化学学报 ›› 2026, Vol. 84 ›› Issue (3): 425-447.DOI: 10.6023/A25110360 上一篇 下一篇
综述
周铭宇a,c, 刘乐彬a,c, 梅雷a, 刘雅兰a,*( ), 石伟群b,*(
), 石伟群b,*( )
)
投稿日期:2025-11-06
发布日期:2025-12-31
作者简介: |
周铭宇, 中国科学院高能物理研究所博士研究生, 主要 从事金属锆的熔盐电化学研究. |
 |
刘乐彬, 中国科学院高能物理研究所硕士研究生, 主要 从事干法后处理研究. |
 |
梅雷, 中国科学院高能物理研究所, 研究员. 主要研究方向为锕系元素化学与分离材料, 致力于将超分子识别与组装的理念和方法应用于锕系固体化学和放射性核素分离化学研究, 在锕系超分子配合物及其配位化学基础、基于协同识别的放射性核素分离新方法、新型锕系固相材料开发等方面开展了系列原创性工作, 为解决乏燃料后处理、环境放射性污染控制与资源回用研究等领域中的关键问题提供了新思路. 以通讯/第一作者身份在Nature Commun., J. Am. Chem. Soc., Angew. Chem. Int. Ed., Adv. Funct. Mater.等国际知名期刊发表研究论文130余篇. 目前担任中国化学会青年化学工作者委员会委员, 中科院青促会化学与材料分会委员, 中国核学会锕系化学与物理分会理事, 并担任《核化学与放射化学》期刊编委, SmartMat、Materials Research Letters、《结构化学》等学术期刊青年编委. |
 |
刘雅兰, 副研究员, 中国科学院高能物理研究所. 多年来致力于氧化物乏燃料干法后处理领域, 聚焦于锕-镧分离研究. 首先开展了锕、镧系氧化物在熔盐中的溶解及其电化学行为研究, 随后在固态活性铝阴极上进行了锕-镧的电化学分离, 并采用原位光谱技术监测了分离过程中锕、镧元素的化学种态变化, 发现了铀的循环电解并将其消除, 提高了电流效率. 最终成功实现了锕-镧元素的有效分离, 与传统的液态Cd阴极相比将分离因子提高了两个数量级. 在此基础上, 进一步总结了锕、镧氧化物在氯化物熔盐中的溶解规律, 提出了利用其溶解性差异实现一步分离的新方法. 基于相关工作, 在电化学领域与核能领域著名期刊Electrochim. Acta, J. Electrochem. Soc., Electrochem. Commun.和 J. Nucl. Mater.等上共发表论文40余篇, 其中第一作者及通讯作者论文20篇. |
 石伟群, 上海交通大学特聘教授, 核燃料循环与核材料研究所所长, 国家杰出青年科学基金获得者. 2007年1月在清华大学化学系获博士学位. 长期致力于核燃料循环化学相关基础研究, 在JACS、Angew. Chem、Chem.、CCS Chem.、Nat. Commun、Adv. Mater.等国际知名期刊发表SCI论文400余篇, 成果被国内外同行广泛关注和引用, 文章总引两万余次, H因子71 (Google Scholar), 2020~2024年连续入选Elsevier中国高被引学者榜单(核科学技术). 分别担任期刊《Supramolecular Materials》副主编, 《Industrial Chemistry & Materials》、《Chinese Chemical Letters》、《Journal of Nuclear Fuel Cycle and Waste Technology》、《International Journal of Advanced Nuclear Reactor Design and Technology》和《Journal of Nuclear Science and Technology》的编委与国际顾问编委, 中文期刊《化学学报》、《高等学校化学学报》、《核化学与放射化学》、《核动力》编委. 现为中国核工业教育学会副理事长、中国核学会锕系物理与化学分会副理事长、中国有色金属学会熔盐化学与技术专业委员会副主任委员、中国化学会核化学与放射化学专业委员会委员、中国核学会核化工分会常务理事兼副秘书长.
