化学学报 ›› 2026, Vol. 84 ›› Issue (3): 275-279.DOI: 10.6023/A26010002 上一篇 下一篇
研究通讯
王翔宇a, 夏天骐a, 冷雪冰a, 王东阳a, 邓亮a,b,*( )
)
Wang Xiangyua, Xia Tianqia, Leng Xuebinga, Wang Dongyanga, Deng Lianga,b,*( )
)
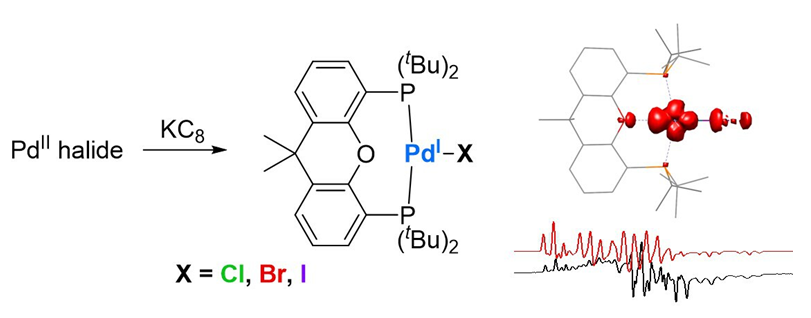
单核一价钯卤化物是许多钯催化反应的中间体, 然而目前报道可分离的单核一价钯卤化物极少. 本研究发现, 使用4,5-双(二叔丁基膦)-9,9-二甲基氧杂蒽(tBuXantphos)作为配体可以有效稳定单核一价钯卤化物. 单核一价钯氯化物[(tBuXantphos)PdCl]可以通过石墨钾还原[(tBuXantphos)PdCl2]得到, 单核一价钯溴化物[(tBuXantphos)PdBr]与碘化物[(tBuXantphos)PdI]则由过量NaX (X=Br, I)与[(tBuXantphos)PdCl2]反应后进一步用石墨钾还原得到. 单晶X-射线衍射确定[(tBuXantphos)PdX] (X=Cl, Br, I)具有扭曲的T-构型结构. 100 K下的电子顺磁共振谱图显示, [(tBuXantphos)PdX]的g因子g=[2.231, 2.079, 2.034] (X=Cl), [2.195, 2.081, 2.035] (X=Br), [2.201, 2.129, 2.074] (X=I), 卤素原子引起的超精细耦合常数A(X)=[33, 27, 90] MHz (X=Cl), [312, 75, 425] MHz (X=Br), [360, 150, 600] MHz (X=I). 密度泛函理论计算结果表明, 这些一价钯卤化物的单占据分子轨道为Pd的dx2-y2轨道与配体np轨道之间所成的σ*轨道, Pd中心的自旋密度分布从氯化物到碘化物依次减少, 分别为0.63, 0.61和0.57, 卤素原子上的自旋密度分布依次增加, 分别为0.17, 0.19和0.22.