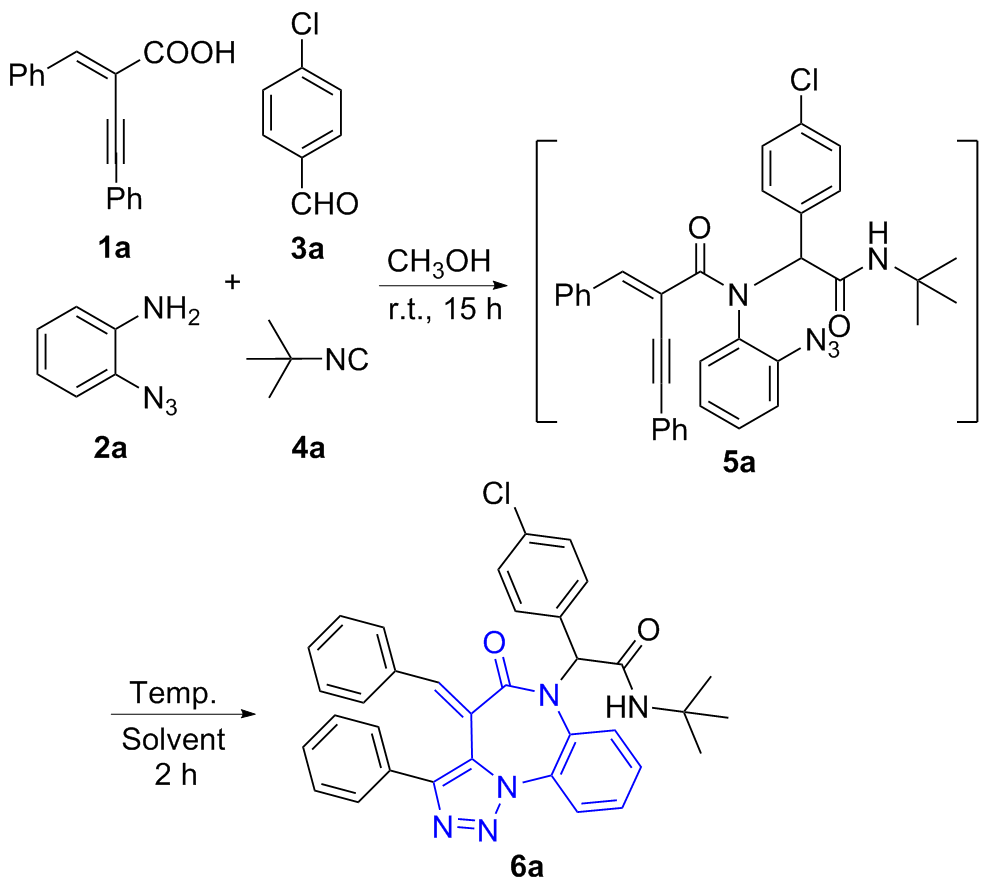
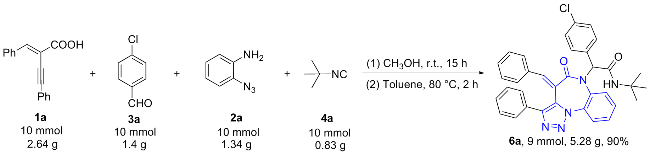(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(4-chlorophenyl)acetamide (6a): White solid (517 mg, yield 88%), m.p. 235~236 ℃; 1H NMR (CDCl3, 400 MHz) δ: 8.03 (d, J=8 Hz, 1H), 7.85 (d, J=8 Hz, 1H), 7.73 (d, J=6.8 Hz, 2H), 7.44 (s, 1H), 7.25~7.13 (m, 7H), 7.08 (t, J=7.0 Hz, 1H), 7.02~6.93 (m, 6H), 5.85 (s, 1H), 5.72 (s, 1H), 1.50 (s, 9H); 13C NMR (CDCl3, 151 MHz) δ: 171.2, 168.3, 144.0, 141.5, 134.3, 133.0, 132.1, 131.1, 130.3, 130.0, 129.6, 129.0, 128.9, 128.8, 128.4, 128.3, 127.7, 126.2, 123.4, 120.8, 66.8, 52.2, 28.7; HRMS (ESI) calcd for C35H30ClN5O2Na [M+Na]+ 610.1980, found 610.1982.