石伟群, 上海交通大学特聘教授, 核燃料循环与核材料研究所所长, 国家杰出青年科学基金获得者. 2007年1月在清华大学化学系获博士学位. 长期致力于核燃料循环化学相关基础研究, 在JACS、Angew. Chem、Chem.、CCS Chem.、Nat. Commun、Adv. Mater.等国际知名期刊发表SCI论文400余篇, 成果被国内外同行广泛关注和引用, 文章总引两万余次, H因子71 (Google Scholar), 2020~2024年连续入选Elsevier中国高被引学者榜单(核科学技术). 分别担任期刊《Supramolecular Materials》副主编, 《Industrial Chemistry & Materials》、《Chinese Chemical Letters》、《Journal of Nuclear Fuel Cycle and Waste Technology》、《International Journal of Advanced Nuclear Reactor Design and Technology》和《Journal of Nuclear Science and Technology》的编委与国际顾问编委, 中文期刊《化学学报》、《高等学校化学学报》、《核化学与放射化学》、《核动力》编委. 现为中国核工业教育学会副理事长、中国核学会锕系物理与化学分会副理事长、中国有色金属学会熔盐化学与技术专业委员会副主任委员、中国化学会核化学与放射化学专业委员会委员、中国核学会核化工分会常务理事兼副秘书长.
Zhou Mingyua,c, Liu Lebina,c, Mei Leia, Liu Yalana,*( ), Shi Weiqunb,*(
), Shi Weiqunb,*( )
)
Received:2025-11-06
Published:2025-12-31
Contact:
*E-mail: liuyalan@ihep.ac.cn;
shiwq@ihep.ac.cn
文章分享
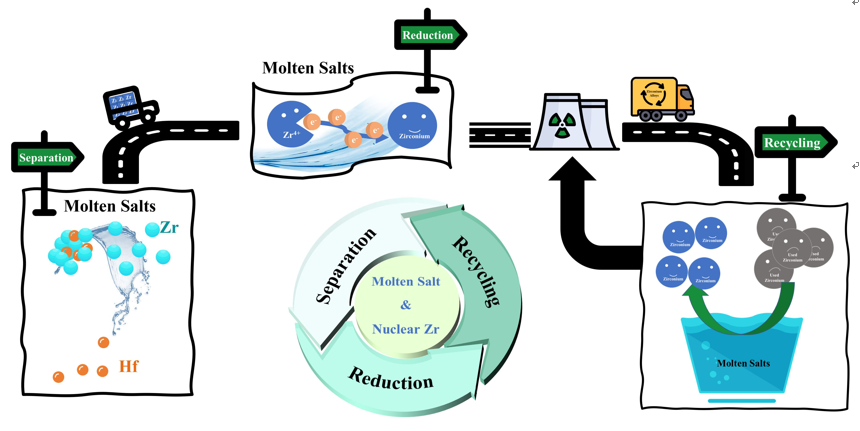
