Acta Chimica Sinica ›› 2022, Vol. 80 ›› Issue (12): 1618-1628.DOI: 10.6023/A22090385 Previous Articles Next Articles
Review
薛晓兰a, 张洋a, 石美瑜a, 李天琳a, 黄天龙a, 戚继球a, 委福祥a, 隋艳伟a,*( ), 金钟b,*(
), 金钟b,*( )
)
投稿日期:2022-09-05
发布日期:2022-10-27
通讯作者:
隋艳伟, 金钟
作者简介: |
薛晓兰, 2015年于陕西师范大学化学化工学院获学士学位, 2020年于南京大学化学化工学院获博士学位. 2020年被聘为中国矿业大学材料与物理学院讲师. 主要研究方向为新型多价态金属离子电池电极材料的设计合成及储能机制研究. |
 |
隋艳伟, 2003年于哈尔滨理工大学获学士学位, 2009年于哈尔滨工业大学获博士学位. 2009年被聘为中国矿业大学材料与物理学院讲师, 2016年于新西兰奥克兰大学进行访学, 2020年被聘为中国矿业大学材料与物理学院教授. 主要从事新能源材料与器件和合金材料及功能化方面的研究. |
 |
金钟, 2003年于北京大学化学与分子工程学院获学士学位, 2008年于北京大学化学与分子工程学院获博士学位. 2008年和2010年分别于美国莱斯大学和麻省理工学院进行博士后研究. 2014年被聘为南京大学化学化工学院教授. 主要从事清洁能源转换/存储材料及器件和新型纳米材料性质调控与光电功能器件方面的研究. |
基金资助:
Xiaolan Xuea, Yang Zhanga, Meiyu Shia, Tianlin Lia, Tianlong Huanga, Jiqiu Qia, Fuxiang Weia, Yanwei Suia( ), Zhong Jinb(
), Zhong Jinb( )
)
Received:2022-09-05
Published:2022-10-27
Contact:
Yanwei Sui, Zhong Jin
Supported by:Share
Xiaolan Xue, Yang Zhang, Meiyu Shi, Tianlin Li, Tianlong Huang, Jiqiu Qi, Fuxiang Wei, Yanwei Sui, Zhong Jin. Recent Progress on Organic Electrode Materials for Nonaqueous Magnesium Secondary Batteries[J]. Acta Chimica Sinica, 2022, 80(12): 1618-1628.
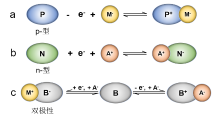

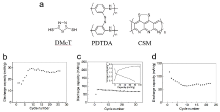

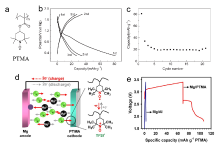

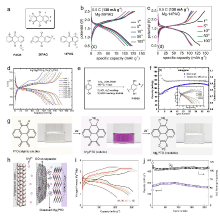





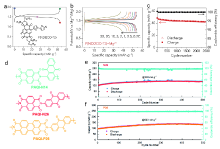
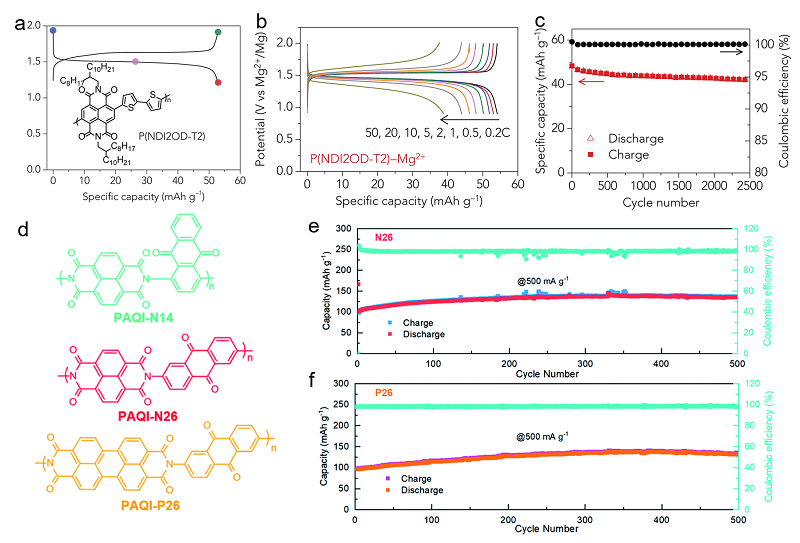

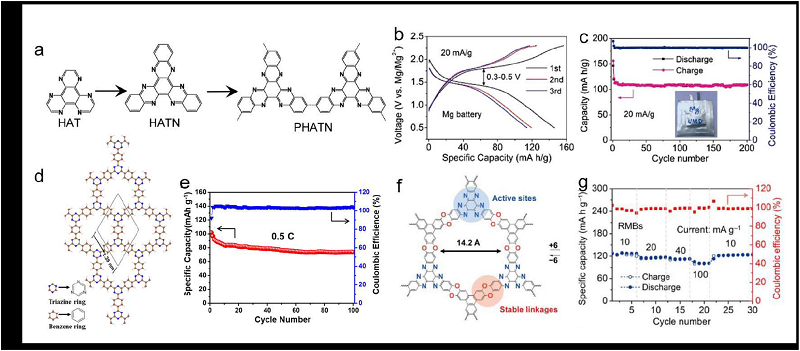
| [1] |
Dunn, B.; Kamath, H.; Tarascon, J. M. Science 2011, 334, 928.
