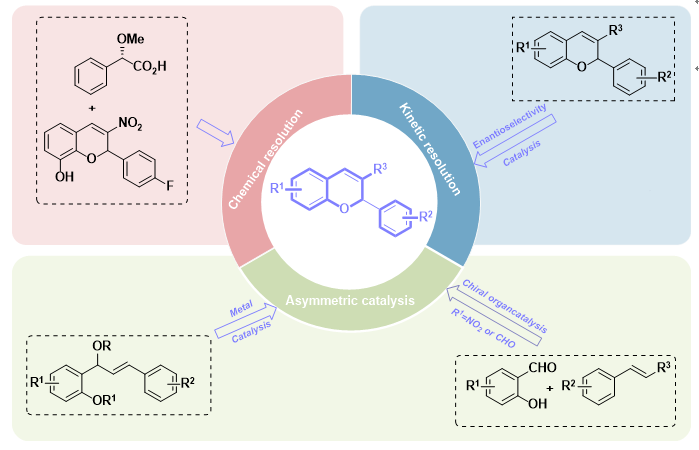
色烯结构是许多天然产物和生物活性分子的重要组成部分,2H-色烯作为色烯的一种,同时也是氧杂环化合物的重要代表,不仅广泛存在于天然产物、药剂和生物相关分子中,而且在材料科学和有机合成中也有广泛的应用。其中手性2-芳基-2H-色烯类化合物具有广泛的药理活性,是合成众多活性天然产物的关键中间体,在药物化学及合成化学等领域具有重要的研究价值。手性化合物或手性药物的对映异构体在生物体内往往会表现出不同的生理活性,自“反应停”事件以来,有机合成工作者在手性合成方面更加重视,使用高光学纯的药物也成为了医药行业的重要指标。因此,手性药物的研究具有重要意义,同时,手性化合物的合成也备受合成工作者的关注。本文归纳总结了近年来2-芳基-2H-色烯类化合物的手性合成研究进展,其中主要介绍了三种该类化合物的手性合成方法,即:化学拆分、动力学拆分、通过手性金属配合物或有机小分子参与的不对称催化,并对该领域未来的发展进行了展望。
Chiral chromene structures are crucial components of numerous natural products and biologically active molecules. As a type of chromene, 2H-chromene is also an important representative of oxygen-containing heterocyclic compounds. It not only widely exists in natural products, pharmaceuticals, and bio-related molecules but also finds extensive applications in materials science and organic synthesis. Among them, chiral 2-aryl-2H-chromene derivatives not only exhibit broad pharmacological activities but also serve as key intermediates for the synthesis of many active natural products, thus holding significant research value in fields such as medicinal chemistry and synthetic chemistry. The enantiomers of chiral compounds or chiral drugs often exhibit distinct physiological activities in living organisms. Since the "The Thalidomide Tragedy", organic synthetic researchers have attached greater importance to chiral synthesis, and the use of pharmaceuticals with high optical purity has become a critical criterion in the pharmaceutical industry. Therefore, the research on chiral drugs is of great significance, and meanwhile, the synthesis of chiral compounds has also attracted considerable attention from synthetic researchers. This paper summarizes the research progress in the chiral synthesis of 2-aryl-2H-chromene compounds in recent years, mainly introducing three chiral synthesis methods for such chiral compounds, namely chemical resolution, kinetic resolution, and asymmetric catalysis involving chiral metal complexes or organosmall molecules. Additionally, the future development of this field is prospected.
[1] Risi C.; Zhao F.; Castagnolo D.ACS. Catal. 2019, 9(8): 7264-7269.
[2] Kaur N.Curr. Org.Synth. 2017, 14(4): 531-556.
[3] Ellis G. P.New York: Wiley-Interscience. 1977, 31.
[4] Ellis G. P.; Lockhart I. M.Chem. Heterocycl. Compd. 1981, 36.
[5] Pratap R.; Ram V. J.Chem. Rev. 2014, 114(20): 10476-10526.
[6] Ruth B.; Jeremy Q.; Peter K.Nat. Rev. Drug. Discov. 2003, 2(3): 177-178.
[7] Thakor V.; Poddar M.; Dey S.; Manjula S. N.; Madhunapantula S. V.; Pawara R.; Patel H. M.; Noolvi M. N.RSC. Adv. 2016, 6, 79166-79179.
[8] Nicolaou K. C.; Pfefferkorn J. A.; Mitchell H. J.; Roecker A. J.; Barluenga S.; Cao G. Q.; Affleck R. L.; Lillig J. E.J. Am. Chem. Soc. 2000, 122(41): 9954-9967.
[9] Aponick A.; Biannic B.; Jong M. R.Chem. Commun.2010, 46(36): 6849-6851.
[10] Zhao Y. J.Master's thesis of Fujian Agriculture and Forestry University. 2017.(in Chinese). (赵旻骏. 福建农林大学硕士学位论文), 2017.
[11] Banerjee D.; Kayal U.Tetrahedron. Letters. 2016, 57(15): 1667-1671.
[12] Ishizuka N.; Matsumura K. I.; Sakai K.J. Med. Chem.2002, 45(10): 2041-2055.
[13] Yu P.; Cen P. L.; Li J. R.China Biotechnology. 2001, (6): 89-94.(in Chinese). 于平, 岑沛霖, 励建荣. 中国生物工程杂志, 2001,(6): 89-94.
[14] Yin S. Q.; Zhang C. F.; Mao X.; Liu Z. P.Tetrahedron: Asymmetry. 2013, 24(5-6): 320-323.
