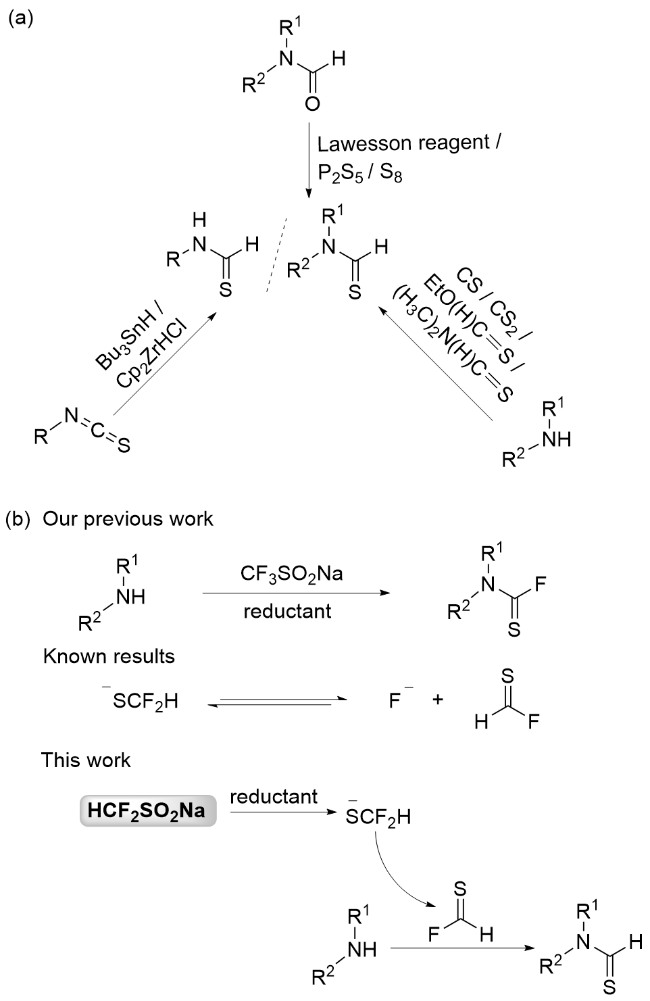
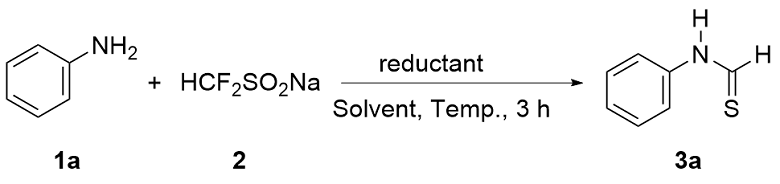
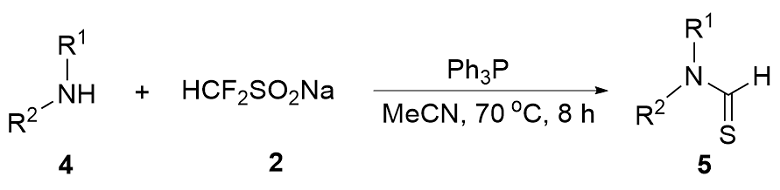
在氮气保护的10 mL反应管中, 分别加入伯胺(0.5 mmol), HCF2SO2Na (0.75 mmol), Ph3P (3 equiv.), MeCN (3 mL), 70 ℃搅拌反应3 h. 反应结束后, 冷却至常温, 向反应管中加入EtOAc (5 mL), 混合物经装有硅藻土垫的沙星漏斗过滤, 滤液浓缩后, 经硅胶柱层析(洗脱剂: 乙酸乙酯/石油醚, 除特别指出外, 体积比为1∶9)分离得产物3.