(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(p-tolyl)acetamide (6b): White solid (454 mg, yield 80%), m.p. 265~266 ℃; 1H NMR (CDCl3, 600 MHz) δ: 8.06 (d, J=8.4 Hz, 1H), 7.82 (d, J=7.8 Hz, 1H), 7.74 (d, J=7.2 Hz, 2H), 7.43 (s, 1H), 7.21~7.16 (m, 5H), 7.07 (t, J=7.2 Hz, 1H), 7.00~6.94 (m, 6H), 6.90 (d, J=8.4 Hz, 2H), 5.75 (s, 2H), 2.21 (s, 3H), 1.50 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 171.1, 168.8, 144.0, 141.1, 138.1, 133.2, 131.1, 130.6, 130.5, 129.8, 129.3, 128.9, 128.8, 128.7, 128.3, 128.2, 127.4, 126.3, 123.2, 121.2, 67.5, 52.0, 28.7, 21.0; HRMS (ESI) calcd for C36H33- N5O2Na [M+Na]+ 590.2526, found 590.2530.
(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-but yl)-2-(thiophen-2-yl)acetamide (6c): White solid (475 mg, yield 85%), m.p. 232~233 ℃; 1H NMR (CDCl3, 600 MHz) δ: 7.97 (d, J=7.2 Hz, 1H), 7.91 (d, J=6.0 Hz, 1H), 7.72 (d, J=7.2 Hz, 2H), 7.44 (s, 1H), 7.34~7.30 (q, J=7.8 Hz, 2H), 7.21~7.16 (m, 4H), 7.08 (t, J=6.6 Hz, 1H), 7.00 (t, J=7.2 Hz, 2H), 6.94 (t, J=6.9 Hz, 3H), 6.82 (s, 1H), 6.23 (s, 1H), 6.01 (s, 1H), 1.47 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 171.0, 167.6, 144.1, 141.6, 135.8, 133.1, 131.3, 130.7, 130.1, 130.0, 129.7, 129.3, 129.2, 128.9, 128.4, 128.2, 128.1, 127.9, 127.2, 126.4, 126.2, 123.6, 121.0, 63.0, 52.0, 28.6; HRMS (ESI) calcd for C33H29N5O2SNa [M+Na]+ 582.1934, found 582.1937.
(
E)-2-(4-Benzylidene-5-
oxo-3-phenyl-4,5-dihydro-6
H- benzo[
b][
1,
2,
3]triazolo[1,5-
d][1,4]diazepin-6-yl)-
N-(
tert-butyl)-2-(furan-2-yl)acetamide (
6d): White solid (467 mg, yield 86%), m.p. 233~234 ℃;
1H NMR (CDCl
3, 600 MHz)
δ: 7.97 (d,
J=7.8 Hz, 1H), 7.93 (d,
J=7.2 Hz, 1H), 7.71 (d,
J=7.2 Hz, 2H), 7.42 (s, 1H), 7.36~7.31 (m, 2H), 7.21~7.14 (m, 4H), 7.08 (t,
J=7.2 Hz, 1H), 7.00 (t,
J=7.2 Hz, 2H), 6.94 (d,
J=6.6 Hz, 2H), 6.45 (s, 1H), 6.17 (s, 1H), 6.15 (s, 1H), 5.84 (s, 1H), 1.48 (s, 9H);
13C NMR (CDCl
3, 101 MHz)
δ: 170.7, 166.5, 146.4, 144.1, 143.4, 141.4, 133.1, 130.8, 130.5, 130.0, 129.9, 129.8, 128.9, 128.4, 128.3, 128.2, 127.7, 127.2, 126.4, 123.6, 121.0, 112.6, 110.3, 61.6, 52.0, 28.6; HRMS (ESI) calcd for C
33H
29N
5O
3Na [M+Na]
+ 566.2163, found 566.2165.