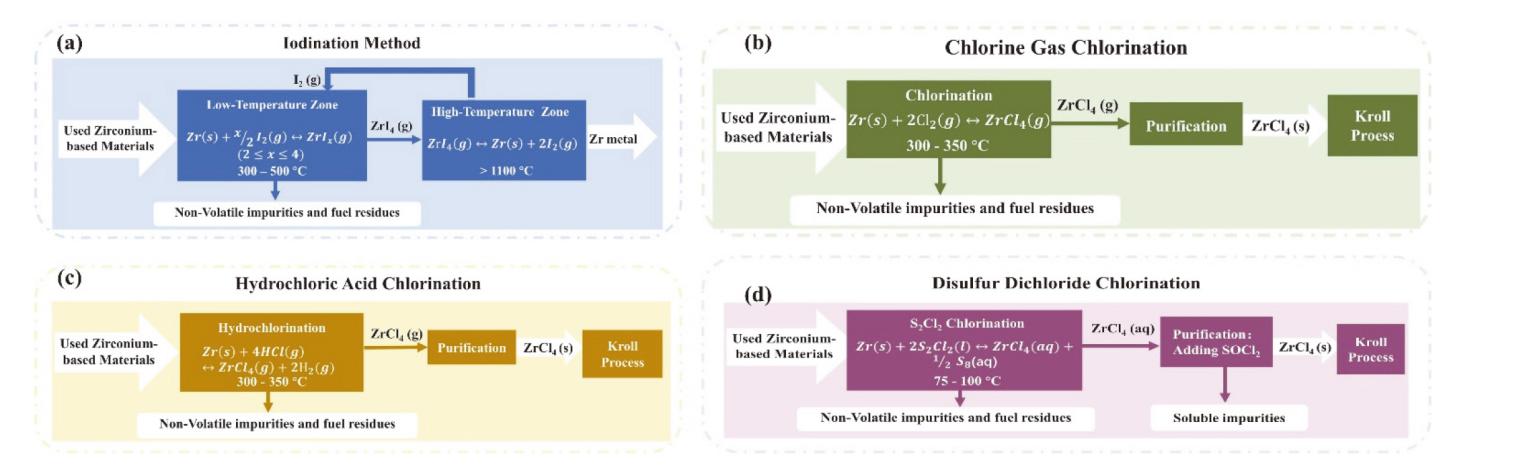
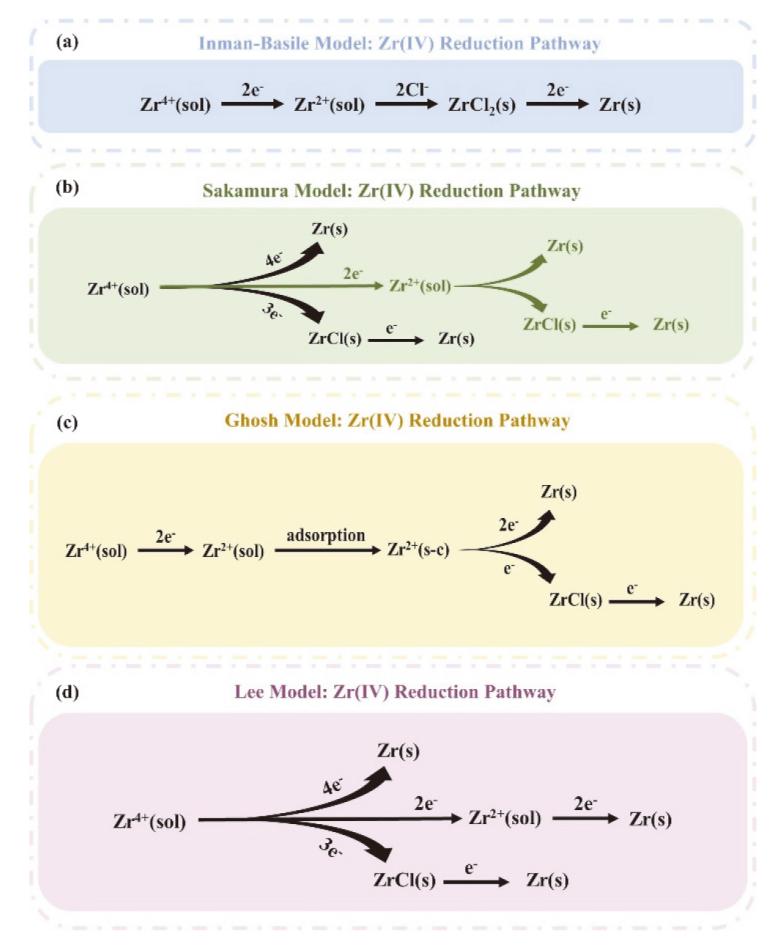
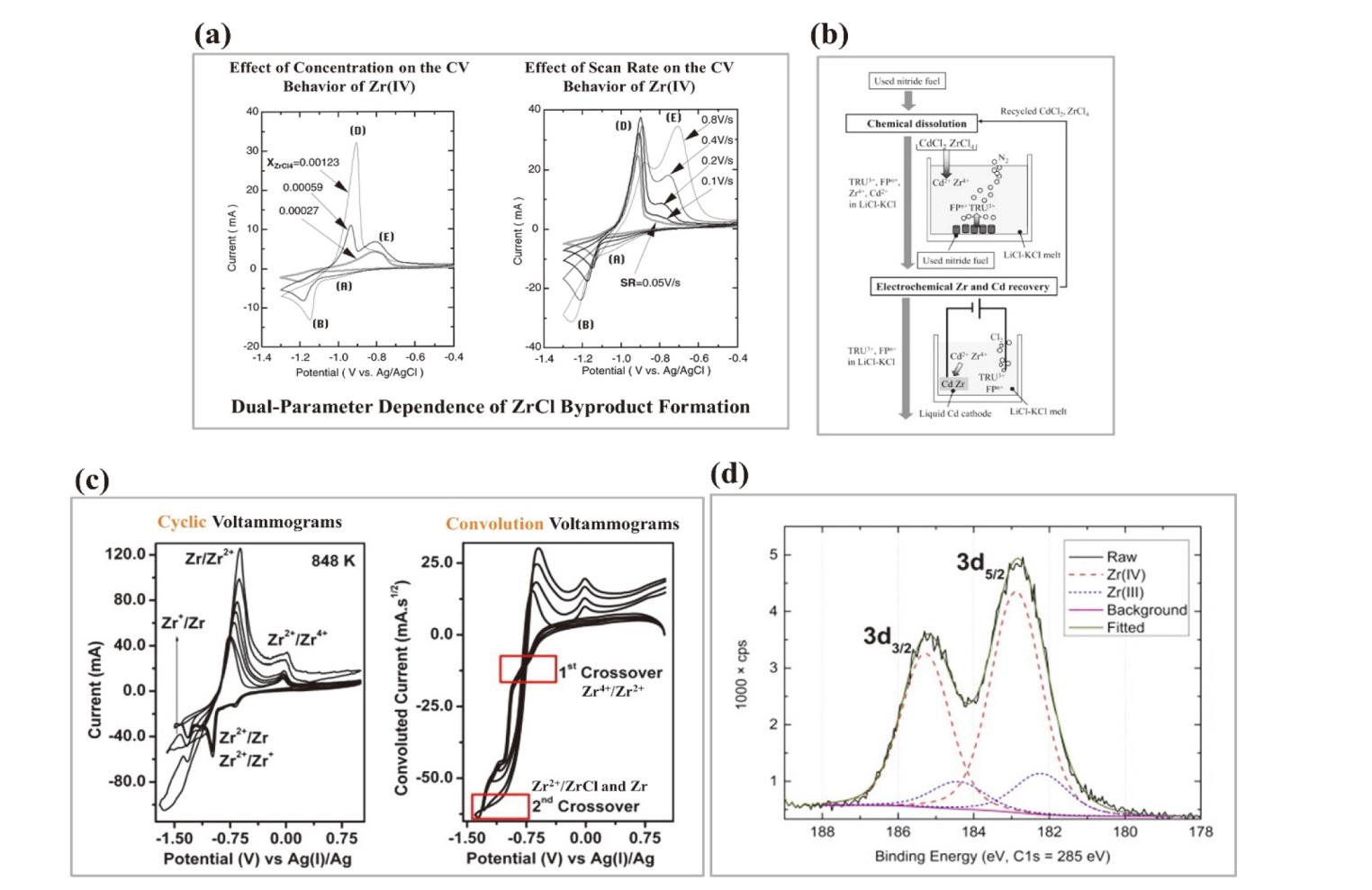
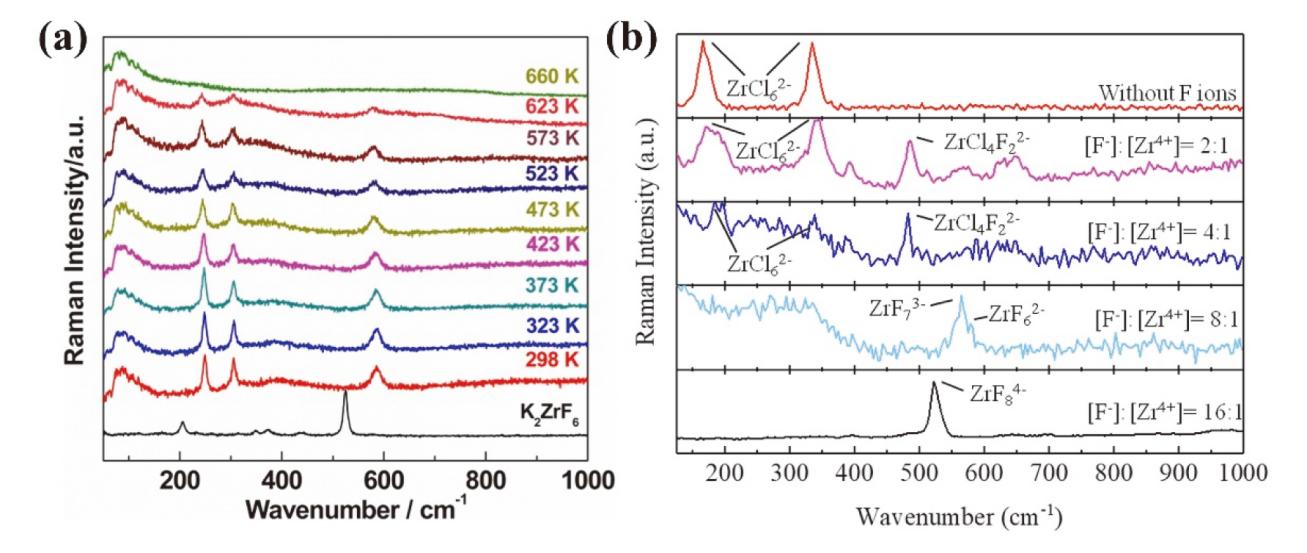
核级锆是核电站反应堆的必备金属材料, 具有普通商用锆材无法企及的低中子吸收截面与耐腐蚀性, 兼具高经济价值与战略意义, 是关乎国家能源安全的储备性资源. 因此, 高效经济地制备和回收核级锆受到核科学工作者们的重视. 熔盐技术作为实现锆铪分离、金属锆制备及退役锆回收的重要途径, 具有流程简洁、适应性强等优势. 此综述概括了熔盐在核级锆全流程管理中的关键作用, 包括不同熔盐体系(氟化物、氯化物及氟氯混合盐体系)在金属锆制备和回收中的应用特性分析和对比. 此外, 重点对熔盐中锆离子的电还原行为进行了系统的梳理, 以推动核级锆的绿色冶金与循环利用.
