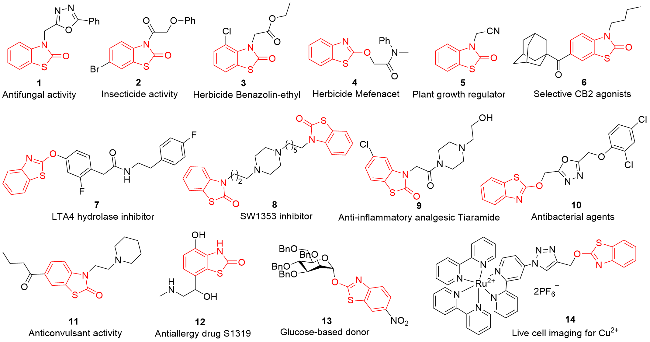
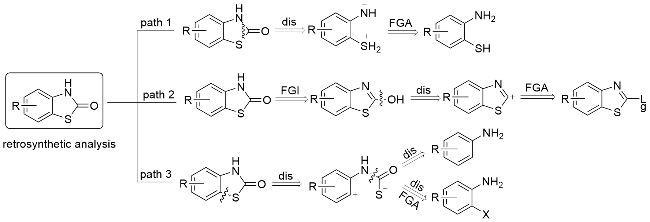
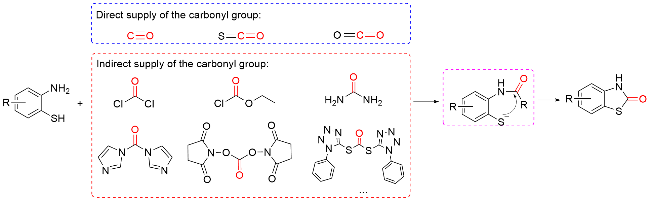
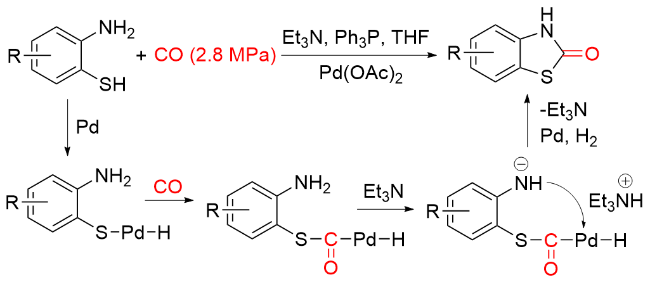
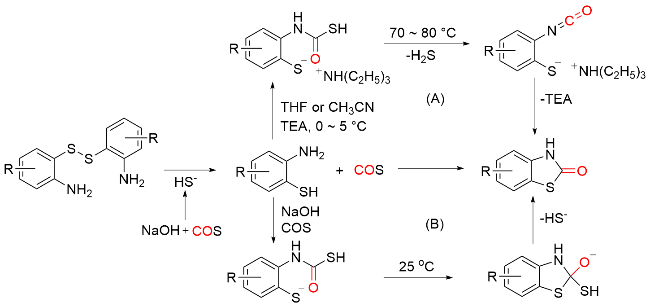
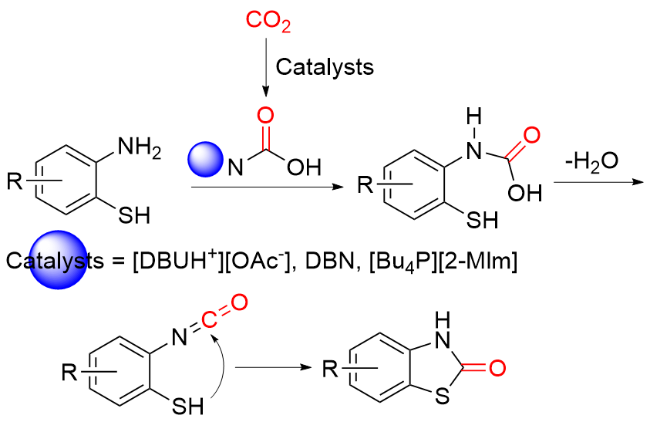
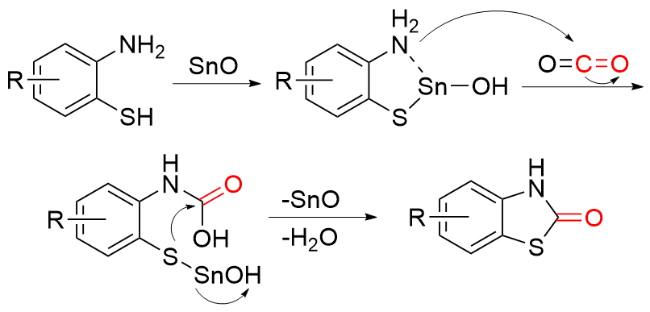
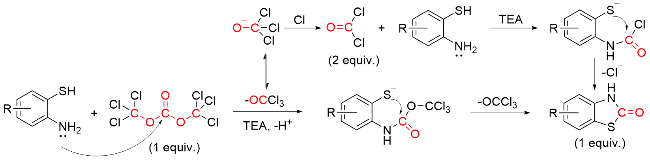
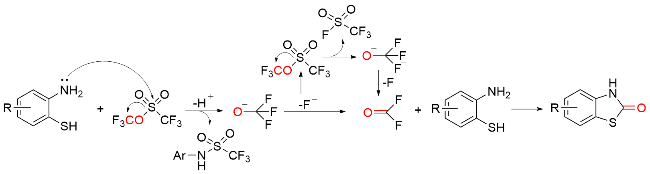
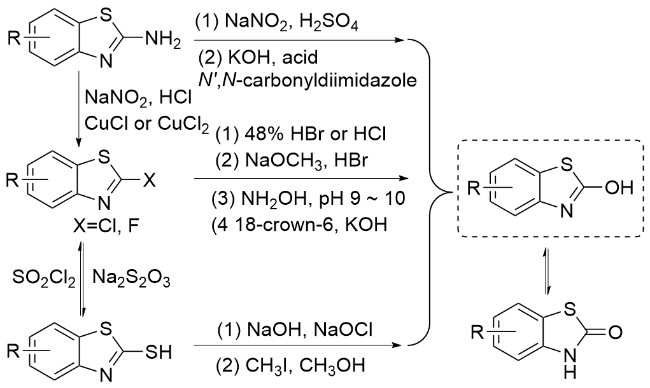
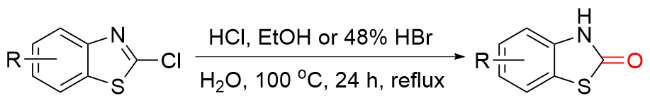
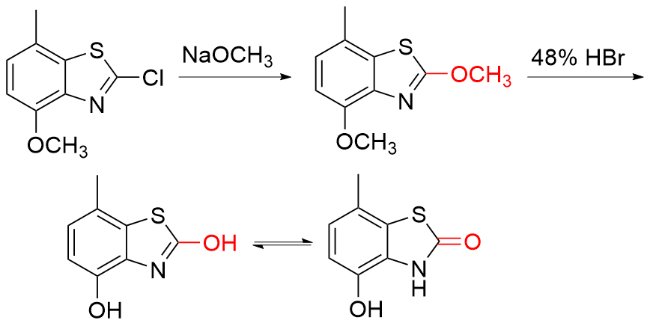
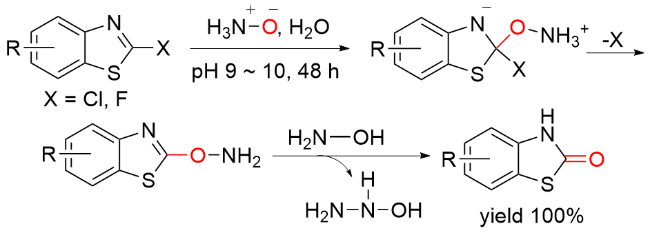
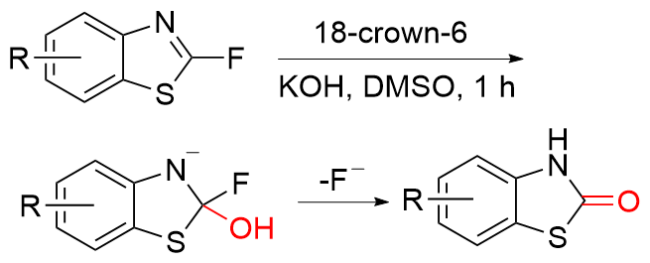
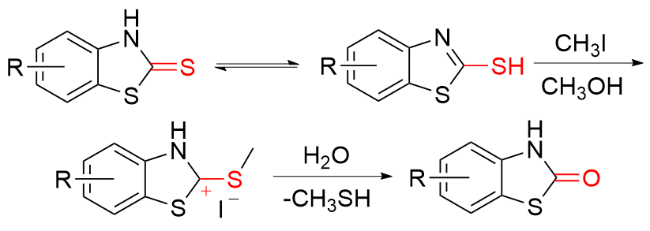
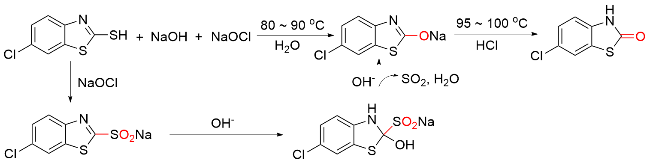
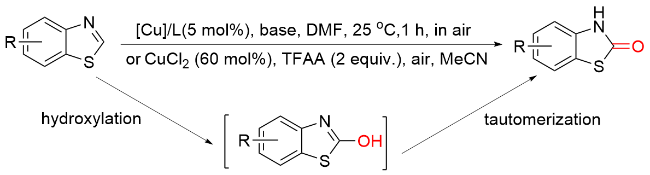
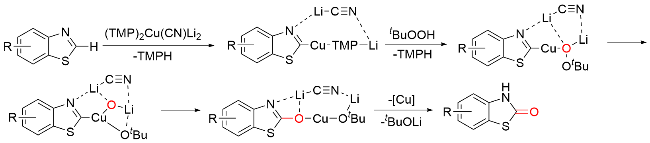
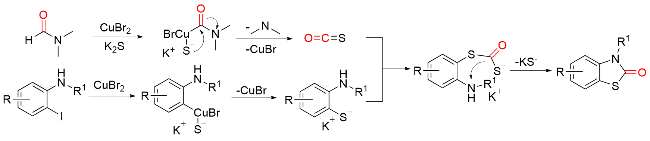
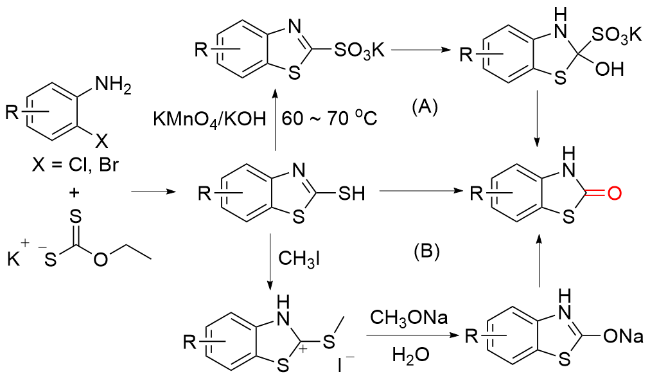