(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-phenylacetamide(6e): White solid (454 mg, yield 82%), m.p. 200~201 ℃; 1H NMR (CDCl3, 600 MHz) δ: 8.04 (d, J=7.8 Hz, 1H), 7.82 (d, J=7.2 Hz, 1H), 7.75 (d, J=7.2 Hz, 2H), 7.45 (s, 1H), 7.21~7.15 (m, 8H), 7.08~6.94 (m, 7H), 5.79 (s, 2H), 1.51 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 171.2, 168.6, 144.0, 141.2, 133.6, 133.1, 131.2, 130.6, 130.4, 129.8, 129.0, 128.9, 128.8, 128.6, 128.4, 128.3, 128.2, 127.4, 126.3, 123.2, 121.2, 67.7, 52.1, 28.7; HRMS (ESI) calcd for C35H31N5O2Na [M+Na]+ 576.2370, found 576.2372.
(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(m-tolyl)acetamide (6h): White solid (454 mg, yield 80%), m.p. 205~206 ℃; 1H NMR (CDCl3, 600 MHz) δ: 8.05 (d, J=8.4 Hz, 1H), 7.82 (d, J=7.8 Hz, 1H), 7.76 (d, J=7.2 Hz, 2H), 7.44 (s, 1H), 7.22~7.15 (m, 5H), 7.08~7.03 (m, 2H), 6.99 (t, J=7.5 Hz, 2H), 6.95 (t, J=8.4 Hz, 3H), 6.83 (d, J=7.8 Hz, 1H), 6.79 (s, 1H), 5.78 (s, 1H), 5.74 (s, 1H), 2.19 (s, 3H), 1.51 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 171.2, 168.7, 144.0, 141.2, 138.4, 133.5, 133.2, 131.3, 130.7, 130.4, 129.8, 129.7, 129.0, 128.9, 128.4, 128.3, 128.2, 127.4, 126.3, 126.0, 123.1, 121.3, 67.7, 52.1, 28.7, 21.2; HRMS (ESI) calcd for C36H33N5O2Na [M+Na]+ 590.2526, found 590.2529.
(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(3-chlorophenyl)acetamide (6i): White solid (511 mg, yield 87%), m.p. 203~204 ℃; 1H NMR (CDCl3, 600 MHz) δ: 8.02 (d, J=7.8 Hz, 1H), 7.86 (d, J=7.8 Hz, 1H), 7.73 (d, J=7.2 Hz, 2H), 7.44 (s, 1H), 7.25~7.16 (m, 6H), 7.12 (d, J=8.4 Hz, 1H), 7.08~7.05 (m, 2H), 6.99 (t, J=7.5 Hz, 2H), 6.95 (d, J=7.8 Hz, 2H), 6.80 (d, J=7.8 Hz, 1H), 5.89 (s, 1H), 5.72 (s, 1H), 1.50 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 171.2, 168.0, 144.1, 141.5, 135.7, 134.5, 133.0, 131.2, 130.5, 130.3, 130.0, 129.8, 129.7, 129.2, 129.0, 128.9, 128.5, 128.4, 128.3, 128.2, 127.8, 127.1, 126.3, 123.5, 120.9, 67.0, 52.2, 28.7; HRMS (ESI) calcd for C35H30ClN5O2Na [M+Na]+ 610.1980, found 610.1981.
