Chinese Journal of Organic Chemistry ›› 2026, Vol. 46 ›› Issue (3): 725-742.DOI: 10.6023/cjoc202509026 Previous Articles Next Articles
REVIEWS
王雪倩a,b, 段宝忠b,*( ), 田红畅a,*(
), 田红畅a,*( ), 张磊a,*(
), 张磊a,*( )
)
收稿日期:2025-09-22
修回日期:2025-10-24
发布日期:2025-12-09
基金资助:
Xueqian Wanga,b, Baozhong Duanb,*( ), Hongchang Tiana,*(
), Hongchang Tiana,*( ), Lei Zhanga,*(
), Lei Zhanga,*( )
)
Received:2025-09-22
Revised:2025-10-24
Published:2025-12-09
Contact:
*Supported by:Share
Xueqian Wang, Baozhong Duan, Hongchang Tian, Lei Zhang. Research Progress in Synthesis of Indole Alkaloid Catharanthine[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 725-742.
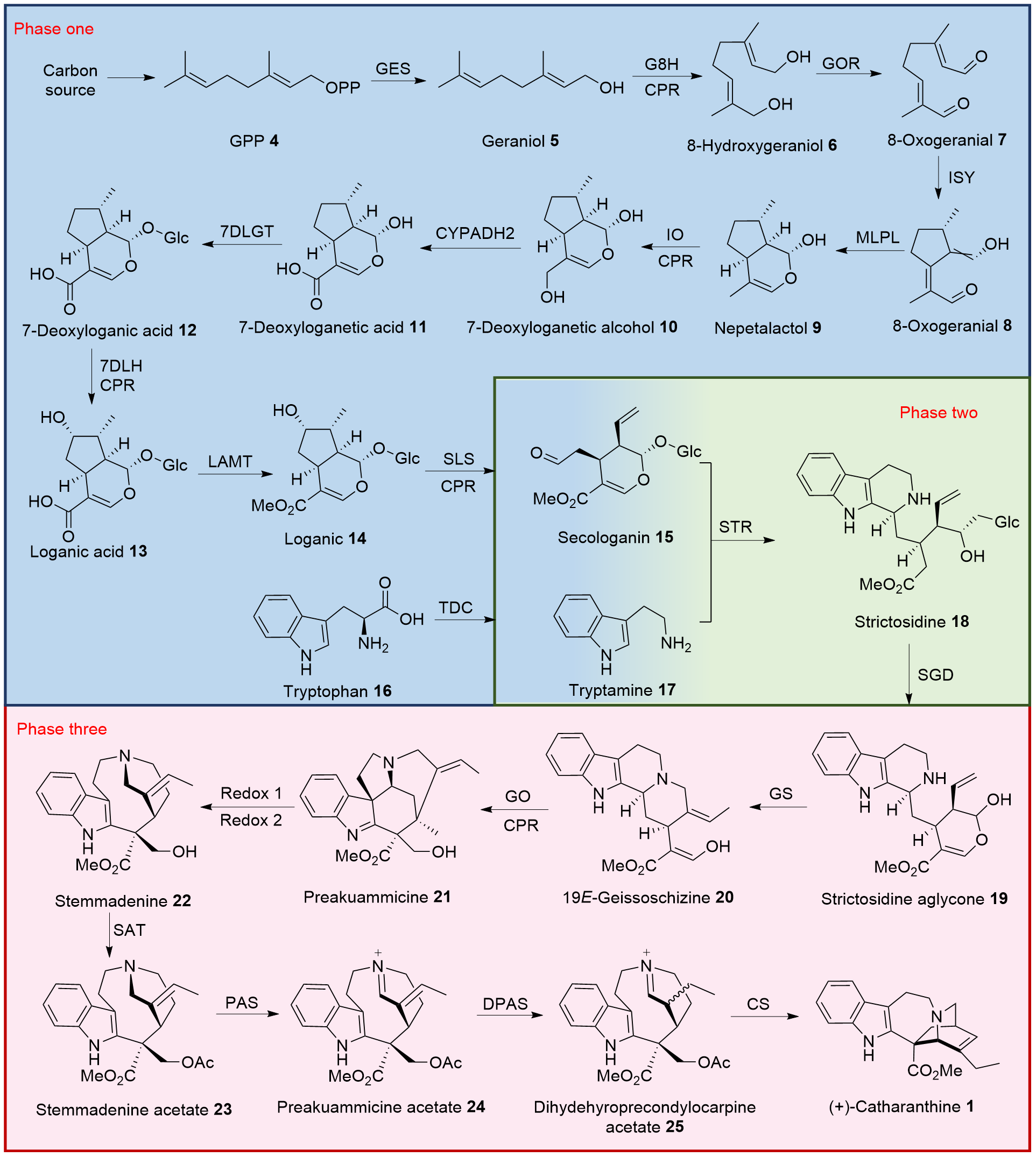
| Entry | Research group | Year | (±/+/-)-Catharanthine | Steps | Yield/% | Reference |
|---|---|---|---|---|---|---|
| 1 | Büchiʼ group | 1969 | (±)-Catharanthine | 17 | 1.4 | [ |
| 2 | Kutneyʼ group | 1975 | (±)-Catharanthine | 12 | <1 | [ |
| 3 | Trostʼ group | 1979 | (±)-Catharanthine | 12 | 13 | [ |
| 4 | Beislerʼ group | 1980 | (±)-Catharanthine | 14 | <1 | [ |
| 5 | Tmanishiʼ group | 1980 | (±)-Catharanthine | 19 | <1 | [ |
| 6 | Dasʼ group | 1981 | (±)-Catharanthine | 10 | 1.4 | [ |
| 7 | Raucherʼ group | 1985 | (±)-Catharanthine | 10 | 13 | [ |
| 8 | Kuehneʼ group | 1986 | (±)-Catharanthine | 9 | 11 | [ |
| 9 | Kuehneʼ group | 2008 | (±)-Catharanthine | 12 | 2.6 | [ |
| 10 | Szántayʼ group | 1990 | (+)-Catharanthine | 9 | 6.3 | [ |
| 11 | Fukuyamaʼ group | 1999 | (±)-Catharanthine | 13 | 12.4 | [ |
| 12 | Dorisʼ group | 2006 | (+)-Catharanthine | 24 | <1 | [ |
| 13 | Oguriʼ group | 2013 | (-)-Catharanthine | 10 | 3.2 | [ |
| 14 | Ishiharaʼ group | 2014 | (-)-Catharanthine | 17 | <1 | [ |
| 15 | Ishiharaʼ group | 2015 | (+)-Catharanthine | 19 | <1 | [ |
| 16 | Tuoping Luoʼ group | 2016 | (+)-Catharanthine | 13 | <1 | [ |
| 17 | Bateyʼ group | 2018 | (+)-Catharanthine | 15 | <1 | [ |
| 18 | Nemotoʼ group | 2019 | (+)-Catharanthine | 27 | <1 | [ |
| 19 | Dixonʼ group | 2021 | (±)-Catharanthine | 5 | 2.1 | [ |
| 20 | Grögerʼ group | 2023 | (-)-Catharanthine | 22 | <1 | [ |
| Entry | Research group | Year | (±/+/-)-Catharanthine | Steps | Yield/% | Reference |
|---|---|---|---|---|---|---|
| 1 | Büchiʼ group | 1969 | (±)-Catharanthine | 17 | 1.4 | [ |
