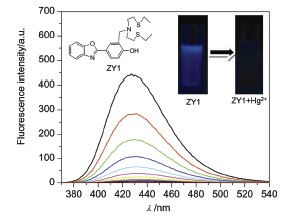
| [1] Clarkson, T. W.; Magos, L.; Myers, G. J. N. Engl. J. Med. 2003, 349, 1731. [2] Nolan, E. M.; Lippard, S. J. Chem. Rev. 2008, 108, 3443. [3] Du, M.-H.; Han, Z.-X.; Wu, X.-Y.; Liang, G.-X.; Zhao, T.; Zhang, H.-Q. Chin. J. Spectrosc. Lab. 2012, 29, 3170. (杜明辉, 韩志湘, 吴向阳, 梁国熙, 赵婷, 张浩琪, 光谱实验室, 2012, 29, 3170.)[4] Wu, X.-L.; Lai, J.-P.; Zhao, Y.-B. Chem. J. Chin. Univ. 2012, 33, 695. (吴小丽, 赖金平, 赵一兵, 高等学校化学学报, 2012, 33, 695.)[5] Huang, W.-J.; Wu, W.-H.; Liang, J.-X. Acta Chim. Sinica 2012, 70, 873. (黄文君, 吴文辉, 梁嘉香, 化学学报, 2012, 70, 873.)[6] Chi, X.-B.; Liu, Y. Chin. J. Org. Chem. 2013, 33, 1545. (迟兴宝, 刘洋, 有机化学, 2013, 33, 1545.)[7] Lü, S.-Y.; Ye, T.; Jiang, X.; Wang, J.-J.; Wei, G.-F.; Lü, J.-Q. Acta Chim. Sinica 2011, 69, 831. (吕诗言, 叶泰, 姜欣, 王晶晶, 魏国芬, 吕鉴泉, 化学学报, 2011, 69, 831.) [8] Nolan, E. M.; Lippard, S. J. J. Am. Chem. Soc. 2007, 129, 5910. [9] Yang, Y.-M.; Zhao, Q.; Feng, W.; Li, F.-Y. Chem. Rev. 2013, 113, 192. [10] Meng, Q.-H.; Cheng, S.; Lan, M.-B.; Wei, G. Chin. J. Chem. 2012, 30, 249 [11] Liu, J.; Sun, Y.-Q.; Wang, P.; Zhang, J.-Y.; Guo, W. Analyst 2013, 138, 2654. [12] Priyanka, S.; Rashid, A.; Syed, S. R.; Mohammad, S.; Satyakam, P.; Arvind, M. Tetrahedron Lett. 2013, 54, 3688. [13] Lin, Q.; Chen, P.; Liu, J.; Fu, Y.-P.; Zhang, Y.-M.; Wei, T.-B. Prog. Chem. 2013, 25, 1177. (林奇, 陈佩, 刘娟, 符永鹏, 张有明, 魏太保, 化学进展, 2013, 25, 1177.)[14] Wang, X.-M.; Wang, J.-G.; Xu, W.-C. Dyestuffs Coloration, 2005, 42, 8. (王雪梅, 王景国, 胥维昌, 染料与染色, 2005, 42, 8.)[15] Pan, J.-X.; Wang, C.-J.; Gao, Z.-H.; Yao, X.-K.; Wang, R.-J. Chem. J. Chin. Univ. 1990, 11, 31. (潘家杏, 王超杰, 高振衡, 姚心侃, 王如骥, 高等学校化学学报, 1990, 11, 31.)[16] Fan, J.-L.; Guo, K.-X.; Peng, X.-J.; Du, J.-J.; Wang, J.-Y.; Sun, S.-G.; Li, H.-L. Sens. Actuators, B: Chem. 2009, 142, 191. [17] Taki, M.; Akaoka, K.; Iyoshi, S.; Yamamoto, Y. Inorg. Chem. 2012, 51, 13075. [18] Du, J.-J.; Fan, J.-L.; Peng, X.-J.; Sun, P.-P.; Wang, J.-Y.; Li, H.-L.; Sun, S.-G. Org. Lett. 2010, 12, 476. [19] Chen, X.-Q.; Nam, S.-W.; Jou, M. J.; Kim, Y.; Kim, S.-J.; Park, S.; Yoon, J. Org. Lett. 2008, 10, 5235. [20] Chen, M.-L.; Lv, X.; Liu, Y.-L.; Zhao, Y.; Liu, J.; Wang, P.; Guo, W. Org. Biomol. Chem. 2011, 9, 2345. [21] Chen, W.-H.; Xing, Y.; Pang, Y. Org. Lett. 2011, 13, 1362. [22] Kwon, J. E.; Lee, S.; You, Y.; Baek, K. H.; Ohkubo, K.; Cho, J.; Fukuzumi, S.; Shin, I.; Park, S. Y.; Nam, W. Inorg. Chem. 2012, 51, 8760.[23] Shortreed, M.; Kopelman, R.; Kuhn, M.; Hoyland, B. Anal. Chem. 1996, 68, 1414. |