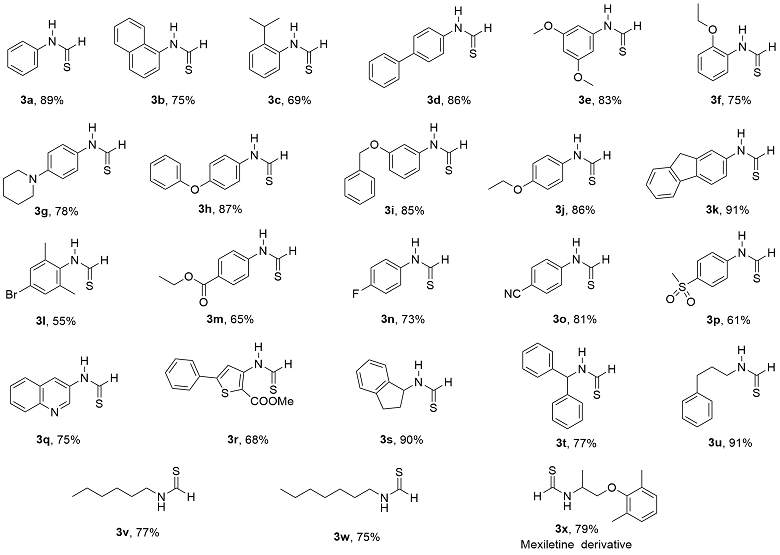
N-苯基甲硫酰胺(3a): 白色固体, 产率89% (61.0 mg). m.p. 136~137 ℃; s-cis rotamer∶s-trans rotamer≈1∶39. s-cis rotamer: 1H NMR (500 MHz, CDCl3) δ: 9.77 (d, J=10.0 Hz, 1H), 9.68 (d, J=10.0 Hz, 1H), 7.82 (d, J=10.0 Hz, 2H), 7.36 (d, J=5.0 Hz, 1H), 7.20 (d, J=10.0 Hz, 2H); 13C NMR (126 MHz, CDCl3) δ: 188.74, 139.95, 131.33, 127.73, 118.90. s-trans rotamer: 1H NMR (500 MHz, CDCl3) δ: 9.94 (d, J=35.0 Hz, 1H), 9.76 (d, J=15.0 Hz, 1H), 7.34 (d, J=10.0 Hz, 2H), 7.21 (d, J=10.0 Hz, 1H), 7.13 (d, J=10.0 Hz, 2H); 13C NMR (126 MHz, CDCl3) δ: 188.74, 139.95, 131.33, 127.73, 118.90; HRMS (EI) calcd for C7H7NS 137.0294, found 137.0296.
N-(萘-1-基)硫代甲酰胺(3b): 白色固体, 产率75% (70.1 mg). m.p. 142~144 ℃; s-cis rotamer∶s-trans rotamer≈1∶2.4. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.11 (s, 1H), 9.84 (s, 1H), 8.01~7.86 (m, 1H), 8.01~7.86 (m, 2H), 7.65~7.50 (m, 4H); 13C NMR (126 MHz, DMSO-d6) δ: 190.30, 164.47, 134.27, 134.19, 128.81, 127.82, 126.96, 126.84, 125.83, 123.88, 119.34. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.59 (s, 1H), 9.81 (s, 1H), 8.16 (d, J=10.0 Hz, 1H), 8.01~7.86 (m, 2H), 7.65~7.50 (m, 4H); 13C NMR (126 MHz, DMSO-d6) δ: 192.85, 160.77, 136.93, 134.11, 128.53, 127.38, 127.19, 127.09, 126.35, 122.92, 122.77; HRMS (EI) calcd for C11H9NS 187.0451, found 187.0452.
N-(2-异丙基苯基)硫代甲酰胺(3c): 白色固体, 产率69% (61.8 mg). m.p. 110~111 ℃. s-cis rotamer∶s-trans rotamer≈1∶1.4. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.01 (s, 1H), 10.05 (s, 1H), 7.86~7.63 (m, 4H), 3.54~3.44 (m, 1H), 1.61 (d, J=5.0 Hz, 6H); 13C NMR (126 MHz, DMSO-d6) δ: 190.59, 142.03, 136.19, 128.77, 127.98, 127.09, 124.47, 28.45, 24.13. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.35 (s, 1H), 9.92 (s, 1H), 7.86~7.63 (m, 4H), 3.77~3.66 (m, 1H), 1.60 (d, J=5.0 Hz, 6H); 13C NMR (126 MHz, DMSO-d6) δ: 192.79, 144.73, 138.76, 128.44, 127.78, 126.93, 126.89, 27.88, 24.03; HRMS (EI) calcd for C10H13NS 179.0764, found 179.0766.