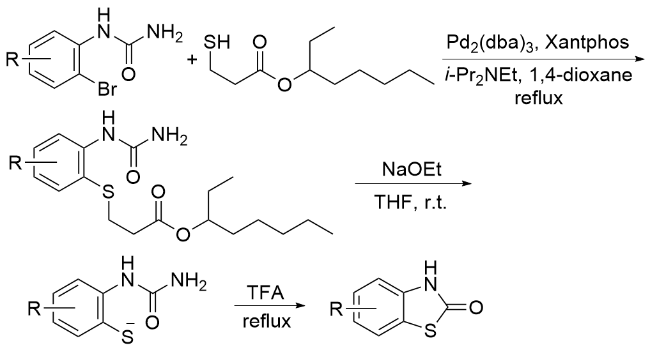
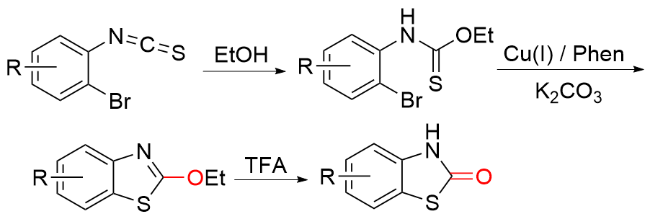
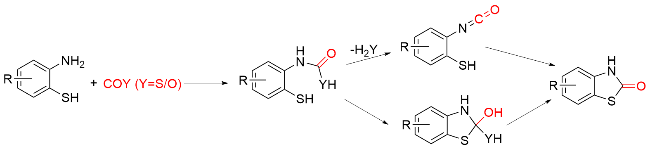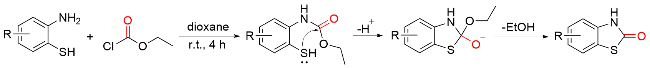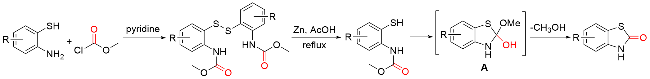1 以2-氨基苯硫酚为底物合成苯并噻唑-2-酮
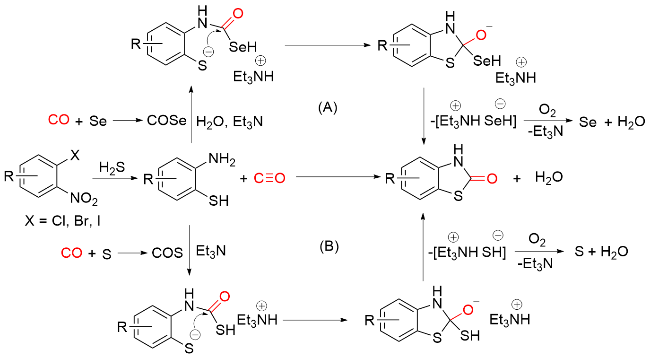
1.1 CO作为羰基化试剂
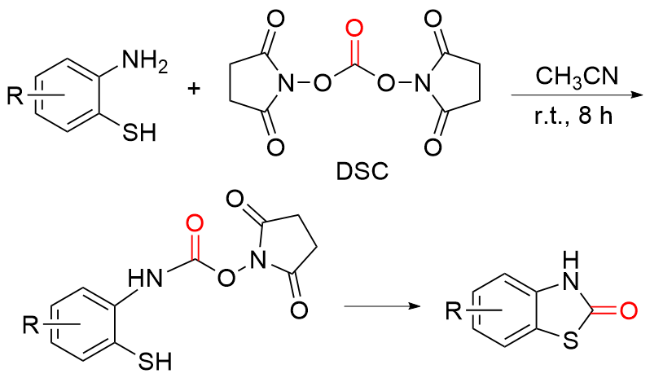
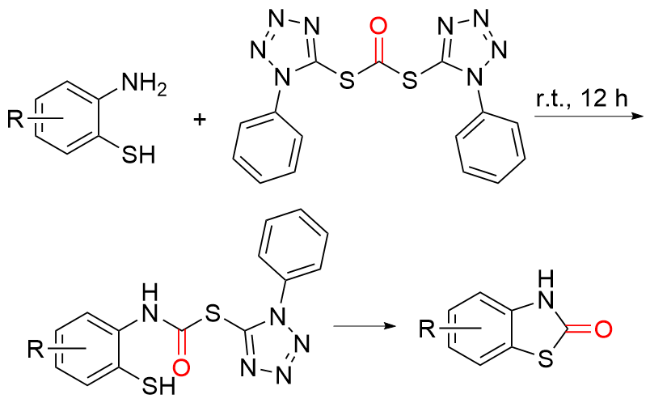
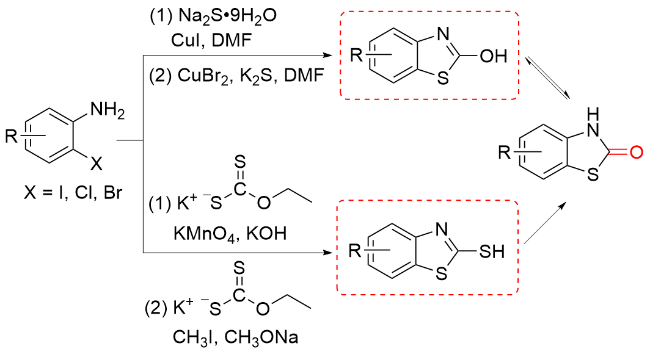
1.2 COY (Y=O/S)作为羰基化试剂
1.3 光气作为羰基化试剂
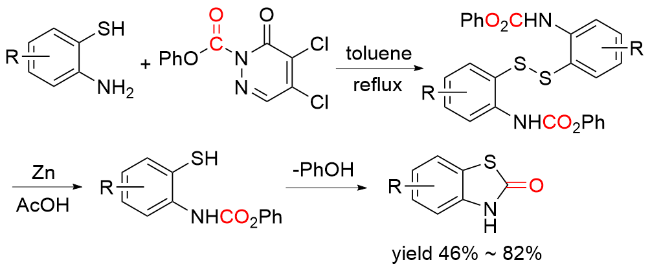
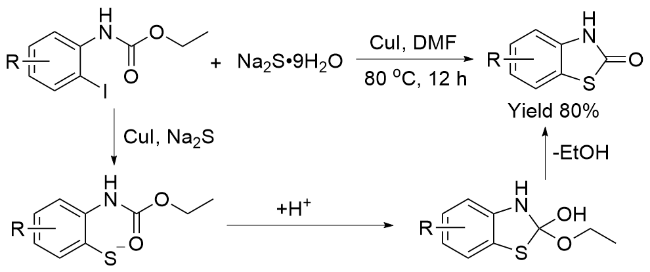
1.4 氯甲酸酯作为羰基化试剂
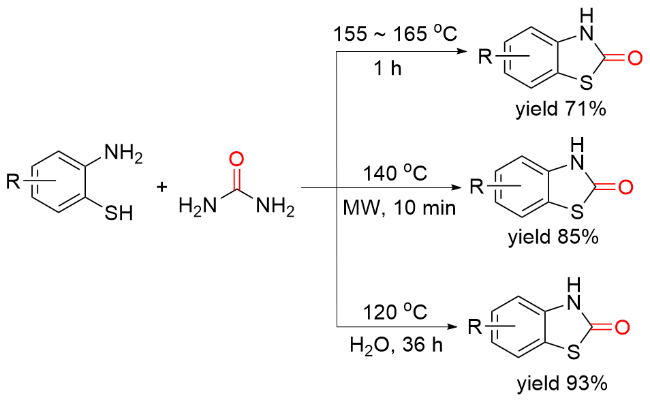
1.5 尿素作为羰基化试剂
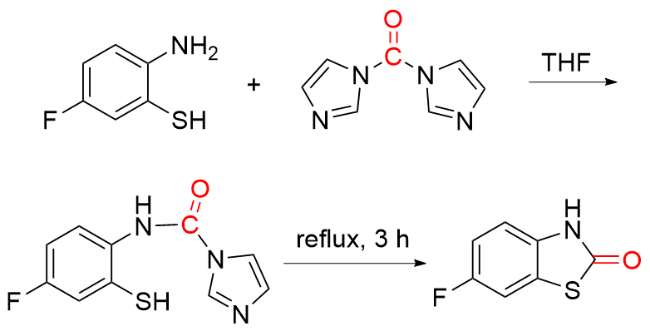
1.6 含羰基杂环化合物作为羰基化试剂
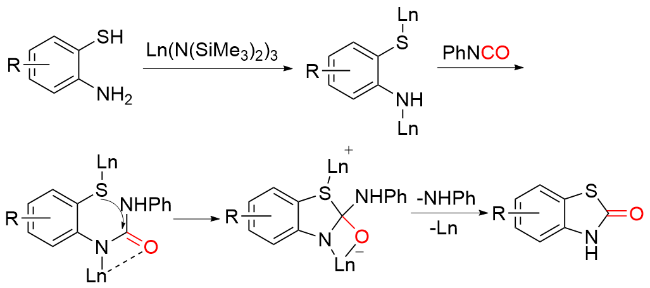
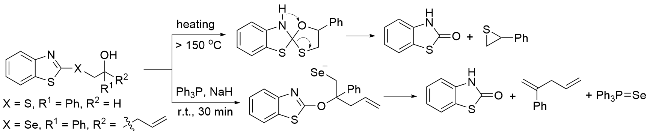
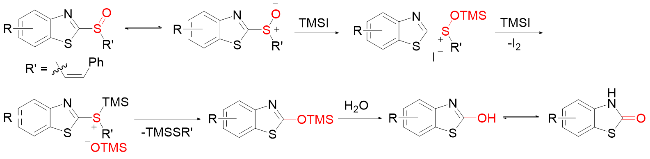
1.7 其他羰基化试剂