周铭宇, 刘乐彬, 梅雷, 刘雅兰, 石伟群. 核级锆的熔盐制备与回收研究进展[J]. 化学学报, 2026, 84(3): 425-447.
Zhou Mingyu, Liu Lebin, Mei Lei, Liu Yalan, Shi Weiqun. Research Progress on the Preparation and Recovery of Nuclear-Grade Zirconium Using Molten Salts[J]. Acta Chimica Sinica, 2026, 84(3): 425-447.
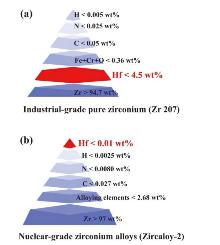
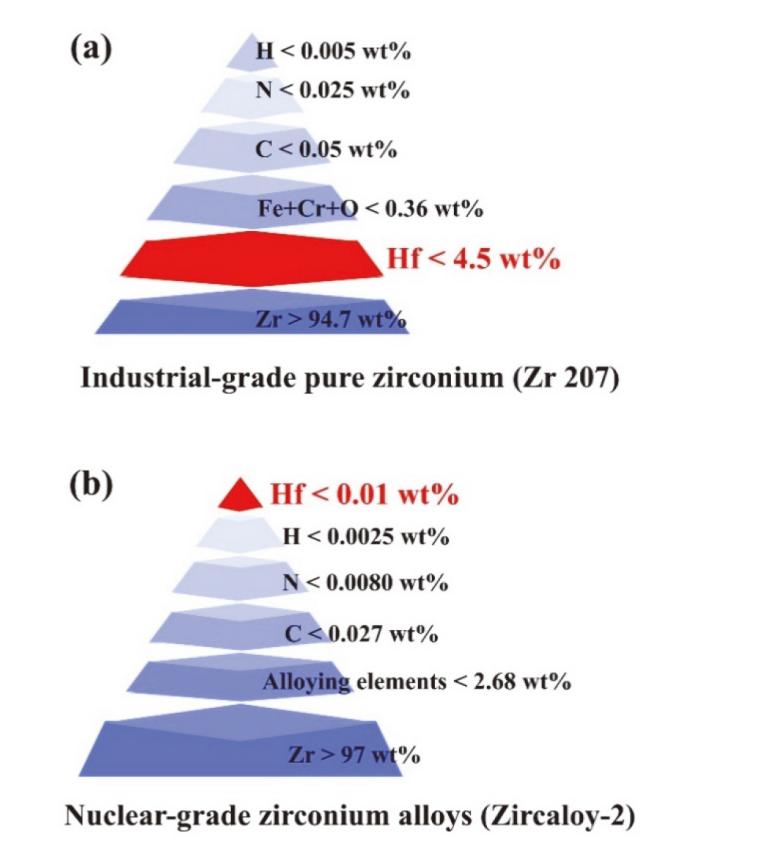
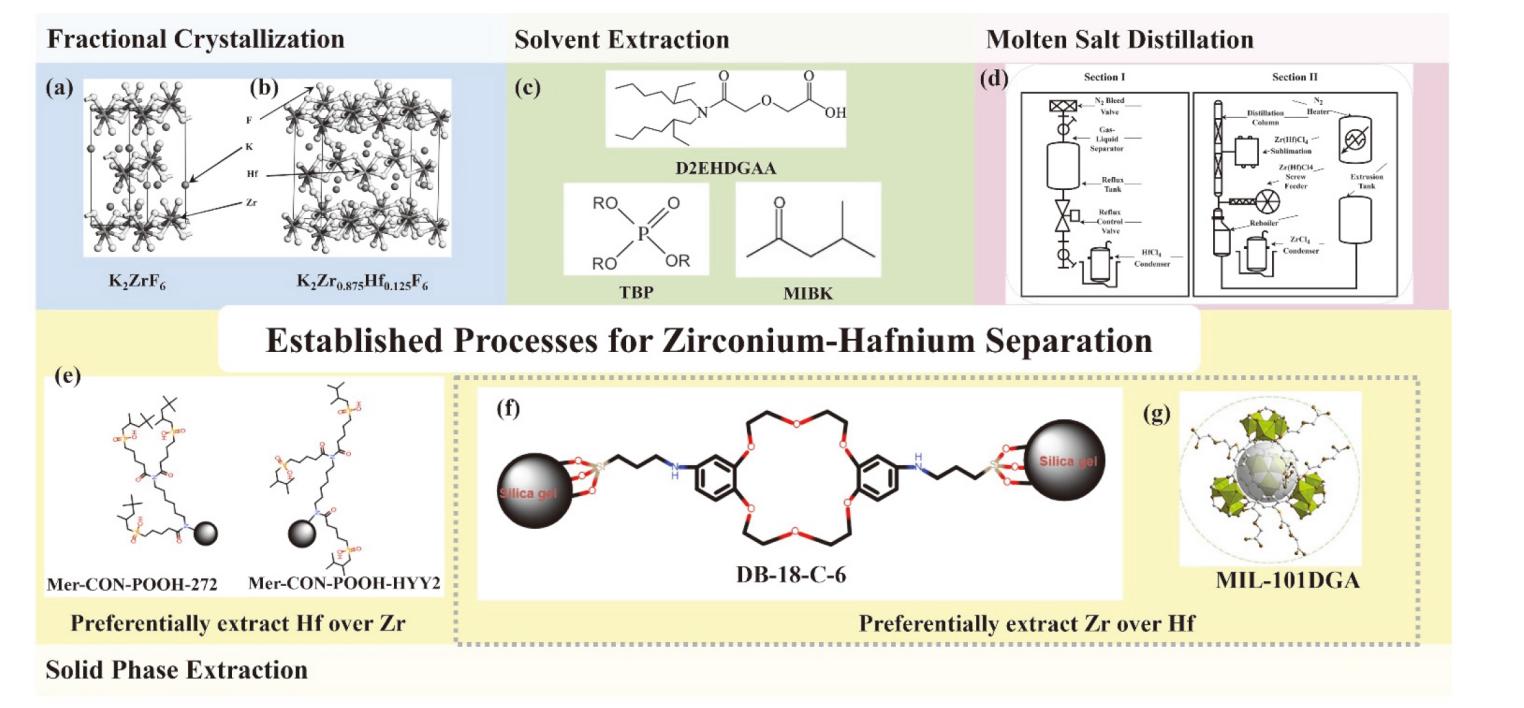
| Methods | Advantage | Challenges |
|---|---|---|
| Fractional Crystallization | Simplicity principle; Mild operating conditions | Low separation factor; Low productivity; Intermittent operation |
| Solvent Extraction | Large-scale production; Mature process; Separation efficiency | High consumption; High waste generation; Process complexity |
| Solid-phase Extraction | Eco-Friendly; Highly efficient | Adsorbent cost; Low stability |
| Molten Salt Distillation | Process simplicity; Simplifies downstream; Waste minimization | High temperature; Corrosivity; High energy consumption |
| Methods | Advantage | Challenges |
|---|---|---|
| Fractional Crystallization | Simplicity principle; Mild operating conditions | Low separation factor; Low productivity; Intermittent operation |
| Solvent Extraction | Large-scale production; Mature process; Separation efficiency | High consumption; High waste generation; Process complexity |
| Solid-phase Extraction | Eco-Friendly; Highly efficient | Adsorbent cost; Low stability |
| Molten Salt Distillation | Process simplicity; Simplifies downstream; Waste minimization | High temperature; Corrosivity; High energy consumption |