N-([1'-联苯]-4-基)甲硫酰胺(3d): 白色固体, 产率86% (91.6 mg). m.p. 180~183 ℃; s-cis rotamer∶s-trans rotamer≈1∶34.3. s-cis rotamer: 1H NMR (500 MHz, CDCl3) δ: 9.73 (d, J=15.0 Hz, 1H), 9.48 (d, J=15.0 Hz, 1H), 7.50 (d, J=5.0 Hz, 2H), 7.45 (d, J=5.0 Hz, 2H), 7.34 (t, J=5.0 Hz, 2H), 7.26 (t, J=5.0 Hz, 1H), 7.11 (d, J=10.0 Hz, 2H); 13C NMR (126 MHz, CDCl3) δ: 187.06, 139.67, 139.35, 137.63, 128.94, 128.58, 127.67, 126.84, 117.77. s-trans rotamer: 1H NMR (500 MHz, CDCl3) δ: 9.91 (d, J=15.0 Hz, 1H), 9.61 (d, J=15.0 Hz, 1H), 7.84 (d, J=10.0 Hz, 2H), 7.52 (d, J=10.0 Hz, 2H), 7.34 (t, J=5.0 Hz, 2H), 7.26 (t, J=5.0 Hz, 1H), 7.13 (d, J=10.0 Hz, 2H); HRMS (EI) calcd for C13H11NS 213.0614, found 213.0611.
N-(3,5-二甲氧基苯基)硫代甲酰胺(3e): 白色固体, 产率83% (81.8 mg). m.p. 179~181 ℃; s-cis rotamer∶s-trans rotamer≈1∶11.5. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 11.35 (d, J=5.0 Hz, 1H), 8.91 (s, 1H), 7.33 (s, 2H), 7.63 (d, J=5.0 Hz, 1H), 6.72 (d, J=5.0 Hz, 1H), 6.21 (d, J=5.0 Hz, 1H), 2.82 (s, 6H); 13C NMR (126 MHz, DMSO-d6) δ: 186.68, 160.08, 140.21, 99.64, 96.82, 55.06. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 11.46 (d, J=15.0 Hz, 1H), 9,39 (d, J=10.0 Hz, 1H), 5.98 (d, J=5.0 Hz, 2H), 5.70 (d, J=5.0 Hz, 1H), 3.12 (s, 6H); 13C NMR (126 MHz, DMSO-d6) δ: 188.83, 160.99, 140.98, 97.58, 95.55, 55.16; HRMS (EI) calcd for C9H11NO2S 197.0503, found 197.0503.
N-(2-乙氧基苯基)硫代甲酰胺(3f): 白色固体, 产率75% (67.9 mg). 洗脱剂: m.p. 138~139 ℃; s-cis rotamer∶s-trans rotamer≈2∶1. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 11.45 (s, 1H), 9.83 (d, J=15.0 Hz, 1H), 7.43 (d, J=5.0 Hz, 1H), 7.27 (d, J=10.0 Hz, 1H), 7.17 (d, J=10.0 Hz, 1H), 7.01 (d, J=10.0 Hz, 1H), 4.17 (q, J=10.0 Hz, 2H), 1.45 (q, J=10.0 Hz, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 190.93, 149.87, 129.23, 127.46, 121.37, 121.14, 113.81, 64.70, 15.05. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 11.48 (s, 1H), 9.60 (d, J=5.0 Hz, 1H), 8.63 (d, J=5.0 Hz, 1H), 7.30 (d, J=10.0 Hz, 1H), 7.19 (d, J=10.0 Hz, 1H), 7.04 (d, J=10.0 Hz, 1H), 4.20 (q, J=10.0 Hz, 2H), 1.45 (q, J=10.0 Hz, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 187.92, 150.58, 127.77, 127.60, 123.84, 120.15, 113.11, 64.67, 15.15; HRMS (EI) calcd for C9H11NOS 181.0554, found 181.0554.
N-(4-(哌啶-1-基)苯基)硫代甲酰胺(3g): 黄绿色固体, 产率78% (85.8 mg). 洗脱剂: 乙酸乙酯/石油醚(V∶V=1∶7). m.p. 134~136 ℃; s-cis rotamer∶s-trans rotamer≈1∶4.8. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.95 (s, 1H), 9.43 (d, J=10.0 Hz, 1H), 7.74 (d, J=10.0 Hz, 2H), 6.93 (d, J=10.0 Hz, 2H), 3.15 (t, J=5.0 Hz, 4H), 1.68~1.61 (m, 4H), 1.59~1.52 (m, 2H); 13C NMR (126 MHz, CD3CN) δ: 186.21, 150.10, 129.34, 122.86, 115.17, 49.66 (t, J=23.9 Hz), 25.06, 23.67. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.12 (s, 1H), 9.63 (s, 1H), 7.11 (d, J=10.0 Hz, 2H), 6.91 (d, J=10.0 Hz, 2H), 3.12 (t, J=5.0 Hz, 4H), 1.68~1.61 (m, 4H), 1.59~1.52 (m, 2H); 13C NMR (126 MHz, CD3CN) δ: 186.59, 150.15, 130.68, 118.46, 116.41, 49.66 (t, J=23.9 Hz), 25.13, 23.63; HRMS (EI) calcd for C12H16N2S 220.1036, found 220.1034.