| 2 | Kutneyʼ group | 1975 | (±)-Catharanthine | 12 | <1 | [ |
| 3 | Trostʼ group | 1979 | (±)-Catharanthine | 12 | 13 | [ |
| 4 | Beislerʼ group | 1980 | (±)-Catharanthine | 14 | <1 | [ |
| 5 | Tmanishiʼ group | 1980 | (±)-Catharanthine | 19 | <1 | [ |
| 6 | Dasʼ group | 1981 | (±)-Catharanthine | 10 | 1.4 | [ |
| 7 | Raucherʼ group | 1985 | (±)-Catharanthine | 10 | 13 | [ |
| 8 | Kuehneʼ group | 1986 | (±)-Catharanthine | 9 | 11 | [ |
| 9 | Kuehneʼ group | 2008 | (±)-Catharanthine | 12 | 2.6 | [ |
| 10 | Szántayʼ group | 1990 | (+)-Catharanthine | 9 | 6.3 | [ |
| 11 | Fukuyamaʼ group | 1999 | (±)-Catharanthine | 13 | 12.4 | [ |
| 12 | Dorisʼ group | 2006 | (+)-Catharanthine | 24 | <1 | [ |
| 13 | Oguriʼ group | 2013 | (-)-Catharanthine | 10 | 3.2 | [ |
| 14 | Ishiharaʼ group | 2014 | (-)-Catharanthine | 17 | <1 | [ |
| 15 | Ishiharaʼ group | 2015 | (+)-Catharanthine | 19 | <1 | [ |
| 16 | Tuoping Luoʼ group | 2016 | (+)-Catharanthine | 13 | <1 | [ |
| 17 | Bateyʼ group | 2018 | (+)-Catharanthine | 15 | <1 | [ |
| 18 | Nemotoʼ group | 2019 | (+)-Catharanthine | 27 | <1 | [ |
| 19 | Dixonʼ group | 2021 | (±)-Catharanthine | 5 | 2.1 | [ |
| 20 | Grögerʼ group | 2023 | (-)-Catharanthine | 22 | <1 | [ |
| [1] |
doi: 10.1073/pnas.2318586121 |
| [2] |
doi: 10.1124/jpet.112.199661 pmid: 23532933 |
| [3] |
doi: 10.1021/acschemneuro.3c00478 |
| [4] |
doi: 10.1007/s00210-024-03191-8 |
| [5] |
doi: 10.1002/jps.3030480419 |
| [6] |
(a)
doi: 10.1021/acs.orglett.7b03694 |
|
(b)
doi: 10.1038/s41586-022-05157-3 |
|
| [7] |
(a)
doi: 10.1007/s00253-020-10592-1 pmid: 15032608 |
|
(b)
pmid: 15032608 |
|
|
(c)
doi: 10.2174/0929867043455846 pmid: 15032608 |
|
| [8] |
(a)
pmid: 18609590 |
|
(b)
doi: 10.1021/ar50029a002 pmid: 18609590 |
|
|
(c)
pmid: 18609590 |
|
|
(d)
doi: 10.1007/BF00233027 pmid: 18609590 |
|
|
(e)
pmid: 18609590 |
|
|
(f)
doi: 10.1016/S0922-338X(97)80984-1 pmid: 18609590 |
|
|
(g)
doi: 10.1016/S0141-0229(01)00306-4 pmid: 18609590 |
|
|
(h)
doi: 10.1007/s00299-010-0874-0 pmid: 18609590 |
|
| [9] |
doi: 10.1126/science.aat4100 |
| [10] |
doi: 10.1021/ol200996a |
| [11] |
doi: 10.4014/jmb.2207.07057 |
| [12] |
(a)
doi: 10.1073/pnas.1501821112 pmid: 34302444 |
|
(b)
doi: 10.1002/bit.v119.5 pmid: 34302444 |
|
|
(c)
doi: 10.1111/1751-7915.13898 pmid: 34302444 |