表1 多种羰基化试剂的特点及其性质Table 1 Carbonylation reagent for the preparation of benzothiazole-2-one using 2-aminothiophenol |
| Structure | Molecular weight | State of matter at room temperature | Carbonylation mechanism | Mass ratio of carbonyl groups in the reagent |
|---|---|---|---|---|
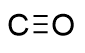 | 27.99 | Gas | CO undergoes in-situ conversion to COS/Se for participation in subsequent reactions or engages in direct reactions mediated by metal catalysis | 100% |
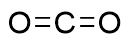 | 43.99 | Gas | The carbonyl group is attacked by a nucleophile to form an amide or ester, which then participates in subsequent reactions | 63.6% |
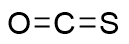 | 59.97 | Gas | The thiocarbonyl group is attacked by an amine nucleophile, forming thiocarbamate intermediates that subsequently engage in further reactions | 46.7% |
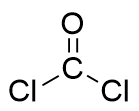 | 97.93 | Gas | The acyl chloride is attacked by an amine nucleophile, forming carbamyl chloride that subsequently engage in further reactions | 28.6% |
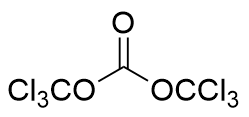 | 293.80 | Solid | Triphosgene cleaves into 3 equiv. of phosgene, which then participate in the reaction | 28.6% |
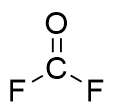 | 65.99 | Gas | The acyl fluoride is attacked by an amine nucleophile, forming carbamyl fluoride that subsequently engage in further reactions | 42.4% |
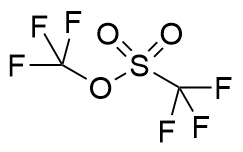 | 217.95 | Gas | The O—S bond cleavage yielded OCF3 and F, generating acyl fluoride, which then participated in the reaction | 12.8% |