N-(4-苯氧基苯基)硫代甲酰胺(3h): 白色固体, 产率87% (99.6 mg). m.p. 90~92 ℃; s-cis rotamer∶s-trans rotamer≈1∶6.3. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.03 (s, 1H), 9.58 (d, J=5.0 Hz, 1H), 7.92 (d, J=10.0 Hz, 2H), 7.40 (d, J=10.0 Hz, 1H), 7.27 (d, J=10.0 Hz, 1H), 7.18 (t, J=10.0 Hz, 1H), 7.06~6.98 (m, 4H); 13C NMR (126 MHz, CD3CN) δ: 186.99, 156.92, 155.12, 133.78, 124.10, 123.72, 119.32, 118.83, 118.58. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.20 (s, 1H), 9.79 (d, J=15.0 Hz, 1H), 7.38 (d, J=10.0 Hz, 2H), 7.27 (d, J=10.0 Hz, 2H), 7.16 (t, J=10.0 Hz, 1H), 7.06~6.98 (m, 4H); 13C NMR (126 MHz, CD3CN) δ: 188.20, 157.05, 155.08, 135.04, 130.02, 123.65, 119.77, 118.91, 118.71; HRMS (EI) calcd for C13H11NOS 229.0563, found 229.0562.
N-(3-(苄氧基)苯基)硫代甲酰胺(3i): 白色固体, 产率85% (103.3 mg). m.p. 102~103 ℃. s-cis rotamer∶s-trans rotamer≈1∶8.3. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.99 (s, 1H), 9.57 (d, J=5.0 Hz, 1H), 7.89 (s, 1H), 7.47~7.26 (m, 5H), 6.91~6.82 (m, 3H), 5.09 (s, 2H); 13C NMR (126 MHz, CD3CN) δ: 187.43, 160.37, 140.64, 137.09, 129.69, 128.51, 127.96, 127.78, 114.54, 112.77, 108.66, 69.84. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.10 (s, 1H), 9.85 (d, J=15.0 Hz, 1H), 7.47~7.26 (m, 5H), 7.42 (s, 1H), 6.91~6.82 (m, 3H), 5.10 (s, 2H); 13C NMR (126 MHz, CD3CN) δ: 188.64, 159.78, 140.48, 136.98, 130.65, 128.55, 128.03, 127.74, 112.02, 110.02, 104.32, 69.89; HRMS (EI) calcd for C14H13NOS 243.0718, found 243.0717.
N-(4-乙氧基苯基)硫代甲酰胺(3j): 白色固体, 产率86% (77.9 mg). m.p. 124~125 ℃; s-cis rotamer∶s-trans rotamer≈1∶4.5. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.96 (s, 1H), 9.54 (d, J=10.0 Hz, 1H), 7.83(d, J=5.0 Hz, 2H), 6.97 (d, J=10.0 Hz, 2H), 4.12~4.03 (m, 2H), 1.39 (t, J=10.0 Hz, 3H); 13C NMR (126 MHz, CD3CN) δ: 186.09, 156.76, 131.00, 123.68, 113.91, 63.27, 13.73. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.13 (s, 1H), 9.72 (s, 1H), 7.23(d, J=5.0 Hz, 2H), 6.97 (d, J=10.0 Hz, 2H), 4.12~4.03 (m, 2H), 1.40 (t, J=10.0 Hz, 3H); 13C NMR (126 MHz, CD3CN) δ: 187.32, 156.81, 132.38, 118.95, 114.93, 63.38, 13.73; HRMS (EI) calcd for C9H11NOS 181.0554, found 181.0553.
