Chinese Journal of Organic Chemistry ›› 2026, Vol. 46 ›› Issue (2): 564-569.DOI: 10.6023/cjoc202509007 Previous Articles Next Articles
ARTICLES
刘辉杨a,c, 陈都a, 苏毅进a,*( ), 张鹏a,*(
), 张鹏a,*( ), 刘超b,*(
), 刘超b,*( )
)
收稿日期:2025-09-05
修回日期:2025-10-07
发布日期:2025-10-23
通讯作者:
苏毅进, 张鹏, 刘超
Huiyang Liua,c, Du Chena, Yijin Sua,*( ), Peng Zhanga,*(
), Peng Zhanga,*( ), Chao Liub,*(
), Chao Liub,*( )
)
Received:2025-09-05
Revised:2025-10-07
Published:2025-10-23
Contact:
Yijin Su, Peng Zhang, Chao Liu
Share
Huiyang Liu, Du Chen, Yijin Su, Peng Zhang, Chao Liu. Rapid and Controlled Reduction of Acyl Chlorides to Aldehydes Using Pinacolborane[J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 564-569.
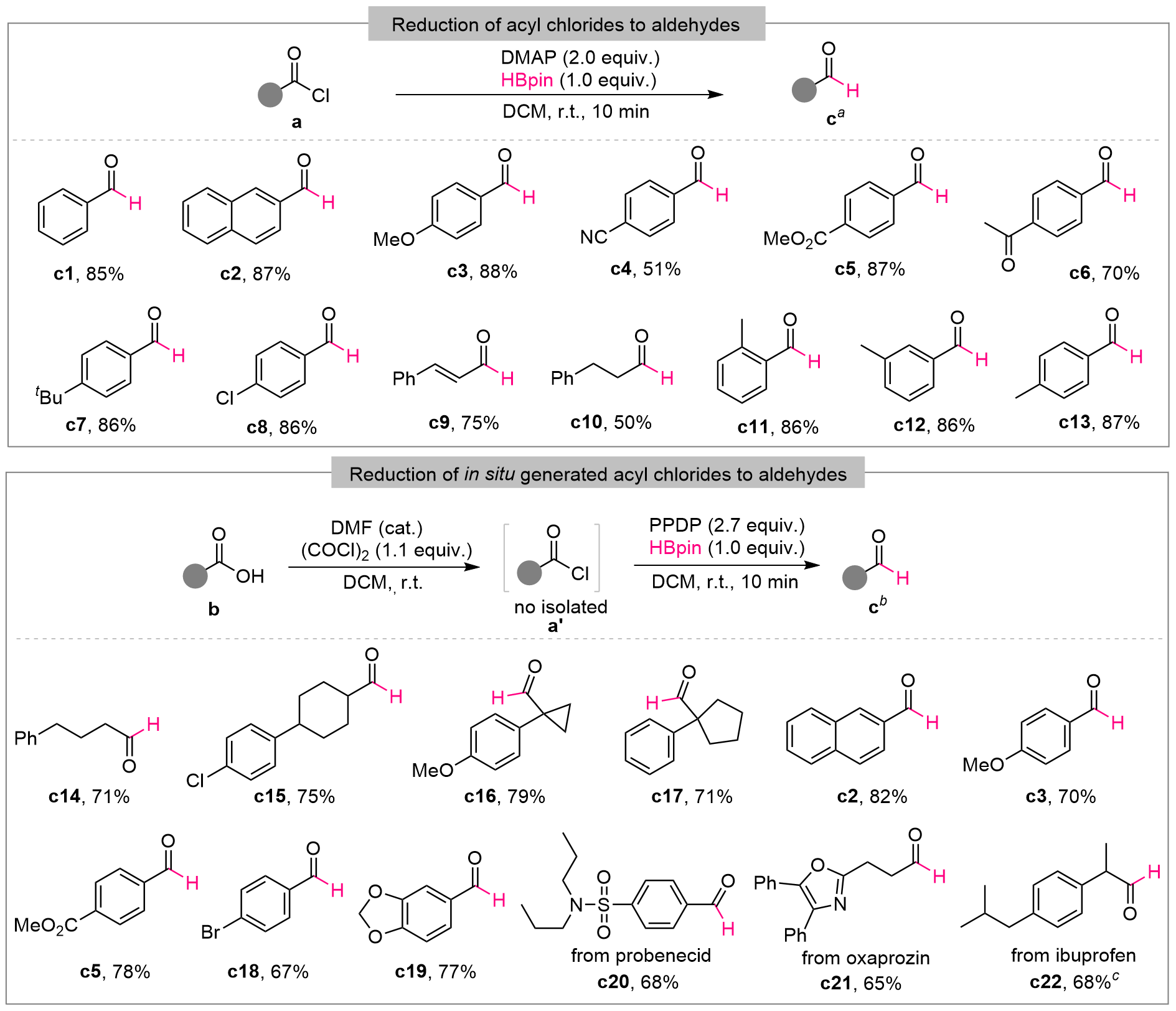


| [1] |
(a)
doi: 10.3390/polym15061517 |
|
(b)
doi: 10.1016/j.indcrop.2023.117372 |
|
|
(c)
|
|
|
(d)
|
|
|
(e)
doi: 10.1021/op2003826 |
|
| [2] |
(a)
doi: 10.1021/jacs.4c03164 |
|
(b)
doi: 10.6023/cjoc202401009 |
|
|
(赵明, 颜瑞, 陈虎, 有机化学, 2024, 44, 2204.)
doi: 10.6023/cjoc202401009 |
|
|
(c)
doi: 10.6023/cjoc202406008 |
|
|
(杨甜甜, 陈都, 张鹏, 刘超, 有机化学, 2025, 45, 212.)
doi: 10.6023/cjoc202406008 |
|
|
(d)
doi: 10.6023/cjoc202504018 |
|
|
(任博文, 方通昌, 刘超, 有机化学, 2025, 45, 3343.)
doi: 10.6023/cjoc202504018 |
|
| [3] |
(a)
doi: 10.1038/s41929-018-0143-0 |
|
(b)
doi: 10.1002/recl.v100:1 |
|
|
(c)
doi: 10.1002/cber.v54:3 |
|
|
(d)
doi: 10.1002/cber.v51:1 |
|
| [4] |
(a)
doi: 10.1021/jo00069a043 |
|
(b)
doi: 10.1016/0040-4020(79)87003-9 |
|
|
(c)
doi: 10.1021/ja01553a014 |
|
|
(d)
doi: 10.1021/ja01582a072 |
|
| [5] |
(a)
doi: 10.1055/s-0029-1219796 |
|
(b)
doi: 10.1002/tcr.v5:6 |
|
|
(c)
doi: 10.1016/S0040-4039(99)02016-X |
|
|
(d)
doi: 10.1016/S0040-4039(97)10117-4 |
|
|
(e)
doi: 10.1021/jo00335a023 |
|
| [6] |
(a)
doi: 10.1080/00397918208065961 |
|
(b)
doi: 10.1016/0040-4039(81)80027-5 |
|
| [7] |
(a)
doi: 10.1016/j.catcom.2014.02.018 |
|
(b)
doi: 10.1055/s-00000083 |
|
|
(c)
doi: 10.1002/adsc.v354.4 |
|
|
(d)
doi: 10.1021/ol060463v |
|
|