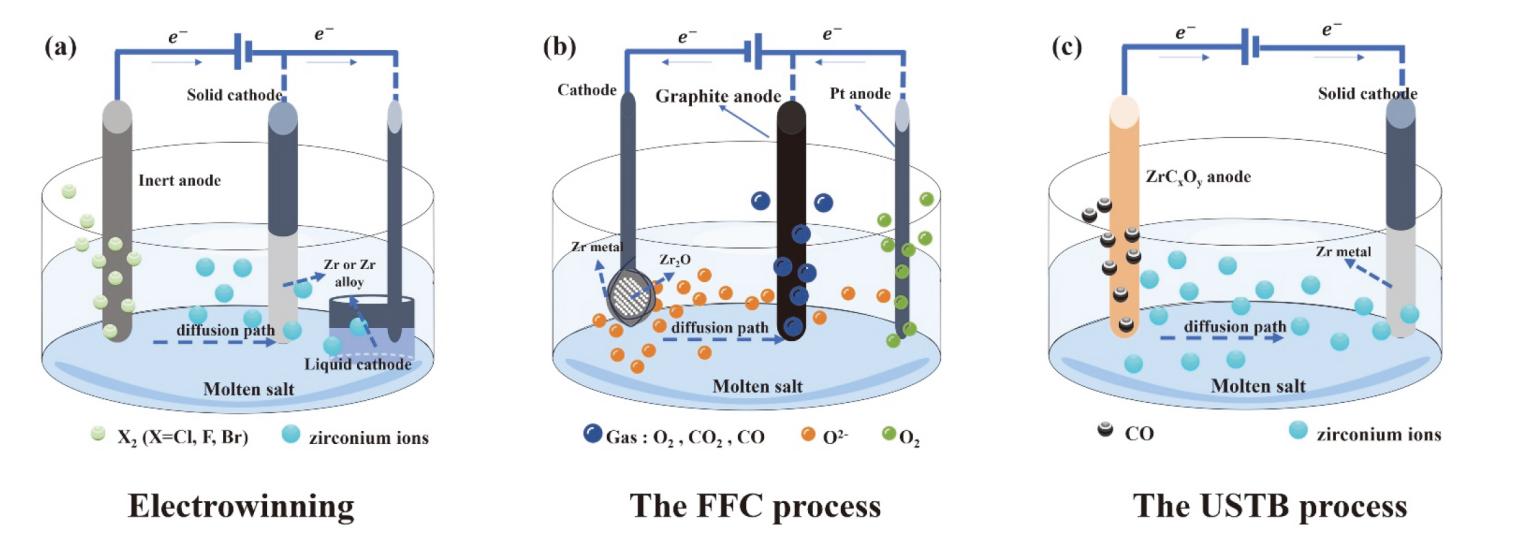


| Components | Melting/Eutectic Temp. (℃) | Characteristics/Properties | Primary Applications in Zr Industry |
|---|---|---|---|
| NaCl | 801 | | Molten Salt Distillation; Molten Salt Electrolysis for Zr/Hf Separation; Electrowinning for Zirconium Production. |
| MgCl2 | 714 | | Kroll Process for Zirconium Production. |
| CaCl2 | 772 | | FFC Process for Zirconium Production. |
| NaCl-KCl | 657 | | Molten Salt Distillation; Molten Salt Extraction for Zr/Hf Separation; Molten Salt Electrolysis for Zr/Hf Separation; Electrowinning for Zirconium Production; USTB Process for Zirconium Production. |
| LiCl-KCl | 352 | | Molten Salt Electrolysis for Zr/Hf Separation; Electrowinning for Zirconium Production; Electrorefining for Zirconium Recovery. |
| LiF-KF | 492 | | Electrorefining for Zirconium Recovery. |
| LiF-NaF | 649 | ||
| LiF-NaF-KF | 460 |
| Components | Melting/Eutectic Temp. (℃) | Characteristics/Properties | Primary Applications in Zr Industry |
|---|---|---|---|
| NaCl | 801 | | Molten Salt Distillation; Molten Salt Electrolysis for Zr/Hf Separation; Electrowinning for Zirconium Production. |
| MgCl2 | 714 | | Kroll Process for Zirconium Production. |
| CaCl2 | 772 | | FFC Process for Zirconium Production. |
| NaCl-KCl | 657 | | Molten Salt Distillation; Molten Salt Extraction for Zr/Hf Separation; Molten Salt Electrolysis for Zr/Hf Separation; Electrowinning for Zirconium Production; USTB Process for Zirconium Production. |