N-(9H-芴-2-基)硫代甲酰胺(3k): 白色固体, 产率91% (102.4 mg). m.p. 196~198 ℃; s-cis rotamer∶s-trans rotamer~1∶7.3. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.09 (d, J=5.0 Hz, 1H), 9.49 (d, J=10.0 Hz, 1H), 8.34 (d, J=5.0 Hz, 1H), 8.25 (d, J=5.0 Hz, 1H), 7.93 (d, J=10.0 Hz, 1H), 7.88 (d, J=5.0 Hz, 1H), 7.52 (d, J=5.0 Hz, 1H), 7.33 (t, J=10.0 Hz, 1H), 7.25 (t, J=10.0 Hz, 1H), 3.96 (s, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 187.19, 145.65, 143.98, 141.59, 139.96, 138.80, 127.79, 127.56, 126.17, 121.62, 120.90, 117.35, 115.06, 37.57. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.27 (d, J=15.0 Hz, 1H), 9.99 (d, J=15.0 Hz, 1H), 7.83 (d, J=10.0 Hz, 1H), 7.82 (d, J=5.0 Hz, 1H), 7.58 (s, 1H), 7.52 (d, J=5.0 Hz, 1H), 7.38 (dd, J=5.0, 5.0 Hz, 1H), 7.32 (t, J=10.0 Hz, 1H), 7.24 (t, J=10.0 Hz, 1H), 3.93 (s, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 189.14, 146.44, 144.50, 141.54, 139.62, 139.45, 127.89, 127.63, 126.14, 121.94, 120.98, 117.32, 115.11, 37.63; HRMS (EI) calcd for C14H11NS 225.0612, found 225.0610.
N-(4-溴-2,6-二甲基苯基)硫代甲酰胺(3l): 白色固体, 产率55% (66.8 mg). m.p. 147~149 ℃; s-cis rotamer : s-trans rotamer≈1∶4.2. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.41 (s, 1H), 9.20 (s, 1H), 7.37 (s, 1H), 2.26 (s, 1H); 13C NMR (126 MHz, CD3CN) δ: 194.19, 137.17, 134.22, 131.11, 120.80, 17.41. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.59 (s, 1H), 9.50 (s, 1H), 7.36 (s, 1H), 2.19 (s, 1H); 13C NMR (126 MHz, CD3CN) δ: 189.96, 138.04, 134.76, 130.65, 120.92, 17.02; HRMS (EI) calcd for C9H10BrNS 242.9717, found 242.9720.
4-甲硫基氨基苯甲酸乙酯(3m): 白色固体, 产率65% (67.9 mg). m.p. 169~171 ℃; s-cis rotamer∶s-trans rotamer≈1∶8.6. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.23 (s, 1H), 9.69 (d, J=5.0 Hz, 1H), 8.15 (d, J=10.0 Hz, 2H), 8.06 (d, J=10.0 Hz, 2H), 4.27 (q, J=5.0 Hz, 2H), 1.38 (t, J=10.0 Hz, 3H); 13C NMR (126 MHz, CD3CN) δ: 189.68, 164.17, 143.32, 130.61, 127.91, 121.91, 61.46, 14.13. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.29 (s, 1H), 10.03 (d, J=15.0 Hz, 1H), 8.04 (d, J=5.0 Hz, 2H), 7.37 (d, J=5.0 Hz, 2H), 4.35 (q, J=5.0 Hz, 2H), 1.37 (t, J=10.0 Hz, 3H). 13C NMR (126 MHz, CD3CN) δ: 189.97, 166.02, 142.98, 131.55, 127.64, 117.38, 61.44, 14.22; HRMS (EI) calcd for C10H11NO2S 209.0508, found 209.0507.
N-(4-氟苯基)硫代甲酰胺(3n): 白色固体, 产率73% (55.6 mg). m.p. 180~182 ℃. s-cis rotamer∶s-trans rotamer≈1∶5.7. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.00 (d, J=5.0 Hz, 1H), 9.48 (d, J=5.0 Hz, 1H), 8.00~7.96 (m, 2H), 7.24~7.14 (m, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 187.94, 161.41, 159.42, 137.29, 136.20, 125.03, 124.96, 116.56, 116.38. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.17 (d, J=10.0 Hz, 1H), 9.88 (d, J=10.0 Hz, 1H), 7.40~7.36 (m, 2H), 7.24~7.14 (m, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 189.95, 161.65, 159.72, 137.31, 136.22, 120.29, 120.22, 117.43, 117.25; HRMS (EI) calcd for C7H6FNS 155.0206, found 155.0204.