(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(2-chlorophenyl)acetamide (6j): White solid (458 mg, yield 78%), m.p. 260~261 ℃; 1H NMR (CDCl3, 600 MHz) δ: 8.13 (d, J=7.8 Hz, 1H), 7.83 (d, J=7.8 Hz, 1H), 7.74 (d, J=7.8 Hz, 2H), 7.50 (d, J=7.8 Hz, 1H), 7.43 (s, 1H), 7.20 (t, J=6.9 Hz, 3H), 7.17~7.05 (m, 6H), 6.99 (t, J=7.5 Hz, 2H), 6.95 (d, J=7.2 Hz, 2H), 5.94 (s, 1H), 5.84 (s, 1H), 1.51 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 170.8, 168.5, 143.9, 140.9, 135.1, 133.1, 131.5, 131.2, 131.1, 130.1, 130.0, 129.9, 129.8, 129.0, 128.9, 128.5, 128.3, 128.1, 127.6, 126.4, 123.5, 121.2, 65.3, 52.2, 28.7; HRMS (ESI) calcd for C35H30ClN5O2Na [M+Na]+ 610.1980, found 610.1982.
(E)-2-(4-Benzylidene-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)pentanamide (6k): White solid (467 mg, yield 90%), m.p. 135~136 ℃; 1H NMR (CDCl3, 600 MHz) δ: 8.06 (d, J=7.8 Hz, 1H), 7.84 (d, J=7.8 Hz, 1H), 7.68 (d, J=7.8 Hz, 2H), 7.55~7.50 (m, 2H), 7.39 (s, 1H), 7.29 (s, 1H), 7.21~7.15 (m, 3H), 7.09 (t, J=7.5 Hz, 1H), 7.01 (t, J=7.5 Hz, 2H), 6.94 (d, J=7.8 Hz, 2H), 4.77 (t, J=7.5 Hz, 1H), 1.78~1.72 (m, 1H), 1.47 (s, 9H), 1.28~1.23 (m, 1H), 1.02~0.94 (m, 1H), 0.89~0.81 (m, 1H), 0.65 (t, J=7.2 Hz, 3H); 13C NMR (CDCl3, 101 MHz) δ: 172.7, 170.4, 143.9, 141.9, 132.9, 131.0, 130.2, 130.1, 130.0, 129.7, 129.6, 128.8, 128.4, 128.3, 128.2, 128.1, 126.4, 126.3, 124.3, 121.1, 61.3, 51.3, 29.8, 28.7, 18.9, 13.4; HRMS (ESI) calcd for C32H33N5O2Na [M+Na]+ 542.2526, found 542.2529.
(
E)-2-(4-Benzylidene-5-
oxo-3-(
p-tolyl)-4,5-dihydro-6
H-benzo[
b][1,2,3]triazolo[5-d][
1,
4]diazepin-6-yl)-
N-(
tert-butyl)-2-(4-chlorophenyl)acetamide (
6m): White solid (463 mg, yield 77%), m.p. 221~222 ℃;
1H NMR (CDCl
3, 600 MHz)
δ: 8.02 (d,
J=8.4 Hz, 1H), 7.83 (d,
J=7.8 Hz, 1H), 7.64 (d,
J=7.8 Hz, 2H), 7.41 (s, 1H), 7.24 (d,
J=7.8 Hz, 1H), 7.19 (t,
J=8.1 Hz, 1H), 7.13 (d,
J=8.4 Hz, 2H), 7.09 (t,
J=7.2 Hz, 1H), 7.01 (t,
J=7.5 Hz, 4H), 6.98~6.96 (m, 4H), 5.86 (s, 1H), 5.71 (s, 1H), 2.25 (s, 3H), 1.49 (s, 9H);
13C NMR (CDCl
3, 101 MHz)
δ: 171.3, 168.3, 144.3, 141.2, 138.1, 134.3, 133.1, 132.2, 131.2, 130.5, 130.3, 130.0, 129.9, 129.1, 129.0, 128.8, 128.4, 128.3, 127.7, 126.8, 126.2, 123.5, 121.0, 66.8, 52.2, 28.7, 21.2; HRMS (ESI) calcd for C
36H
32ClN
5O
2Na [M+Na]
+ 624.2137, found 624.2138.