|
| [13] |
doi: 10.1038/s44160-022-00205-2 |
| [14] |
(a)
doi: 10.1146/phyto.2018.56.issue-1 |
|
(b)
doi: 10.1007/s00299-018-2296-3 |
|
| [15] |
doi: 10.1016/j.molp.2023.10.016 |
| [16] |
doi: 10.1093/mp/ssq020 pmid: 20457641 |
| [17] |
doi: 10.1038/s41586-020-2546-8 |
| [18] |
doi: 10.1021/acssynbio.2c00434 |
| [19] |
doi: 10.1021/ja00707a043 |
| [20] |
pmid: 5459202 |
| [21] |
(a)
doi: 10.1021/ja01065a038 pmid: 5418451 |
|
(b)
doi: 10.1021/ja00972a061 pmid: 5418451 |
|
|
(c)
pmid: 5418451 |
|
|
(d)
pmid: 5418451 |
|
| [22] |
doi: 10.1021/jo01326a044 |
| [23] |
doi: 10.1016/0040-4020(80)80061-5 |
| [24] |
pmid: 6037875 |
| [25] |
doi: 10.1021/ja01059a063 |
| [26] |
|
| [27] |
|
| [28] |
pmid: 1176299 |
| [29] |
doi: 10.1021/jo00217a052 |
| [30] |
(a)
doi: 10.1021/jo00365a012 |
|
(b)
doi: 10.1016/j.tet.2008.07.044 |
|
| [31] |
doi: 10.1016/S0040-4020(01)81977-3 |
| [32] |
(a)
doi: 10.1021/ol990749i |
|
(b)
doi: 10.3987/COM-01-S(K)42 |
|
| [33] |
doi: 10.1002/anie.v45:32 |
| [34] |
doi: 10.1038/nchem.1798 pmid: 24345948 |
| [35] |
doi: 10.1039/C4CC01445F |
| [36] |
doi: 10.1021/jacs.5b08693 |
| [37] |
doi: 10.1039/c6sc00932h pmid: 30034694 |
| [38] |
pmid: 17941641 |
| [39] |
doi: 10.1039/C8QO00849C |
| [40] |
doi: 10.1021/acs.orglett.9b01198 |
| [41] |
doi: 10.1021/ja00236a023 |
| [42] |
doi: 10.1021/jacs.1c04980 pmid: 34254792 |
| [43] |
doi: 10.1055/a-2086-0690 |
| [1] | Wenfu Cheng, Minghao Zhao, Yuchen Yang, Lijia Wang. The First Total Synthesis of N-Methyl Pratensilin B [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 670-677. |
| [2] | Weigang He, Mingyue Duan, Shuai Yan, Weijie Ye, Xueyuan Xu, Xianyu Sun. Progress in the Total Synthesis of the Kopsia Alkaloids Arboridinine and Arborisidine [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 443-454. |
| [3] | Lilin Zhao, Zhaoqi Li, Liping Zhong, Long Min. Recent Progress in the Total Synthesis of abeo-Steroids [J]. Chinese Journal of Organic Chemistry, 2026, 46(1): 21-38. |
| [4] | Xinyi Cui, Lifan Guo, Congxuan Ma, Yun Li, Jianhua Liang. Structural Modifications, Structure-Activity Relationships, and Total Synthesis Advances in Erythromycin Analogs against Resistant Pathogens [J]. Chinese Journal of Organic Chemistry, 2026, 46(1): 39-73. |