N-(4-氰基苯基)硫代甲酰胺(3o): 白色固体, 产率81% (65.6 mg). m.p. 248~250 ℃; s-cis rotamer∶s-trans rotamer≈11∶1. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.20 (s, 1H), 9.67 (s, 1H), 8.27 (d, J=10.0 Hz, 2H), 7.87 (d, J=10.0 Hz, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 189.38, 142.98, 133.62, 122.02, 117.91. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.41 (s, 1H), 10.18 (s, 1H), 7.81 (d, J=10.0 Hz, 2H), 7.56 (d, J=10.0 Hz, 2H); 13C NMR (126 MHz, DMSO-d6) δ: 191.22, 143.43, 134.38, 119.19, 107.43; HRMS (EI) calcd for C8H6N2S 162.0247, found 162.0247.
N-(4-(甲磺酰基)苯基)硫代甲酰胺(3p): 白色固体, 产率61% (65.6 mg). m.p. 200~202 ℃; s-cis rotamer∶s-trans rotamer≈1∶7.3. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.38 (s, 1H), 9.62 (d, J=5.0 Hz, 2H), 8.19 (d, J=5.0 Hz, 2H), 7.86 (d, J=10.0 Hz, 2H), 2.98 (s, 1H); 13C NMR (126 MHz, CD3CN) δ: 188.75, 143.00, 136.14, 127.96, 121.66, 43.35. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 10.44 (s, 1H), 9.97 (d, J=15.0 Hz, 2H), 7.83 (d, J=10.0 Hz, 2H), 7.38 (d, J=10.0 Hz, 2H), 2.98 (s, 1H); 13C NMR (126 MHz, CD3CN) δ: 189.67, 142.69, 136.94, 128.91, 117.10, 43.35; HRMS (EI) calcd for C8H9NO2S2 215.0072, found 215.0072.
N-(喹啉-3-基)硫代甲酰胺(3q): 白色固体, 产率75% (70.5 mg). 洗脱剂: 乙酸乙酯/石油醚(V∶V=1∶7). m.p. 112~114 ℃; s-cis rotamer∶s-trans rotamer≈1∶4.9. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.47 (s, 1H), 9.75 (s, 1H), 9.34 (d, J=5.0 Hz, 1H), 9.08 (d, J=5.0 Hz, 1H), 8.02 (d, J=10.0 Hz, 1H), 8.00 (d, J=10.0 Hz, 1H), 7.79~7.75 (m, 1H), 7.66~7.60 (m, 1H); 13C NMR (126 MHz, DMSO-d6) δ: 189.80, 147.17, 145.32, 133.39, 130.19, 129.61, 129.12, 128.57, 126.83, 118.88. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.52 (s, 1H), 10.22 (s, 1H), 8.99 (d, J=5.0 Hz, 1H), 8.37 (d, J=5.0 Hz, 1H), 8.00 (d, J=10.0 Hz, 1H), 7.92 (d, J=10.0 Hz, 1H), 7.74~7.68 (m, 1H), 7.66~7.60 (m, 1H); 13C NMR (126 MHz, DMSO-d6) δ: 191.25, 146.27, 143.88, 134.27, 129.75, 129.63, 128.69, 128.62, 128.34, 121.54; HRMS (EI) calcd for C10H8N2S 188.0403, found 188.0404.
3-甲硫氨基-5-苯基噻吩-2-甲酸甲酯(3r): 浅黄色固体, 产率68% (94.5 mg). 洗脱剂: 乙酸乙酯/石油醚(V∶V=1∶6). m.p. 158~161 ℃; s-cis rotamer∶s-trans rotamer≈5∶1. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 11.99 (d, J=10.0 Hz, 1H), 10.24 (d, J=15.0 Hz, 1H), 8.19 (s, 1H), 7.79 (d, J=10.0 Hz, 2H), 7.56~7.45 (m, 3H), 3.91 (s, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 191.40, 162.77, 149.33, 144.53, 132.11, 129.96, 129.45, 125.87, 115.98, 108.97, 52.56. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 11.39 (d, J=15.0 Hz, 1H), 9.69 (d, J=10.0 Hz, 1H), 8.98 (s, 1H), 7.73 (d, J=5.0 Hz, 2H), 7.56~7.45 (m, 3H), 3.90 (s, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 189.76, 162.10, 147.41, 142.57, 131.90, 129.72, 129.55, 119.87, 115.61, 108.50, 52.46; HRMS (ESI) calcd for C13H11NO2S2 [M+H]+ 278.0304, found 278.0301.