|
| [2] |
Etacheri, V.; Marom, R.; Elazari, R.; Salitra, G.; Aurbach, D. Energy Environ. Sci. 2011, 4, 3243.
|
| [3] |
Melot, B. C.; Tarascon, J. M. Acc. Chem. Res. 2013, 46, 1226.
|
| [4] |
Lei, X.; Liang, X.; Yang, R.; Zhang, F.; Wang, C.; Lee, C. S.; Tang, Y. Small 2022, 18, 2200418.
|
| [5] |
Yoo, H. D.; Shterenberg, I.; Gofer, Y.; Gershinsky, G.; Pour, N.; Aurbach, D. Energy Environ. Sci. 2013, 6, 2265.
|
| [6] |
Gregory, T. D.; Hoffman, R. J.; Winterton, R. C. J. Electrochem. Soc. 1990, 137, 775.
|
| [7] |
Aurbach, D.; Lu, Z.; Schechter, A.; Gofer, Y.; Gizbar, H.; Turgeman, R.; Cohen, M.; Moshkovich, M.; Levi, E. Nature 2000, 407, 724.
|
| [8] |
Zhang, R.; Yu, X.; Nam, K. W.; Ling, C.; Arthur, T. S.; Song, W.; Knapp, A. M.; Ehrlich, S. N.; Yang, X.; Matsui, M. Electrochem. Commun. 2012, 23, 110.
|
| [9] |
Cheng, Y.; Shao, Y.; Raju, V.; Ji, X.; Mehdi, B. L.; Han, K. S.; Engelhard, M. H.; Li, G.; Browning, N. D.; Mueller, K. T.; Liu, J. Adv. Funct. Mater. 2016, 26, 3446.
|
| [10] |
Wang, L.; Asheim, K.; Vullum, P. E.; Svensson, A. M.; Vullum- Bruer, F. Chem. Mater. 2016, 28, 6459.
|
| [11] |
Truong, Q. D.; Devaraju, M. K.; Honma, I. J. Power Sources 2017, 361, 195.
|
| [12] |
NuLi, Y.; Yang, J.; Li, Y.; Wang, J. Chem. Commun. 2010, 46, 3794.
|
| [13] |
Sun, X.; Bonnick, P.; Nazar, L. F. ACS Energy Lett. 2 016, 1, 297.
|
| [14] |
Liang, Y.; Feng, R.; Yang, S.; Ma, H.; Liang, J.; Chen, J. Adv. Mater. 2012, 23, 640.
|
| [15] |
Liu, B.; Luo, T.; Mu, G.; Wang, X.; Chen, D.; Shen, G. ACS Nano 2013, 7, 8051.
|
| [16] |
Duffort, V.; Sun, X.; Nazar, L. F. Chem. Commun. 2016, 52, 12458.
|
| [17] |
Ding, Y.; Ren, X.; Chen, D.; Wen, F.; Li, T.; Xu, F. ACS Appl. Energy Mater. 2022, 5, 3004.
|
| [18] |
Sun, X.; Bonnick, P.; Duffort, V.; Liu, M.; Rong, Z.; Persson, K. A.; Ceder, G.; Nazar, L. F. Energy Environ. Sci. 2016, 9, 2273.
|
| [19] |
Tang, M.; Li, H.; Wang, E.; Wang, C. Chin. Chem. Lett. 2018, 29, 232.
