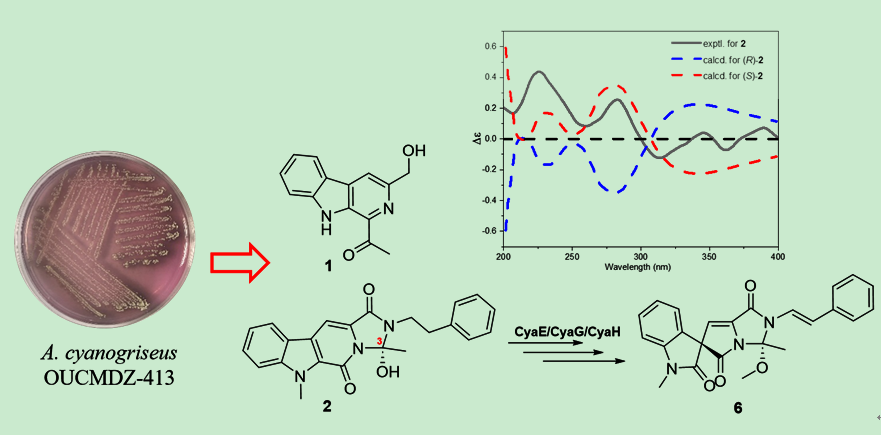
首次从海洋来源的异壁放线菌Actinoalloteichus cyanogriseus OUCMDZ-413的发酵产物中发现了一种新的1-乙酰基-3-羟甲基-β-咔啉 (1) 和一种关键中间体 (S)-cyanogramide B (2),该中间体参与了 cyanogramide (6) 的生物合成;同时还获得了三种已知的类似物 (3-5)。通过核磁共振波谱全解析、比旋光和电子圆二色谱分析,确定了它们的化学结构。(S)-cyanogramide B (2) 的分离以及 cyanogramide (6) 的再次分离强调了发酵优化是挖掘微生物代谢潜力的有效策略,从而为发现菌株A. cyanogriseus OUCMDZ-413的化学多样性提供了参考。
A new 1-acetyl-3-hydroxymethyl-β-carboline (1) and a key intermediate (S)-cyanogramide B (2), involved in the biosynthesis of cyanogramide (6), were isolated for the first time from the marine derived Actinoalloteichus cyanogriseus OUCMDZ-413, along with three know analogues (3-5). Their structures were fully elucidated through comprehensive nuclear magnetic resonance (NMR), specific rotation, and electronic circular dichroism (ECD) techniques. The isolation of (S)-cyanogramide B (2), along with the re-isolation of cyanogramide (6), highlights that fermentation optimization is an effective strategy for exploring the metabolic potential of microorganisms. These findings provide valuable insights into the metabolite diversity of A. cyanogriseus OUCMDZ-413.
[1]Hutchings M. I.; Truman A. W.; Wilkinson, B. Curr. Opin. Microbiol.2019, 51, 72.
[2]Zhao C.; Zhu T.; Zhu, W. Chin. J. Org. Chem.2013, 33, 1195 (in Chinese).
(赵成英, 朱统汉, 朱伟明, 有机化学, 2013, 33, 1195.)
[3]Bu S.; Wang Y.; Yin W.; Wen Q.; Wang P.; Zhu, W. Chin. J. Org. Chem.2025, 45, 2486 (in Chinese).
(卜淑甜, 王永, 殷文剑, 温青云, 王佩, 朱伟明, 有机化学, 2025, 45, 2486.)
[4]Barzkar N.; Jahromi S. T.; Poorsaheli H. B.; Vianello F. Mar. Drugs2019, 17, 464.
[5]Liu H.; Zhu G.; Zhao S.; Fu P.; Zhu, W. Chin. J. Org. Chem.2019, 39, 507 (in Chinese).
(刘海珊, 朱国良, 赵水鸽, 付鹏, 朱伟明, 有机化学, 2019, 39, 507.)