N-(2,3-二氢-1H-茚-1-基)甲硫酰胺(3s): 白色固体, 产率90% (79.7 mg). m.p. 124~126 ℃; s-cis rotamer∶s-trans rotamer≈1∶4. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.23 (s, 1H), 8.42 (s, 1H), 7.26~7.10 (m, 4H), 5.12 (t, J=10.0 Hz, 1H), 2.97~2.90 (m, 1H), 2.87~2.73 (m, 1H), 2.56~2.42 (m, 1H), 1.98~1.81 (m, 1H); 13C NMR (126 MHz, CD3CN) δ: 190.44, 143.10, 141.11, 128.13, 124.64, 123.79, 64.46, 32.59, 29.65. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.32 (s, 1H), 8.59 (s, 1H), 7.26~7.10 (m, 4H), 6.04 (q, J=10.0 Hz, 1H), 2.97~2.90 (m, 1H), 2.87~2.73 (m, 1H), 2.56~2.42 (m, 1H), 1.98~1.81 (m, 1H). 13C NMR (126 MHz, CD3CN) δ: 188.20, 143.63, 141.57, 127.88, 124.53, 123.77, 57.17, 31.63, 29.36; HRMS (EI) calcd for C10H11NS 177.0607, found 177.0606.
N-二苯甲硫酰胺(3t): 白色固体, 产率77% (87.4 mg). m.p. 109~111 ℃. s-cis rotamer∶s-trans rotamer≈1∶10. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.25 (s, 1H), 9.04 (s, 1H), 7.35~7.20 (m, 10H), 5.98 (s, 1H); 13C NMR (126 MHz, CD3CN) δ: 191.65, 139.86, 128.47, 127.63, 127.14, 66.37. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.42 (s, 1H), 9.17 (s, 1H), 7.35~7.20 (m, 10H), 6.83 (s, 1H); 13C NMR (126 MHz, CD3CN) δ: 188.00, 139.98, 128.37, 127.32, 116.94, 59.38; HRMS (EI) calcd for C14H13NS 227.0769, found 227.0767.
N-(3-苯基丙基)甲硫酰胺(3u): 无色液体, 产率91% (81.4 mg). s-cis rotamer∶s-trans rotamer≈1∶4. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.19 (d, J=10.0 Hz, 1H), 8.46 (s, 1H), 7.43~7.28 (m, 5H), 3.50 (q, J=10.0 Hz, 2H), 2.75 (t, J=10.0 Hz, 2H), 2.07~2.02 (m, 2H); 13C NMR (126 MHz, CD3CN) δ: 191.18, 141.05, 128.15, 128.10, 125.69, 48.39, 31.89, 31.08. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.42 (d, J=10.0 Hz, 1H), 8.55 (s, 1H), 7.43~7.28 (m, 5H), 3.71 (q, J=10.0 Hz, 2H), 2.77 (t, J=10.0 Hz, 2H), 2.07~2.02 (m, 2H); 13C NMR (126 MHz, CD3CN) δ: 188.45, 141.34, 128.10, 128.06, 125.63, 42.06, 32.36, 28.82; HRMS (EI) calcd for C10H13NS 179.0762, found 179.0759.
N-己基甲硫酰胺(3v): 无色液体, 产率77% (55.86 mg). s-cis rotamer∶s-trans rotamer≈1∶6.3. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 10.11 (s, 1H), 9.08 (d, J=14.6 Hz, 1H), 3.32 (q, J=6.7 Hz, 2H), 1.48 (t, J=6.8 Hz, 2H), 1.29 (d, J=14.1 Hz, 6H), 0.87 (t, J=6.7 Hz, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 191.12, 49.45, 31.20, 30.01, 25.96, 14.33. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 10.21 (s, 1H), 9.22 (d, J=6.5 Hz, 1H), 3.51 (q, J=6.7 Hz, 2H), 1.56 (q, J=7.1 Hz, 2H), 1.29 (d, J=14.1 Hz, 6H), 0.87 (t, J=6.7 Hz, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 188.35, 43.01, 31.34, 27.53, 26.54, 22.47, 14.35; HR-MS (EI) calcd for C7H15NS 145.0920, found 145.0918.
N-庚基甲硫酰胺(3w): 无色液体, 产率75% (59.67 mg). s-cis rotamer∶s-trans rotamer≈1∶6.3. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 10.11 (s, 1H), 9.08 (d, J=14.7 Hz, 1H), 3.33~3.29 (m, 2H), 1.48 (t, J=7.0 Hz, 2H), 1.33~1.21 (m, 8H), 0.90~0.81 (m, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 191.14, 49.44, 30.05, 28.66, 26.26, 22.48. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 10.20 (s, 1H), 9.21 (dd, J=6.5, 1.1 Hz, 1H), 3.51 (q, J=6.5, 5.7 Hz, 2H), 1.55 (t, J=7.1 Hz, 2H), 1.31~1.23 (m, 8H), 0.88~0.84 (m, 3H); 13C NMR (126 MHz, DMSO-d6) δ: 188.35, 42.99, 31.64, 28.78, 27.57, 26.83, 22.48, 14.40; HR-MS (EI) calcd for C8H17NS 159.1076, found 159.1075.