| [5] | Qianqian Zhao, Peiyao Wei, Sundian Liu, Boxin Zhang, Chengyuan Liang. Applications of Palladium-Catalyzed Cascade Reactions in Total Synthesis of Complex Natural Products with Quaternary Carbon Centers [J]. Chinese Journal of Organic Chemistry, 2025, 45(9): 3289-3300. |
| [6] | Ying Xia, Chenlong Zhu, Bingfeng Sun. Progress in Total Synthesis of Entecavir, an Antiviral Drug for Hepatitis B★ [J]. Chinese Journal of Organic Chemistry, 2025, 45(9): 3186-3202. |
| [7] | Xiaoyu Liu, Tingrui Xu, Yong Qin. Recent Progress in the Total Synthesis of Morphine Alkaloids★ [J]. Chinese Journal of Organic Chemistry, 2025, 45(9): 3098-3112. |
| [8] | Baichuan Mo, Tingting Li, Fang Wang, Xiaojie Li, Darifu Ba. Recent Progress in the Synthesis of Carbazolequinone Natural Products [J]. Chinese Journal of Organic Chemistry, 2025, 45(8): 2746-2766. |
| [9] | Yiming Ding, Tingrong Zhang, Jingwei Zhang, Jun Deng. Progress in the Total Synthesis of Cephalotane Diterpenoid Natural Products [J]. Chinese Journal of Organic Chemistry, 2025, 45(6): 2048-2073. |
| [10] | Weigang He, Yadong Liu, Yingkai Deng, Xianyu Sun. Recent Progress in the Synthesis of the Malagasy Alkaloids [J]. Chinese Journal of Organic Chemistry, 2025, 45(4): 1153-1165. |
| [11] | Jiuzhou Yi, Liang Huo, Jinyan Chen, Meng Liu, Huilin Li, Xuegong She. Protecting Group Effects on the Total Syntheses of Several Classes of Natural Products [J]. Chinese Journal of Organic Chemistry, 2025, 45(3): 1030-1039. |
| [12] | Jie Chen, Jun Li, Xianwen Long, Haixiang Shen, Jun Deng. Recent Advances of Wagner-Meerwein Rearrangement in Natural Product Synthesis [J]. Chinese Journal of Organic Chemistry, 2025, 45(3): 896-912. |
| [13] | Chuang Li, Cheng Zhang, Xiaoyu Liu, Yong Qin. Recent Progress in the Total Synthesis of Diterpenoid Alkaloids [J]. Chinese Journal of Organic Chemistry, 2025, 45(3): 881-895. |
| [14] | Qingxing Yang, Xuan Liu, Shuo Ma, Xinxin Li, Dongxu Ma, Tao Xu. Total Syntheses of Marine-Derived Polyhalogenated Natural Products [J]. Chinese Journal of Organic Chemistry, 2025, 45(3): 764-803. |
| [15] | Yunbo Yang, Hanfeng Ding. Advances in the Total Syntheses of Tetraquinane and Tetraquinane-Type Terpenes [J]. Chinese Journal of Organic Chemistry, 2025, 45(3): 725-747. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||