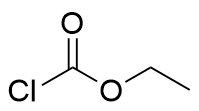 | 108.00 | Liquid | The acyl chloride is attacked by an amine nucleophile, forming carbamate that subsequently engage in further reactions | 25.9% |
 | 60.03 | Solid | The urea is attacked by an amine nucleophile, forming another urea that subsequently engage in further reactions | 46.6% |
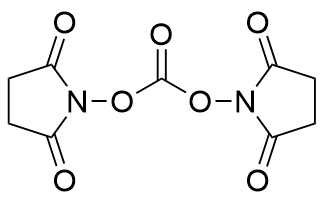 | 256.03 | Solid | The electron-withdrawing inductive effect reduces the conjugation of the ester bond, facilitating the cleavage of the C—O bond to yield the carbonyl group | 11.0% |
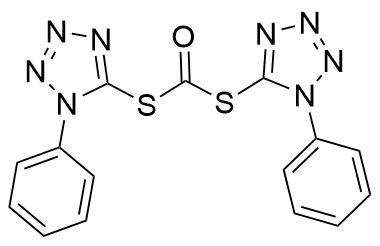 | 382.04 | Solid | The conjugation effect of the aromatic ring reduces the conjugation of the C—S bond in the thioester, promoting the cleavage of the C—S bond to form the carbonyl group | 7.4% |
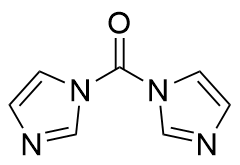 | 162.05 | Solid | The conjugation effect of the aromatic ring reduces the bond energy of the C—N bond in the amide, facilitating the cleavage of the C—N bond to yield the carbonyl group | 17.2% |
 | 283.98 | Liquid | The conjugation effect of the double bond facilitates the cleavage of the C—N bond in the amide, while the conjugation effect of the aromatic ring reduces the conjugation of the ester bond, promoting the cleavage of both the C—N and C—O bonds to yield the carbonyl group | 9.8% |
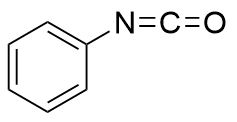 | 119.04 | Liquid | The carbon atom in isocyanate, influenced by the electron-withdrawing inductive effects of both the oxygen and nitrogen atoms, is highly susceptible to nucleophilic attack, leading to the formation of urea or carbamate | 23.5% |