[6]Wang C.; Wang L.; Fan J.; Sun K.; Zhu, W. Chin. J. Org. Chem.2017, 37, 658 (in Chinese).
(王聪, 王立平, 范杰, 孙坤来, 朱伟明, 有机化学, 2017, 37, 658.)
[7]Carroll A. R.; Copp B. R.; Davis R. A.; Keyzers R. A.; Prinsep, M. R. Nat. Prod. Rep.2020, 37, 175.
[8]Du Y.; Chen Z.; Li H.; Wang Y.; Fu P.; Zhu ,W. Chin. Chem. Lett.2019, 30, 981.
[9]Luo Y.; Gao Y.; Yan P.; Zhu, W. Chin. J. Org. Chem.2022, 42, 2840 (in Chinese).
(罗云, 高谕康, 燕鹏程, 朱伟明, 有机化学, 2022, 42, 2840.)
[10]Fu P.; Wang S.; Hong K.; Li X.; Liu P.; Wang Y.; Zhu, W. J. Nat. Prod.2011, 74, 1751.
[11]Fu P.; Liu P.; Li X.; Wang Y.; Wang S.; Hong K.; Zhu W. Org. Lett.2011, 13, 5948.
[12]Fu P.; Zhu Y.; Mei X.; Wang Y.; Jia H.; Zhang C.; Zhu W. Org. Lett.2014, 16, 4264.
[13]Mei X.; Lan M.; Cui G.; Zhang H.; Zhu, W. Org. Chem. Front.2019, 6, 3566.
[14]Gao Y.; Lan M.; Shao J.; Wen Q.; Fu P.; Zhu, W. J. Nat. Prod.2025, 88, 1847.
[15]Mei X.; W L.; Wang D.; Fan J.; Zhu ,W. Chin. J. Org. Chem.2017, 37, 2352 (in Chinese).
(梅显贵, 王立平, 王冬阳, 范杰, 朱伟明, 有机化学, 2017, 37, 2352.)
[16]Fu P.; Kong F.; Li X.; Wang Y.; Zhu W. Org. Lett.2014, 16, 3708.
[17]Zhu Y.; Zhang Q.; Fang C.; Zhang Y.; Ma L.; Liu Z.; Zhai S.; Peng J.; Zhang L.; Zhu W.; Zhang, C. Angew. Chem. Int. Ed.2020, 59, 14065.
[18]Dai J.; Dan W.; Li N.; Wang R.; Zhang Y.; Li N.; Wang R.; Wang J. Food Chem.2018, 253, 211.
[19]Qin L.; Yi W.; Lian X.; Zhang, Z. J. Nat. Prod.2020, 83, 2686.
[20]Wang P.; Kong F.; Wei J.; Wang Y.; Wang W.; Hong K.; Zhu W. Mar. Drugs2014, 12, 477.
[21]Yang S.; Cordell, G. A. J. Nat. Prod.1997, 60, 44.
[22]Yin S.; Liu Z.; Shen J.; Xia Y.; Wang W.; Gui P.; Jia Q.; Kachanuban K.; Zhu W.; Fu P. Nat. Commun.2022, 13, 5169.
[23]Yadav A. N.; Kumar R.; Kumar S.; Kumar V.; Sugitha T.; Singh B.; Chauahan V. S.; Dhaliwal H. S.; Saxena, A. K. J. Appl. Biol. Biotechnol.2017, 5, 45.
[24]Zhao Y.; Xu C.; Wang Q.; Wei Y.; Liu F.; Xu S.; Zhang Z.; Mu, W. Pestic. Biochem. Physiol.2016, 129, 49.
[25]Zheng M.; Shi J.; Shi J.; Wang Q.; Li Y. Biol. Control.2013, 65, 200.
[26]Gao Y.; He L.; Li X.; Lin J.; Mu W.; Liu, F. Pestic. Biochem. Physiol.2018, 147, 51.