| LiCl-KCl | 352 | | Molten Salt Electrolysis for Zr/Hf Separation; Electrowinning for Zirconium Production; Electrorefining for Zirconium Recovery. |
| LiF-KF | 492 | | Electrorefining for Zirconium Recovery. |
| LiF-NaF | 649 | ||
| LiF-NaF-KF | 460 |
| Technique | Description/Formula Name | Equation | Applicable System & Notes |
|---|---|---|---|
| EFMa | Nernst Equation | | Ecell: Measured potential (V) C: Concentration of the electroactive species (mol/L) |
| LSV/CV | Reversible System Half-Wave Potential | | Reversible System: Fast electron transfer. EP/2: Half-wave potential (V) EP: Peak potential (V) Constant 2.2 is characteristic of a reversible process. |
| Irreversible System Half-Wave Potential | | Totally Irreversible System: Slow electron transfer. Constant 1.85 is characteristic of an irreversible process. | |
| SWV | Peak Width at Half Height | | W1/2: Peak width at half height (V) Applicable when electron transfer is sufficiently fast. |
| CP | Sand's Equation | | Used to calculate the diffusion coefficient D or n. i: Applied constant current (A) A: Electrode area (cm2) C: Bulk concentration (mol/cm3) |
| Calculation | Randles-Sevcik Equation | | Primarily used in LSV/CV to calculate the diffusion coefficient D from the peak current ip. ip: Peak current (A) ν: Scan rate (V/s) |
| Technique | Description/Formula Name | Equation | Applicable System & Notes |
|---|---|---|---|
| EFMa | Nernst Equation | | Ecell: Measured potential (V) C: Concentration of the electroactive species (mol/L) |