|
| [20] |
Dawut, G.; Lu, Y.; Zhao, Q.; Liang, J.; Tao, Z. L.; Chen, J. Acta Phys.-Chim. Sin. 2016, 32, 1593. (in Chinese)
|
|
( 古丽巴哈尔•达吾提, 卢勇, 赵庆, 梁静, 陶占良, 陈军, 物理化学学报, 2016, 32, 1593.)
|
|
| [21] |
McAllister, B. T.; Kyne, L. T.; Schon, T. B.; Seferos, D. S. Joule 2019, 3, 620.
doi: 10.1016/j.joule.2018.12.005 |
| [22] |
Qin, K.; Huang, J.; Holguin, K.; Luo, C. Energy Environ. Sci. 2020, 13, 3950.
|
| [23] |
Song, Z.; Zhou, H. Energy Environ. Sci. 2013, 6, 2280.
|
| [24] |
Williams, D. L.; Byrne, J. J.; Driscoll, J. S. J. Electrochem. Soc. 1969, 116, 2.
|
| [25] |
Kumar, G.; Sivashanmugam, A.; Muniyandi, N.; Dhawan, S. K.; Trivedi, D. C. Synth. Met. 1996, 80, 279.
|
| [26] |
Shadike, Z.; Tan, S.; Wang, Q. C.; Lin, R.; Hu, E.; Qu, D.; Yang, X. Q. Mater. Horiz. 2021, 8, 471.
|
| [27] |
NuLi, Y.; Guo, Z.; Liu, H.; Yang, J. Electrochem. Commun. 2007, 9, 1913.
|
| [28] |
NuLi, Y.; Chen, Q.; Wang, W.; Wang, Y.; Yang, J.; Wang, J. Sci. World J. 2014, 2014, 107918.
|
| [29] |
Lu, Y.; Zhang, Q.; Li, L.; Niu, Z.; Chen, J. Chem 2018, 4, 2786.
|
| [30] |
Chen, Q.; NuLi, Y.; Guo, W.; Yang, J.; Wang, J.; Guo, Y. Acta Phys.- Chim. Sin. 2013, 29, 2295.
|
| [31] |
Ha, S. Y.; Lee, Y. W.; Woo, S. W.; Koo, B.; Kim, J. S.; Cho, J.; Lee, K.; Choi, N. S. ACS Appl. Mater. Interfaces 2014, 6, 4063.
|
| [32] |
Häupler, B.; Wild, A.; Schubert, U. S. Adv. Energy Mater. 2015, 5, 1402034.
|
| [33] |
Xie, J.; Zhang, Q. Small 2019, 15, 1805061.
|
| [34] |
Sano, H.; Senoh, H.; Yao, M.; Sakaebe, H.; Kiyobayashi, T. Chem. Lett. 2012, 41, 1594.
|
| [35] |
Senoh, H.; Sakaebe, H.; Sano, H.; Yao, M.; Kuratani, K.; Takeichi, N.; Kiyobayashi, T. J. Electrochem. Soc. 2014, 161, A1315.
|
| [36] |
Pan, B.; Zhou, D.; Huang, J.; Zhang, L.; Burrell, A. K.; Vaughey, J. T.; Zhang, Z.; Liao, C. J. Electrochem. Soc. 2016, 163, A580.
|
| [37] |
Debashis, T.; Viswanatha, H. M.; Harish, M. N. K.; Sampath, S. J. Electrochem. Soc. 2020, 167, 070561.
|
| [38] |
Bitenc, J.; Pirnat, K.; Bančič, T.; Gaberšček, M.; Genorio, B.; Randon-Vitanova, A.; Dominko, R. ChemSusChem 2015, 8, 4128.
doi: 10.1002/cssc.201500910 pmid: 26610185 |
| [39] |
Song, Z.; Qian, Y.; Gordin, M. L.; Tang, D.; Xu, T.; Otani, M.; Zhan, H.; Zhou, H. Angew. Chem. Int. Ed. 2015, 54, 13947.
|
| [40] |
Pan, B.; Huang, J.; Feng, Z.; Zeng, L.; He, M.; Zhang, L.; Vaughey, J. T.; Bedzyk, M. J.; Fenter, P.; Zhang, Z.; Burrell, A. K.; Liao, C. Adv. Energy Mater. 2016, 6, 1600140.
|
| [41] |
Bitenc, J.; Pirnat, K.; Žagar, E.; Randon-Vitanova, A.; Dominko, R. J. Power Sources 2019, 430, 90.