(E)-2-(4-Benzylidene-3-(4-fluorophenyl)-5-oxo-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(4-chlorophenyl)acetamide (6n): White solid (551 mg, yield 91%), m.p. 170~171 ℃; 1H NMR (CDCl3, 600 MHz) δ: 8.03 (d, J=8.4 Hz, 1H), 7.85 (d, J=7.8 Hz, 1H), 7.69 (dd, J=9.0, 5.4 Hz, 2H), 7.44 (s, 1H), 7.21 (t, J=8.7 Hz, 1H), 7.14 (d, J=8.4 Hz, 2H), 7.10 (t, J=7.2 Hz, 1H), 7.03~6.87 (m, 9H), 5.83 (s, 1H), 5.71 (s, 1H), 1.50 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 171.1, 168.2, 162.7 (d, J=249.5 Hz), 143.1, 141.6, 134.4, 133.0, 132.1, 131.1, 130.4, 130.3, 130.1, 129.1, 128.8, 128.6, 128.4, 128.2 (d, J=9.1 Hz), 127.8, 125.9 (d, J=3.0 Hz), 123.4, 120.9, 115.4 (d, J=21.2 Hz), 66.9, 52.2, 28.7; HRMS (ESI) calcd for C35H29ClFN5O2Na [M+Na]+ 628.1886, found 628.1887.
(E)-2-(4-Benzylidene-9-methyl-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(4-chlorophenyl)acetamide (6o): White solid (553 mg, yield 92%), m.p. 236~237 ℃; 1H NMR (CDCl3, 600 MHz) δ: 7.88 (d, J=8.4 Hz, 1H), 7.73 (d, J=7.2 Hz, 2H), 7.65 (s, 1H), 7.42 (s, 1H), 7.21~7.14 (m, 5H), 7.08 (t, J=7.2 Hz, 1H), 7.01~6.93 (m, 7H), 5.83 (s, 1H), 5.70 (s, 1H), 2.33 (s, 3H), 1.49 (s, 9H); 13C NMR (CDCl3, 101 MHz) δ: 171.3, 168.3, 144.0, 141.3, 138.1, 134.3, 133.1, 132.2, 130.8, 130.4, 130.3, 129.9, 129.7, 128.9, 128.8, 128.4, 128.3, 128.2, 128.0, 126.3, 123.8, 121.2, 66.7, 52.1, 28.7, 20.8; HRMS (ESI) calcd for C36H32ClN5O2Na [M+Na]+ 624.2137, found 624.2138.
(E)-2-(4-Benzylidene-9-methyl-5-oxo-3-phenyl-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-2-(4-chlorophenyl)-N-cyclohexylacetamide (6q): White solid (571 mg, yield 91%), m.p. 219~220 ℃; 1H NMR (CDCl3, 600 MHz) δ: 7.91 (d, J=8.4 Hz, 1H), 7.72 (d, J=7.8 Hz, 2H), 7.65 (s, 1H), 7.40 (s, 1H), 7.22~7.13 (m, 6H), 7.07 (t, J=6.6 Hz, 1H), 7.00~6.93 (m, 6H), 5.93 (d, J=7.8 Hz, 1H), 5.79 (s, 1H), 4.00 (s, 1H), 2.32 (s, 3H), 2.10~2.01 (m, 2H), 1.75 (t, J=12.6 Hz, 3H), 1.46~1.40 (q, J=12 Hz, 2H), 1.29 (t, J=10.5 Hz, 1H), 1.22~1.19 (m, 2H); 13C NMR (CDCl3, 101 MHz) δ: 171.4, 168.1, 144.0, 141.3, 138.1, 134.3, 133.1, 132.1, 130.8, 130.3, 130.2, 129.9, 129.7, 128.9, 128.8, 128.4, 128.3, 128.2, 128.0, 127.9, 126.3, 123.8, 121.1, 66.2, 49.1, 32.9, 25.4, 24.7, 20.8; HRMS (ESI) calcd for C38H34ClN5O2Na [M+Na]+ 650.2293, found 650.2294.
