强, 史瑞雪, 有机化学, 2016, 36, 1814.) |
| [6] | (c) Abedinifar, F.; Bahadorikhalili, S.; Larijani, B.; Mahdavi, M.; Verpoort, F. Appl. Organomet. Chem. 2022, 36, e6482. |
| [6] | (d) Qin, S. Chin. J. Org. Chem. 2023, 43, 3761 (in Chinese). |
| [6] | (秦思凝, 有机化学, 2023, 43, 3761.) |
| [6] | (e) Parumala, S. K. R.; Surasani, S. R.; Peddinti, R. K. New J. Chem. 2014, 38, 5268. |
| [6] | (f) Zhou, Q.; Zhang, B.; Gu, H.; Zhong, A.; Du, T.; Zhou, Q.; Ye, Y.; Jin, Z.; Jiang, H.; Chen, R. Lett. Org. Chem. 2012, 9, 175. |
| [6] | (g) Zhang, Y.; Wang, Y.; Wang, L.; Han, J. Org. Biomol. Chem. 2024, 22, 486. |
| [7] | Jang, H. Org. Biomol. Chem. 2021, 19, 8656. |
| [8] | (a) Li, Y.; Liu, L.; Shan, D.; Liang, F.; Wang, S.; Yu, L.; Liu, J.-Q.; Wang, Q.; Shao, X.; Zhu, D. ACS Catal. 2023, 13, 13474. |
| [8] | (b) Liu, Y.; Xing, S.; Zhang, J.; Liu, W.; Xu, Y.; Zhang, Y.; Yang, K.; Yang, L.; Jiang, K.; Shao, X. Org. Chem. Front. 2022, 9, 1375. |
| [8] | (c) Zhang, W.; Huang, M.; Zou, Z.; Wu, Z.; Ni, S.; Kong, L.; Zheng, Y.; Wang, Y.; Pan, Y. Chem. Sci. 2021, 12, 2509. |
| [8] | (d) Gra?l, S.; Hamze, C.; Koller, T. J.; Knochel, P. Chem.-Eur. J. 2019, 25, 3752. |
| [8] | (e) Dong, Z.-B.; Balkenhohl, M.; Tan, E.; Knochel, P. Org. Lett. 2018, 20, 7581. |
| [8] | (f) Wang, X.; Meng, J.; Zhao, D.; Tang, S.; Sun, K. Chin. Chem. Lett. 2023, 34, 107736. |
| [9] | (a) Wang, X.; Chen, J.-Q.; Yang, X.-X.; Hao, E.-J.; Dong, Z.-B. Eur. J. Org. Chem. 2022, e202200015. |
| [9] | (b) Li, F.; Wang, D.; Chen, H.; He, Z.; Zhou, L.; Zeng, Q. Chem. Commun. 2020, 56, 13029. |
| [9] | (c) Zou, L.-H.; Zhao, C.; Li, P.-G.; Wang, Y.; Li, J. J. Org. Chem. 2017, 82, 12892. |
| [9] | (d) Prasad, C. D.; Balkrishna, S. J.; Kumar, A.; Bhakuni, B. S.; Shrimali, K.; Biswas, S.; Kumar, S. J. Org. Chem. 2013, 78, 1434. |
| [9] | (e) Fernández-Salas, J. A.; Pulis, A. P.; Procter, D. J. Chem. Commun. 2016, 52, 12364. |
| [9] | (f) Xia, D.; Luo, J.; He, L.; Cai, Z.; Du, G. Chin. J. Org. Chem. 2024, 44, 622 (in Chinese). |
| [9] | (夏登鹏, 罗锦昀, 何林, 蔡志华, 杜广芬, 有机化学, 2024, 44, 622.) |
| [10] | (a) Bacchi, S.; Benaglia, M.; Cozzi, F.; Demartin, F.; Filippini, G.; Gavezzotti, A. Chem.-Eur. J. 2006, 12, 3538. |
| [10] | (b) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320. |
| [10] | (c) Berger, R.; Resnati, G.; Metrangolo, P.; Weber, E.; Hulliger, J. Chem. Soc. Rev. 2011, 40, 3496. |
| [10] | (d) Merritt, E. A.; Olofsson, B. Angew. Chem., Int. Ed. 2009, 48, 9052. |
| [10] | (e) Yoshimura, A.; Zhdankin, V. V. Chem. Rev. 2016, 116, 3328. |
| [11] | (a) Liu, C.; Zhang, B. Chem. Rec. 2016, 16, 667. |
| [11] | (b) Li, X.; Fu, B.; Zhang, Q.; Yuan, X.; Zhang, Q.; Xiong, T.; Zhang, Q. Angew. Chem., Int. Ed. 2020, 59, 23056. |
| [11] | (c) Zhao, Y.; Fu, B.; Wang, S.; Li, Y.; Yuan, X.; Yin, J.; Xiong, T. Zhang, Q. Org. Lett. 2023, 25, 2492. |
| [11] | (d) Linde, E.; Bulfield, D.; Kervefors, G.; Purkait, N.; Olofsson, B. Chem 2022, 8, 850. |
| [11] | (e) Stuart, D. R. Chem.-Eur. J. 2017, 23, 15852. |
| [11] | (f) Fa?anás-Mastral, M. Synthesis 2017, 49, 1905. |
| [12] | (a) Yang, L.; Qin, T.; Liu, B. Synlett 2023, 34, 2336. |
| [12] | (b) Liao, W.; Du, H.; Chen, M.; Xiong, Y.; Zhou, H.; Qin, T.; Liu, B. Org. Biomol. Chem. 2024, 22, 708. |
| [13] | (a) Fortugno, C.; Varchi, G.; Guerrini, A. Biomed. Anal. 2014, 95, 151. |
| [13] | (b) Gambini, L.; Udompholkul, P.; Salem, A. F.; Baggio, C.; Pellecchia, M. ChemMedChem 2020, 15, 2176. |
| [13] | (c) Tang, X.; Zhang, N.; He, G.; Li, C.-H.; Huang, W.; Wang, X.-Y.; Zhan, G.; Han, B. Org. Lett. 2020, 22, 7909. |
| [14] | (a) Mondal, S.; Di Tommaso, E. M.; Olofsson, B. Angew. Chem., Int. Ed. 2023, 62, No. e202216296. |
| [14] | (b) Wu, S.; Wong, T. H.-F.; Righi, P.; Melchiorre, P. J. Am. Chem. Soc. 2024, 146, 2907. |
| [15] | Wang, D.; Yu, X.; Zhao, K.; Li, L.; Ding, Y. Tetrahedron Lett. 2014, 55, 5739. |
/
| 〈 |
|
〉 |