doi: 10.1016/j.jpowsour.2019.04.114 |
| [42] |
Bitenc, J.; Pirnat, K.; Mali, G.; Novosel, B.; Vitanova, A. R.; Dominko, R. Electrochem. Commun. 2016, 69, 1.
|
| [43] |
Tian, J.; Cao, D.; Zhou, X.; Hu, J.; Huang, M.; Li, C. ACS Nano 2018, 12, 3424.
|
| [44] |
Dong, H.; Tutusaus, O.; Liang, Y.; Zhang, Y.; Lebens-Higgins, Z.; Yang, W.; Mohtadi, R.; Yao, Y. Nat. Energy 2020, 5, 1043.
|
| [45] |
Rodríguez-Pérez, I. A.; Yuan, Y.; Bommier, C.; Wang, X.; Ma, L.; Leonard, D. P.; Lerner, M. M.; Carter, R. G.; Wu, T.; Greaney, P. A.; Lu, J.; Ji, X. J. Am. Chem. Soc. 2017, 139, 13031.
doi: 10.1021/jacs.7b06313 pmid: 28823162 |
| [46] |
Cui, L.; Zhou, L.; Zhang, K.; Xiong, F.; Tan, S.; Li, M.; An, Q.; Kang, Y. M.; Mai, L. Nano Energy 2019, 65, 103902.
|
| [47] |
Yang, H.; Xu, F. ChemPhysChem 2021, 22, 1455.
|
| [48] |
Bančič, T.; Bitenc, J.; Pirnat, K.; Lautar, A. K.; Grdadolnik, J.; Vitanova, A. R.; Dominko, R. J. Power Sources 2018, 395, 25.
|
| [49] |
Fan, X.; Wang, F.; Ji, X.; Wang, R.; Gao, T.; Hou, S.; Chen, J.; Deng, T.; Li, X.; Chen, L.; Luo, C.; Wang, L.; Wang, C. Angew. Chem. Int. Ed. 2018, 57, 7146.
|
| [50] |
Wang, Y.; Liu, Z.; Wang, C.; Hu, Y.; Lin, H.; Kong, W.; Ma, J.; Jin, Z. Energy Storage Mater. 2020, 26, 494.
|
| [51] |
Dong, H.; Liang, Y.; Tutusaus, O.; Mohtadi, R.; Zhang, Y.; Hao, F.; Yao, Y. Joule 2019, 3, 782.
doi: 10.1016/j.joule.2018.11.022 |
| [52] |
Ding, Y.; Chen, D.; Ren, X.; Cao, Y.; Xu, F. J. Mater. Chem. A 2022, 10, 14111.
|
| [53] |
Cao, Y.; Wang, M.; Wang, H.; Han, C.; Pan, F.; Sun, J. Adv. Energy Mater. 2022, 12, 2200057.
|
| [54] |
Li, J.; Jing, X.; Li, Q.; Li, S.; Gao, X.; Feng, X.; Wang, B. Chem. Soc. Rev. 2020, 49, 3565.
|
| [55] |
Mao, M.; Luo, C.; Pollard, T. P.; Hou, S.; Gao, T.; Fan, X.; Cui, C.; Yue, J.; Tong, Y.; Yang, G.; Deng, T.; Zhang, M.; Ma, J.; Suo, L.; Borodin, O.; Wang, C. Angew. Chem., Int. Ed. 2019, 58, 17820.
|
| [56] |
Sun, R.; Hou, S.; Luo, C.; Ji, X.; Wang, L.; Mai, L.; Wang, C. Nano Lett. 2020, 20, 3880.
|
| [57] |
Li, S.; Liu, Y.; Dai, L.; Li, S.; Wang, B.; Xie, J.; Li, P. Energy Storage Mater. 2022, 48, 439.