(
E)-
N-Benzyl-2-(4-benzylidene-9-methyl-5-
oxo-3-phenyl-4,5-dihydro-6
H-benzo[
b][1,2,3]triazolo[1,5-
d]-[
1,
4]diazepin-6-yl)-2-(4-chlorophenyl)acetamide (
6r): White solid (572 mg, yield 90%), m.p. 202~203 ℃;
1H NMR (CDCl
3, 600 MHz)
δ: 7.88 (d,
J=8.4 Hz, 1H), 7.72 (d,
J=7.2 Hz, 2H), 7.65 (s, 1H), 7.40 (s, 1H), 7.37 (d,
J=4.2 Hz, 4H), 7.30 (t,
J=4.2 Hz, 1H), 7.21~7.16 (m, 3H), 7.13~7.07 (m, 3H), 7.02~6.94 (m, 7H), 6.42 (t,
J=5.4 Hz, 1H), 5.90 (s, 1H), 4.70 (dd,
J=14.7, 5.6 Hz, 1H), 4.62 (dd,
J=14.4, 5.4 Hz, 1H), 2.33 (s, 3H);
13C NMR (CDCl
3, 101 MHz)
δ: 171.3, 169.2, 143.9, 141.3, 138.2, 137.6, 134.4, 133.1, 131.9, 130.4, 130.2, 129.9, 129.7, 128.9, 128.8, 128.3, 128.2, 127.9, 127.8, 127.7, 127.6, 126.3, 123.9, 121.1, 65.9, 44.1, 20.8; HRMS (ESI) calcd for C
39H
30ClN
5O
2Na [M+Na]
+ 658.1980, found 658.1980.
(E)-2-(4-Benzylidene-3-butyl-5-oxo-4,5-dihydro-6H-benzo[b][1,2,3]triazolo[1,5-d][1,4]diazepin-6-yl)-N-(tert-butyl)-2-(4-chlorophenyl)acetamide (6s): White solid (528 mg, yield 91%), m.p. 140~141 ℃; 1H NMR (CDCl3, 600 MHz) δ: 7.98 (d, J=8.4 Hz, 1H), 7.82 (d, J=7.2 Hz, 1H), 7.54 (s, 1H), 7.32~7.28 (m, 2H), 7.25~7.20 (m, 3H), 7.16 (d, J=7.8 Hz, 2H), 7.10 (d, J=7.2 Hz, 2H), 6.97 (d, J=8.4 Hz, 2H), 5.82 (s, 1H), 5.67 (s, 1H), 2.32~2.27 (m, 1H), 2.15~2.10 (m, 1H), 1.48 (s, 9H), 1.40~1.36 (m, 1H), 1.23~1.15 (m, 2H), 0.91~0.88 (m, 1H), 0.79 (t, J=7.2 Hz, 3H); 13C NMR (CDCl3, 151 MHz) δ: 171.0, 168.2, 145.1, 141.3, 134.3, 133.5, 132.3, 131.6, 131.5, 130.4, 130.3, 130.0, 129.0, 128.8, 128.2, 127.7, 123.4, 121.3, 67.0, 52.1, 29.9, 28.6, 25.0, 22.4, 13.6. HRMS (ESI) calcd for C33H34ClN5O2Na [M+Na]+ 590.2293, found 590.2294.
Supporting Information Experimental procedures along with copies of NMR spectra and HRMS data (
6a~
6e,
6h~
6k,
6m~
6o,
6q~
6s). The Supporting Information is available free of charge via the Internet at
http://sioc-journal.cn/.