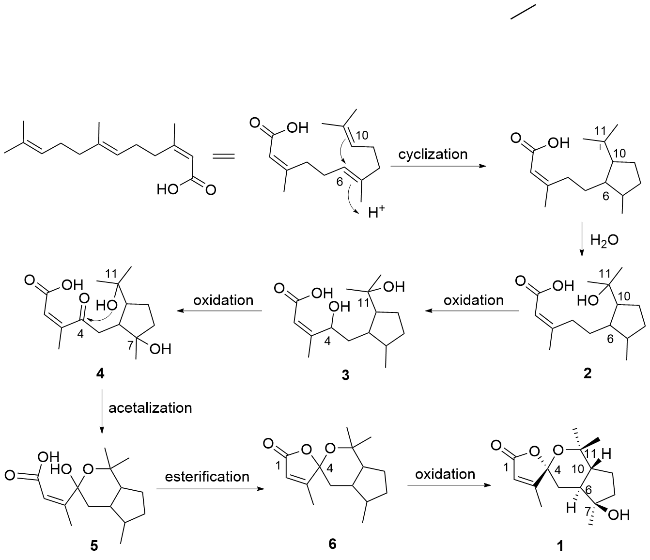
Compound
1 was obtained as a colorless needle-like crystal, and its molecular formula was determined to be C
15H
22O
4 based on the HRESIMS ([M-H]
- at
m/
z 265.1471, calcd for C
15H
21O
4 m/
z 265.1445), which required five degrees of unsaturation.
1H NMR (
Table 1) and HSQC spectra exhibited three singlet methyls at
δH 1.26, 1.28, and 1.33 (s, each 3H); one doublet methyl at
δH 2.01 (d,
J=1.5 Hz, 3H); three methylene groups at
δH 1.63~1365 and 1.8~1.80 (m, each 1H),
δH 1.34~1.36 and 1.88~1.91 (m, each 1H), and
δH 1.75 and 1.85 (overlapped, each 1H); two methine groups at
δH 1.75 and 1.85 (overlapped, each 1H); and one olefinic proton at
δH 5.77 (q,
J=1.5 Hz, 1H). The
13C NMR and DEPT spectra exhibited 15 carbon resonances, including four methyl carbons at
δC 12.9, 22.1, 27.0, and 30.6; three methylene carbons at
δC 24.8, 31.8, and 40.4; two methane carbons at
δC 43.5 and 49.0; three oxygenated quaternary carbons at
δC 79.5, 80.6, and 108.8; and three sp
2 hybridized carbons at
δC 117.9, 168.4, and 171.1. The
13C NMR data indicated a total of 15 carbon atoms, while the
1H NMR data revealed the presence of multiple methyl groups suggested that the skeleton of compound
1 should be sesquiterpene.
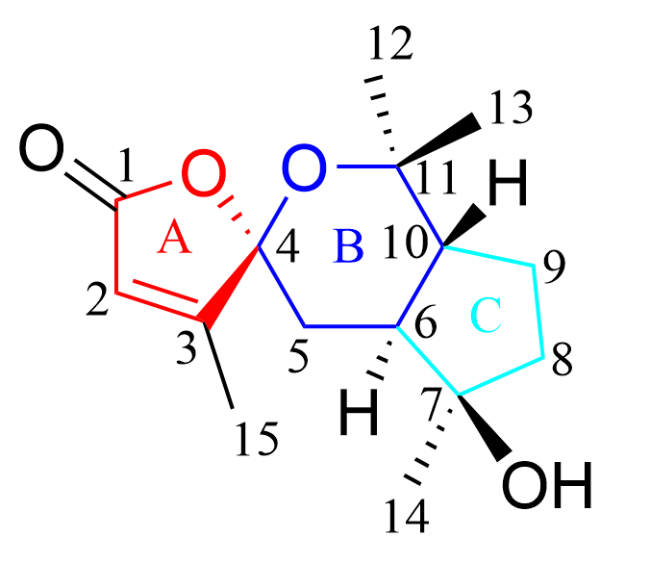
[19] The presence of an unsaturated five-membered lactone moiety
[20] (subunit A in
Figure 1) was confirmed by
1H-
1H correlated spectroscopy (COSY) correlations of H-2 (
δH 5.77) with 15-CH
3 (
δH 2.01), together with the heteronuclear multiple bond correlation (HMBC) correlations from H-2 to C-1 (
δC 171.1), C-3 (
δC 168.4), and C-4 (
δC 108.8), from 15-CH
3 to C-2 (
δC 117.9), C-3, and C-4 (
Figure 2). Furthermore, the HMBC correlations from H-5 (