(e)
doi: 10.1016/S0040-4039(00)80274-9 |
|
|
(f)
doi: 10.1021/jo01258a105 |
|
| [8] |
doi: 10.1021/acscatal.0c04051 |
| [9] |
|
| [10] |
doi: 10.1039/C7GC01632H |
| [11] |
(a)
doi: 10.1002/anie.v43:41 |
|
(b)
|
|
|
(c)
doi: 10.1021/acs.orglett.7b01213 |
|
| [12] |
doi: 10.1002/adsc.v362.24 |
| [13] |
doi: 10.1039/D1CC00965F |
| [14] |
doi: 10.1002/anie.v55.14 |
| [15] |
doi: 10.1021/ol403183a |
| [16] |
doi: 10.1021/acscatal.8b03592 pmid: 31367474 |
| [17] |
doi: 10.1021/ol060463v |
| [18] |
doi: 10.1039/D5OB00704F |
| [19] |
doi: 10.1021/acscatal.0c02755 |
| [20] |
|
| [21] |
doi: 10.1021/acs.orglett.7b00958 |
| [22] |
doi: 10.1021/ja910778p pmid: 20377222 |
| [1] | Nana Wei, Wanzhen Guo, Xing Lu, Zhiqiang Ren, Haojie Ma, Yuqi Zhang, Jijiang Wang, Bo Han. Magnesium-Promoted Selectively Reduction of Unprotected Indoles and Quinoxalines [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 1027-1038. |
| [2] | Jialong Xu, Quan Kong, Jianhui Chen, Mingyue Zhang, Xiaoming Ji, Biao Cheng. Recent Advances in Low-Valent Tungsten-Catalyzed Organic Reactions [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 759-772. |
| [3] | Xiaoxiang Xi, Ming Gao, Libiao Han. Rhodium-Catalyzed Reduction of RS—SR to Thiophenols/Thiols with H2 [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 859-865. |
| [4] | Ke Ren, Guanglu Zhang, Yifan Niu, Xiaomeng Wang, Canyu Chen, Min Jiang. Photocatalytic Radical-Mediated Synthesis of Unnatural α-Amino Acids from Heteroaryl Aldehydes [J]. Chinese Journal of Organic Chemistry, 2026, 46(1): 215-224. |
| [5] | Shunli Du, Yaya Wang, Jiaming Guo, Xuewei Xu, Xinhua Peng. Study on the Borylation of β-C Positions of Aryl Olefins Catalyzed by Copper Organophosphorus Complexes [J]. Chinese Journal of Organic Chemistry, 2025, 45(7): 2435-2443. |
| [6] | Jiangyan Tian, Chi Li, Jianfei Chen, Hui Lv. Nickel and Cobalt-Catalyzed Asymmetric Reductive Coupling of Halides with Aldehydes, Ketones and Imines [J]. Chinese Journal of Organic Chemistry, 2025, 45(6): 1946-1960. |