|
| [1] | Jiawei He, Liu Jiao, Xueyi Cheng, Guanghai Chen, Qiang Wu, Xizhang Wang, Lijun Yang, Zheng Hu. Structural Regulation of Metal Organic Framework-derived Hollow Carbon Nanocages and Their Lithium-Sulfur Battery Performance [J]. Acta Chimica Sinica, 2022, 80(7): 896-902. |
| [2] | Shouxiao Chen, Junke Liu, Weichen Zheng, Guozhen Wei, Yao Zhou, Juntao Li. Electron/ion Conductor Double-coated LiNi0.8Co0.1Mn0.1O2 Li-ion Battery Cathode Material and Its Electrochemical Performance [J]. Acta Chimica Sinica, 2022, 80(4): 485-493. |
| [3] | Kai Qiu, Mingxia Yan, Shouwang Zhao, Shengli An, Wei Wang, Guixiao Jia. Theoretical Study on the Structural Stability and Oxygen Ion Oxidation of Al-doped Lithium-ion Battery Layered Cathode Li(Li0.17Ni0.17Al0.04Fe0.13Mn0.49)O2 [J]. Acta Chimica Sinica, 2021, 79(9): 1146-1153. |
| [4] | Tongxin Li, Donglin Li, Qingbo Zhang, Jianhang Gao, Xiangze Kong, Xiaoyong Fan, Lei Gou. Preparation and Electrochemical Performance of Macroporous Ni-rich LiNi0.8Co0.1Mn0.1O2 Cathode Material [J]. Acta Chimica Sinica, 2021, 79(5): 678-684. |
| [5] | Bixia Lin, Yingshan Huang, Shuai Chen, Zhenyu Xing. Research Progress of Key Materials for Sodium-selenium Batteries [J]. Acta Chimica Sinica, 2021, 79(5): 641-648. |
| [6] | Yanli Li, Dandan Yu, Sen Lin, Dongfei Sun, Ziqiang Lei. Preparation of α-MnO2 Nanorods/Porous Carbon Cathode for Aqueous Zinc-ion Batteries [J]. Acta Chimica Sinica, 2021, 79(2): 200-207. |
| [7] | Lu Zhang, Wenfeng Wang, Hongming Zhang, Shumin Han, Limin Wang. Research Progress and Challenge of Aqueous Zinc Ion Battery [J]. Acta Chimica Sinica, 2021, 79(2): 158-175. |
| [8] | Qimei Liang, Yujiao Guo, Junming Guo, Mingwu Xiang, Xiaofang Liu, Wei Bai, Ping Ning. Preparation and High Temperature Electrochemical Performance of LiNi0.08Mn1.92O4 Cathode Material of Submicron Truncated Octahedron [J]. Acta Chimica Sinica, 2021, 79(12): 1526-1533. |
| [9] | Jisheng Xie, Zhumei Xiao, Wenhua Zuo, Yong Yang. Research Progresses of Sodium Cobalt Oxide as Cathode in Sodium Ion Batteries [J]. Acta Chimica Sinica, 2021, 79(10): 1232-1243. |
| [10] | Tang Gong-ao, Mao Kun, Zhang Jing, Lyu Pin, Cheng Xueyi, Wu Qiang, Yang Lijun, Wang Xizhang, Hu Zheng. Hierarchical Nitrogen-doped Carbon Nanocages as High-rate Long-life Cathode Material for Rechargeable Magnesium Batteries [J]. Acta Chimica Sinica, 2020, 78(5): 444-450. |
| [11] | Liu Jiuding, Zhang Yudong, Liu Junxiang, Li Jinhan, Qiu Xiaoguang, Cheng Fangyi. In-situ Li3PO4 Coating of Li-Rich Mn-Based Cathode Materials for Lithium-ion Batteries [J]. Acta Chimica Sinica, 2020, 78(12): 1426-1433. |
| [12] | Ren Xuqiang, Li Donglin, Zhao Zhenzhen, Chen Guangqi, Zhao Kun, Kong Xiangze, Li Tongxin. Dual Effect of Aluminum Doping and Lithium Tungstate Coating on the Surface Improves the Cycling Stability of Lithium-rich Manganese-based Cathode Materials [J]. Acta Chimica Sinica, 2020, 78(11): 1268-1274. |
| [13] | Song Xuexi, Li Jicheng, Li Zhaohui, Li Xifei, Ding Yanhuai, Xiao Qizhen, Lei Gangtie. Effect of K-Doping on the Sodium-storage Performance of Sodium Vanadate Nanoplates [J]. Acta Chim. Sinica, 2019, 77(7): 625-633. |
| [14] | Li Zhao, Wang Zhong, Ban Liqin, Wang Jiantao, Lu Shigang. Recent Advances on Surface Modification of Li- and Mn-Rich Cathode Materials [J]. Acta Chimica Sinica, 2019, 77(11): 1115-1128. |
| [15] | Wang Ling, Yang Guorui, Wang Jianan, Wang Silan, Peng Shengjie, Yan Wei. Research Progress on Electrospun Materials for Sodium-Ion Batteries [J]. Acta Chim. Sinica, 2018, 76(9): 666-680. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||