δH 1.64) to C-4 (
δC 108.8), C-6 (
δC 43.5), and C-10 (
δC 49.0), from 12-CH
3 (
δH 1.28) and 13-CH
3 (
δH 1.26) to C-11 (
δC 80.6) and C-10, in conjunction with the nuclear Overhauser effect spectroscopy (NOESY) interactions (
Figure 2) of 13-CH
3 and 15-CH
3, demonstrated that the six-membered spiro-heterocycle (subunit B in
Figure 1) was connected to C-4. The C-7 signal (
δC 79.5), together with HMBC correlations from 14-CH
3 (
δH 1.33) to C-6, C-7, and C-8 (
δC 40.4), suggested that C-7 was an oxygenated carbon, and 14-CH
3 connected to C-7. These data accounted for only four degrees of unsaturation, necessitating the addition of one more ring in the molecule. Further analysis of the
13C NMR spectrum of compound
1 implied the presence of a cyclopentane moiety (subunit C in
Figure 1). Thus, compound
1 was characterized as a rare sesquiterpene featuring a unique spiro-heterocyclic skeleton. The NOESY correlations of 15-CH
3 with 13-CH
3, 15-CH
3 with H-10, 12-CH
3 with
δH 1.64 (H-5
α), H-5
α with 14-CH
3, and 14-CH
3 with H-6, indicated that 15-CH
3, 13-CH
3 and H-10 were in the
β-orientation, while 12-CH
3, 14-CH
3 and H-6 were in the
α-orientation. The relative configuration of compound
1, (4
R*,6
S*,7
R*,10
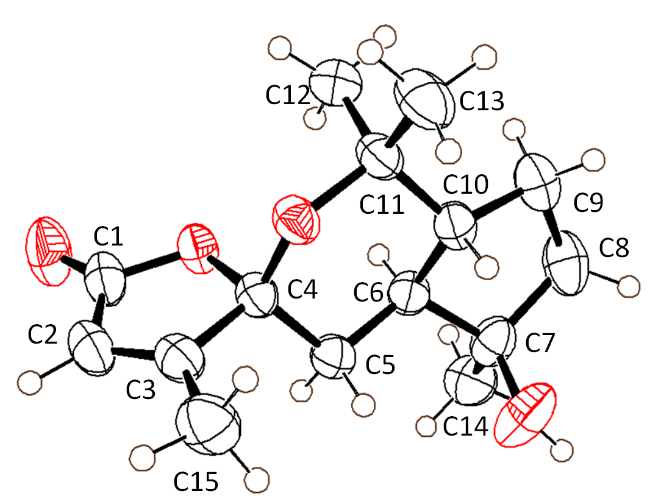
R*), was established using single crystal X-ray diffraction (
Figure 3) (CCDC No. 2403747). The CD spectrum of the compound displayed a straight line, leading to the presumption that the compound was a racemate. Therefore, the structure of compound
1 was determined and named hawkteasesquioid A.




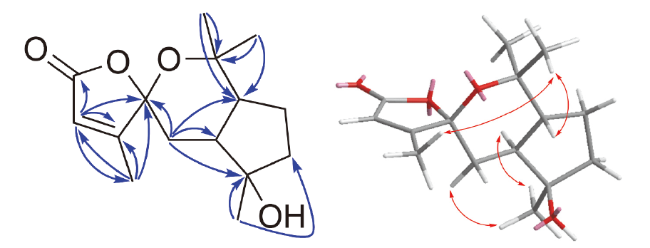
 ) and NOESY (
) and NOESY ( ) correlations of compound 1
) correlations of compound 1