| [7] | Yepeng Xu, Yi Ruan, Jianfeng Zheng, Peiqiang Huang. Synthesis of (–)-Sedacryptine and (–)-Geissman-Waiss Lactone by Applying Methods for the Direct Transformation of Amides [J]. Chinese Journal of Organic Chemistry, 2025, 45(3): 988-995. |
| [8] | Tiantian Yang, Du Chen, Peng Zhang, Chao Liu. Application of Triborylmethane Reagent in the Stereoselective Synthesis of Dissymmetric gem-Diborylalkenes [J]. Chinese Journal of Organic Chemistry, 2025, 45(1): 212-219. |
| [9] | Mengjin Liu, Yan Xiao, Kai Zhou, Zicheng Li, Wencai Huang. Synthesis of 3,5-Diaryl-1,2,4-oxadiazole by One-Pot Homocoupling Reaction [J]. Chinese Journal of Organic Chemistry, 2024, 44(7): 2251-2256. |
| [10] | Junyong Wang, Na Li, Jie Ke, Chuan He. Recent Advances in Electrochemical Silylation [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 927-939. |
| [11] | Hongbing Chen, Sijia Yang, Zhipeng Ye, Kai Chen, Haoyue Xiang, Hua Yang. Electrocatalytic Reduction of Quinolines and Ketones by Using Lewis Base-Ligated Borane as a Hydrogen Donor [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 966-971. |
| [12] | Jian Huang, Wenzhen Zhang. Advances in Electrochemical Cathodic Reductive Reactions Involving Carbon-Nitrogen Bonds [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 825-839. |
| [13] | Fangying Duan, Menglei Yuan, Jian Zhang. Paired Electrolysis for Inorganic Small Molecules Reduction Coupled with Alternative Oxidation Reactions [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 809-824. |
| [14] | Jing Xiao, Yuxiang Xie, Libiao Han. Advances in Organic Reactions Using Phosphorus Acid as Reducing Agent [J]. Chinese Journal of Organic Chemistry, 2024, 44(12): 3702-3712. |
| [15] | Qiuting Zhao, Wenguang Wang. Iron-Catalyzed Selective Hydrogenation and Hydroboration/Hydrosilylation of CO2 [J]. Chinese Journal of Organic Chemistry, 2024, 44(10): 3106-3116. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||