1 分子间反应构建十元氮杂环化合物
1.1 串联反应构建十元氮杂环化合物
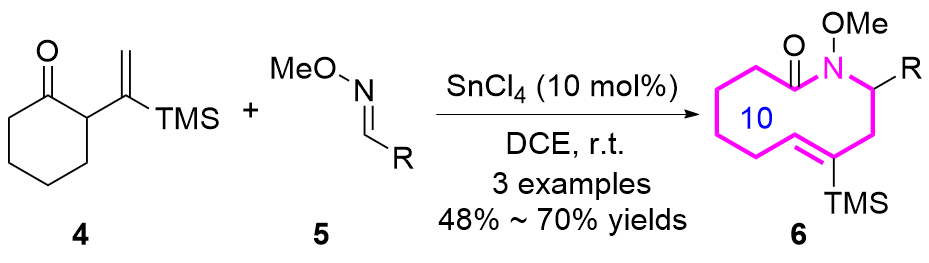
1.2 环加成反应构建十元氮杂环化合物
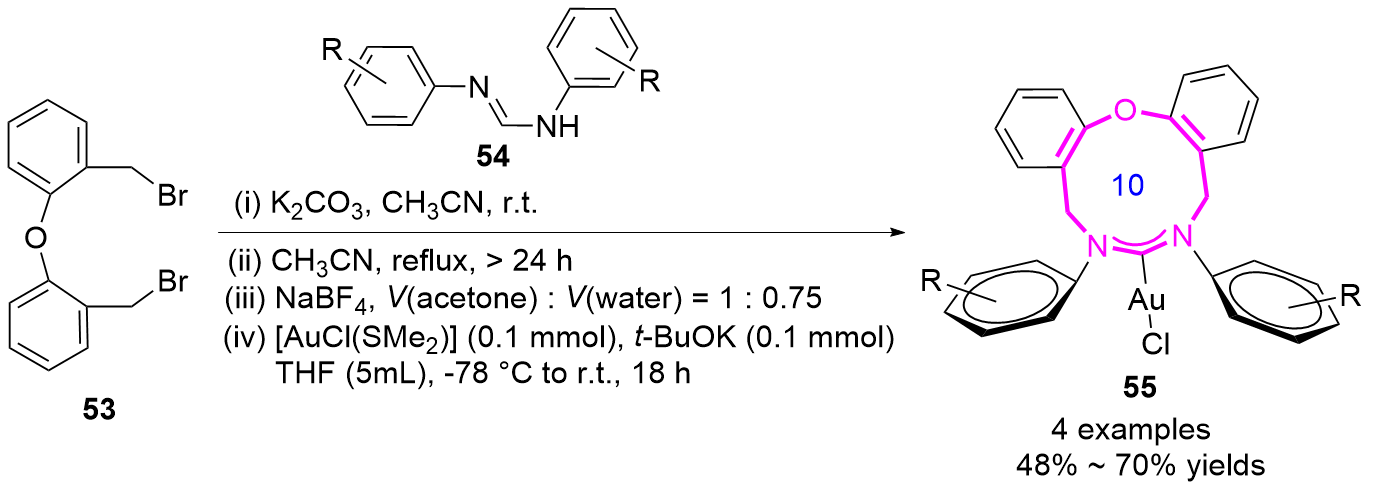

十元氮杂环化合物合成最新研究进展
† 共同第一作者
收稿日期: 2025-09-28
修回日期: 2025-10-08
网络出版日期: 2025-10-29
基金资助
广西自然科学基金(2025GXNSFGA069003)
广西自然科学基金(2023GXNSFDA026025)
国家自然科学基金(22571050)
广西八桂青年学者计划资助项目
Recent Advances on the Synthesis of Ten-Membered Nitrogen Heterocyclic Compounds
† These authors contributed equally to this work.