[15] Hardouin C.; Burgaud L.; Valleix A.; Doris E.Tetrahedron Lett. 2003, 44(16): 435-437.
[16] Farina V.; Krishnan B.J. Am.Chem. Soc. 1991, 113: 9585-9595.
[17] Pfenninger A.Synthesis. 1986, 87: 89-116.
[18] Kim D. J.; Lee Y. M.; Choi E. T.; Lee M. H.; Park Y. S.Bull. Korean Chem. Soc. 2009, 30(5): 1211-1214.
[19] Xie J. W.; Fan L. P.; Su H.; Li X. S.; Xu D. C.Org. Biomol. Chem. 2010, 8(9): 2117-2122.
[20] Kang S.; Baek J.; Ko Y.; Im C.; Park K.Synlett. 2013, 24(05): 630-634.
[21] Maji B.; Bhattacharaya A.; Shukla P. M.; Kaushik L. K.Org. Chem.Front. 2019 , 6(20): 3523-3529.
[22] Yang Q. J.; Wang Y. B.; Luo S. H.; W, J. Angew.Chem. Int. Ed. 2019, 58(16): 5343-5347.
[23] a) Bauer, D. J.; Selway, J. W. T.; Batchelor, J. F.; Tisdale, M.; Caldwell, I. C.; Young, D. A. B.Nature, 1981, 292, 369; b) Quaglia, M. G.; Desideri, N.; Bossù, E.; Sestili, I.; Conti, C. Chirality, 1992, 4, 65.
[24] Su Y.; Xie C.; Hu Z.; Zi G.; Li C.; Hou G.Org. Lett. 2025, 27: 3095-3100.
[25] David J.; Maloney J. Z.D.; Shelley, R.; Starck, Z. G.; Sidney, M. H. J. Am. Chem. Soc. 2005, 127: 4140-4141.
[26] Harathi D. S.; Prantik M.; Glenn P. A.Y.; Mary, P.W. J. Org. Chem. 2015, 80(8): 4003-4016.
[27] Majumdar N.; Paul N. D.; Mandal S.; Bruin B.; Wulff W. D.ACS. Catalysis.2015, 5(4): 2329-2366.
[28] Teller H.; Corbet M.; Mantilli L.; Gopakumar G.; Goddard R.; Thiel W.; Furstner A.J. Am. Chem. Soc. 2012, 134(37): 15331-15342.
[29] Reynolds T. E.; Scheidt K. A.Angew. Chem. Int. Ed. 2007, 46(41): 7806-7809.
[30] He H.; Ye K. Y.; Wu Q. F.; Dai L. X.; You S. L.Adv. Synth.Catal. 2012, 354(6): 1084-1094.
[31] Zeng B. S.; Yu X. Y.; Siu P. W.; Scheidt K. A.Chem. Sci.2014, 5(6): 2277-2281.
[32] Jiang Z. Z.; Gao A.; Li H.; Chen D.; Ding C. H.; Xu B.; Hou X. L.Chem-Asian. J.2017, 12(24): 3119-3122.
[33] Wang J. Q.; Crane E. A.; Scheidt K. A.Org. Lett.2011, 13(12): 3086-3089.
[34] Rueping M.; Nachtsheim B. J.; Ieawsuwan W.; Atodiresei I.Angew. Chem.Int. Ed. 2011, 50(30): 6706-6720.
[35] Li H.; Wang J.; E-Nunu, T.; Zu, L.; Jiang, W.; Wei, S. H.; Wang, W. Chem. Commun. 2007, 38(5): 507-509.
[36] Xu,.D.Q.; Wang, Y. F.; Luo, S. P.; Zhang, S.; Zhong, A. G.; Chen, H.; Xu, Z. Y. Adv. Synth. Catal. 2008, 350(16): 2610-2616.
[37] Luo S. P.; Li Z. B.; Wang L. P.; Guo Y. X.; A, B.; Xu, D. Q.Org. Biomol. Chem. 2009; 7(21): 4539.
[38] Moquist P. N.; Kodama T.; Schaus S. E.Angew. Chem.Int. Ed. 2010, 49(39): 7096-7100.
[39] Shen H.; Yang K. F.; Shi Z. H.; Jiang J. X.; Lai J. Q.; Xu L. W.Eur. J. Org. Chem. 2011, 2011(26): 5031-5038.
[40] Zhang Z. G.; Jakab G.; Schreiner P. R.Synlett. 2011(9): 1262-1264.
[41] Rueping M.; Uria U.; Lin M. Y.; Atodiresei I.J. Am.Chem. Soc. 2011, 133(11): 3732-3735.
[42] Yin G. H.; Zhang R. C.; Lei L.; Tan J.; Chen L. G.Eur. J. Org. Chem. 2013, 24: 5431-5438.
[43] Hu Z.; Song L.; Jacobsen E. N.J. Am.Chem. Soc. 2014, 136(47): 16485-16488.
[44] Denmark S. E.; Kornfilt D.J. Org.Chem. 2017, 82(6): 3192-3222.
[45] Mohanta R.; Bez G.J. Org.Chem. 2020, 85(7): 4627-4636.
[46] Rani D.; Gulati V.; Guleria M.; Singh S. P.; Agarwal J.J. Mol. Struct. 2022, 1265: 133341.
[47] Li C. L.; Chen C. X.; Zhan M. R.; Wang Z. X.; Wang S. Y.Russ. J. Org. Chem. 2024,60(S1): S140-S146.