N-(1-(2,6-二甲基苯氧基)丙-2-基)甲硫酰胺(3x): 黄色液体, 产率79% (88.12 mg). s-cis rotamer∶s-trans rotamer≈1∶3.2. s-cis rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.31 (d, J=15.0 Hz, 1H), 8.50 (s, 1H), 7.05 (d, J=5.0 Hz, 2H), 6.95 (t, J=10.0 Hz, 1H), 4.16~4.09 (m, 1H), 3.77 (ddd, J=40.0, 10.0, 5.0 Hz, 2H), 2.27 (s, 6H), 1.36(d, J=5.0 Hz, 3H); 13C NMR (126 MHz, CD3CN) δ: 191.4, 154.8, 130.8, 128.9, 124.2, 74.1, 56.0, 15.9, 15.4. s-trans rotamer: 1H NMR (500 MHz, CD3CN) δ: 9.40 (d, J=10.0 Hz, 1H), 8.67 (s, 1H), 7.05 (d, J=5.0 Hz, 2H), 6.95 (t, J=10.0 Hz, 1H), 5.07~4.97 (m, 1H), 3.89 (ddd, J=20.0, 10.0, 5.0 Hz, 2H), 2.28 (s, 6H), 1.45(d, J=5.0 Hz, 3H); 13C NMR (126 MHz, CD3CN) δ: 188.5, 155.0, 130.9, 128.9, 124.1, 72.3, 48.6, 15.4, 15.3; HR-MS (EI) calcd for C12H17NOS 223.1025, found 223.1024.
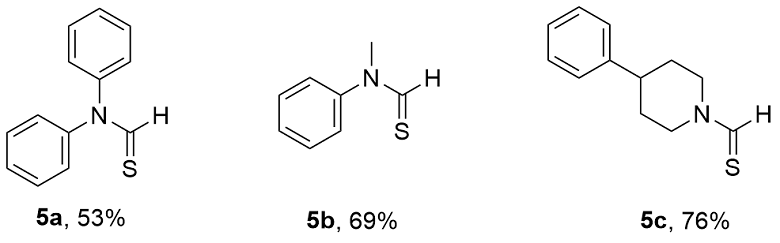
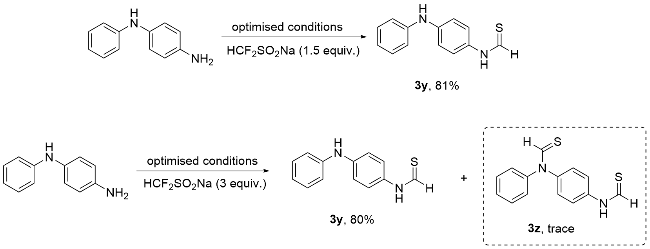
N-(4-(苯氨基)苯基)甲硫酰胺(3y): 无色固体, 产率81% (92.37 mg). s-cis rotamer∶s-trans rotamer≈1∶3.7. s-cis rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 11.85 (d, J=7.0 Hz, 1H), 9.38 (d, J=6.9 Hz, 1H), 8.30 (s, 1H), 7.90 (d, J=8.9 Hz, 2H), 7.30~7.20 (m, 2H), 7.09 (d, J=8.7 Hz, 4H), 6.86 (s, 1H); 13C NMR (126 MHz, DMSO-d6) δ: 123.36, 117.32, 116.59. s-trans rotamer: 1H NMR (500 MHz, DMSO-d6) δ: 12.10 (d, J=14.0 Hz, 1H), 9.76 (d, J=14.1 Hz, 1H), 8.22 (s, 1H), 7.29~7.21 (m, 4H), 7.05 (dd, J=8.2, 3.9 Hz, 4H), 6.83 (t, J=7.4 Hz, 1H); 13C NMR (126 MHz, DMSO-d6) δ: 186.79, 143.72, 141.59, 132.81, 129.68, 120.23, 119.05, 117.94, 117.08; HR-MS (EI) calcd for C13H12N2S 228.0716, found 228.0716.