Received date: 2025-09-28
Revised date: 2025-10-08
Online published: 2025-10-29
Supported by
Natural Science Foundation of Guangxi(2025GXNSFGA069003)
Natural Science Foundation of Guangxi(2023GXNSFDA026025)
National Natural Science Foundation of China(22571050)
Guangxi Bagui Youth Program
罗艳 , 刘章伟 , 胡朝蕾 , 闭红艳 , 莫冬亮 . 十元氮杂环化合物合成最新研究进展[J]. 有机化学, 2026 , 46(2) : 420 -442 . DOI: 10.6023/cjoc202508004
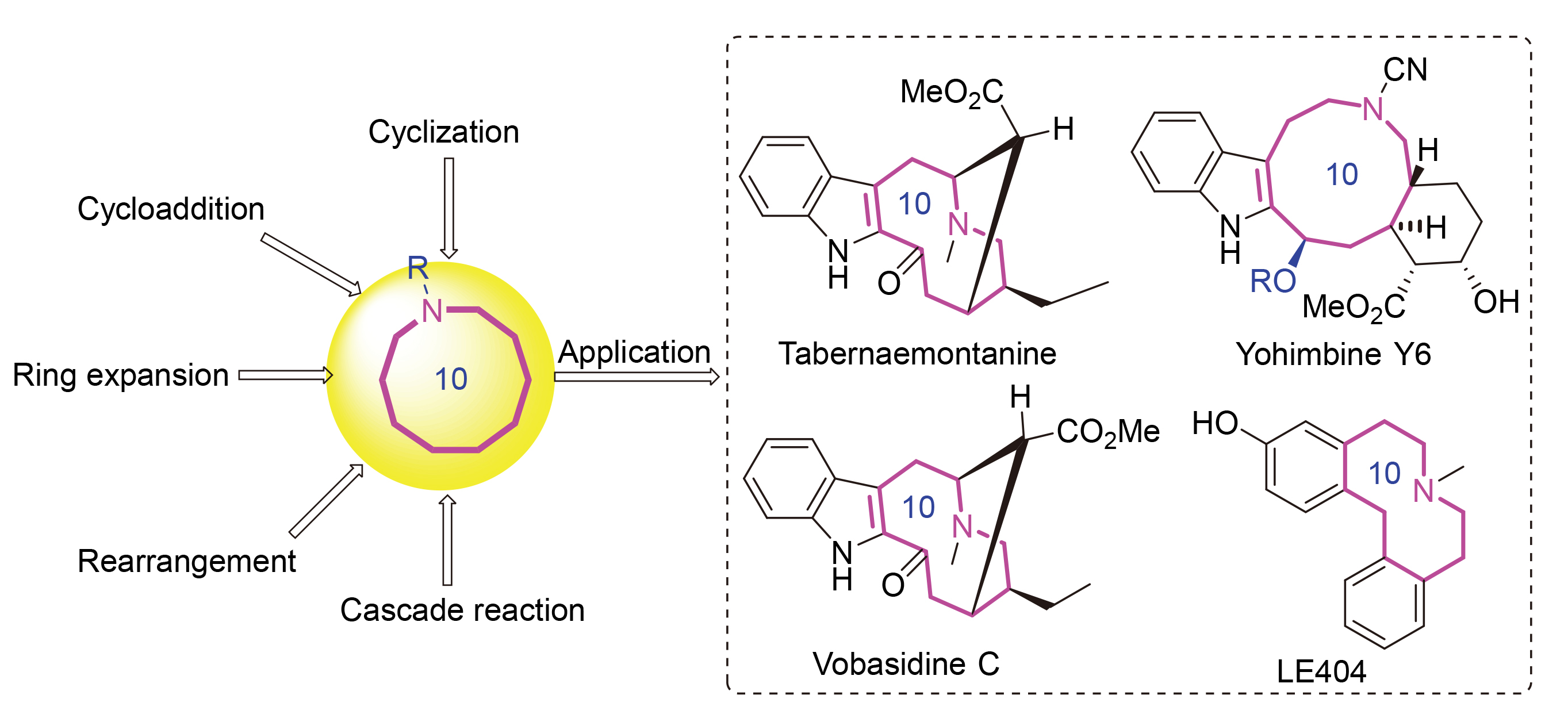
Ten-membered nitrogen heterocyclic compounds are not only the cores of many nitrogen-containing alkaloids, but also the key intermediates for the construction of other natural products. Consequently, their efficient synthesis has garnered significant attention in the field of organic and medicinal chemistry. Over the past decade, since their special medium-sized rings and possessing good biological activities, the synthetic methodologies toward the preparation of ten-membered N-hetero- cyclic compounds have been greatly developed and have also become one of the hottest topics in the medium-sized ring synthetic fields. This review summarizes the recent advances in the synthesis of ten-membered N-heterocyclic compounds in the last decade. It coveres the innovative synthetic methodologies for these ten-membered nitrogen heterocyclic compounds and their applications in the total synthesis of natural products.


| [1] |
(a)
(b)
(c)
(d)
(张馨元, 林礼, 李静, 段世妤, 隆宇航, 李加洪, 有机化学, 2021, 41. 1878.)
(e)
(覃小婷, 邹宁, 农彩梅, 莫冬亮, 有机化学, 2023, 43, 130.)
|
| [2] |
(a)
(b)
(c)
(赵荣宝, 吴毓林, 有机化学, 1988, 8, 97.)
|
| [3] |
(a)
(b)
(c)
(d)
(e)
|
| [4] |
(a)
(b)
(c)
(d)
(e)
(辛翠, 蒋俊, 邓紫微, 欧丽娟, 何卫民, 化学学报, 2024, 82, 1109.)
(f)
|
| [5] |
(a)
(b)
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
(a)
(b)
(c)
(d)
(e)
(f)
(g)
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
(a)
(b)
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
Voskressensky,
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [45] |
|
| [46] |
(a)
(b)
|
| [47] |
|
| [48] |
|
| [49] |
(a)
(b)
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
(a)
(b)
(c)
|
| [54] |
|
| [55] |
(a)
(b)
|
| [56] |
|
| [57] |
|
/
| 〈 |
|
〉 |