| LSV/CV | Reversible System Half-Wave Potential | | Reversible System: Fast electron transfer. EP/2: Half-wave potential (V) EP: Peak potential (V) Constant 2.2 is characteristic of a reversible process. |
| Irreversible System Half-Wave Potential | | Totally Irreversible System: Slow electron transfer. Constant 1.85 is characteristic of an irreversible process. | |
| SWV | Peak Width at Half Height | | W1/2: Peak width at half height (V) Applicable when electron transfer is sufficiently fast. |
| CP | Sand's Equation | | Used to calculate the diffusion coefficient D or n. i: Applied constant current (A) A: Electrode area (cm2) C: Bulk concentration (mol/cm3) |
| Calculation | Randles-Sevcik Equation | | Primarily used in LSV/CV to calculate the diffusion coefficient D from the peak current ip. ip: Peak current (A) ν: Scan rate (V/s) |
| FLiNaK (LiF:NaF:KF) | |||||
|---|---|---|---|---|---|
| Author | Time | Zr(IV) Ion Source | Electrode | Electroreduction behavior of Zr(IV) | D(Zr4+)/(cm2•s−1) |
| Mellors[ | 1966 | ZrF4 & K2ZrF6 | Mo | Zr(IV)→Zr | — |
| Li[ | 2023 | K2ZrF6 | Cu | | — |
| Zuo[ | 2025 | ZrF4 | W | | 3.85×10−6 (600 ℃) |
| LiF-CaF2 | |||||
| Gibilaro[ | 2013 | ZrF4 | Ta, Cu, Ni | Zr(IV)→Zr | 9.96×10−6 (840 ℃) |
| Fabian[ | 2022 | ZrF4 | Mo, Ni | Zr(IV)→Zr0(sol)→Zr | 2.6×10−5 (840 ℃) |
| LiF-KF | |||||
| Mellors[ | 1966 | ZrF4 & K2ZrF6 | Mo | Zr(IV)→Zr | — |
| Park[ | 2013 | ZrF4 | Mo | Zr(IV)→Zr | — |
| Xu[ | 2016 | ZrF4 | Mo | Step 1: Zr4+→Zr2+ Step 2: Zr4+, Zr2+→Zr+ Step 3: Zr+, Zr2+, Zr4+→Zr | 1.32×10−6~1.53×10−5 (600 ℃) |
| LiF-NaF | |||||
| Groult[ | 2007 | ZrF4 | W, Mo, C, Ni | Zr(IV)→Zr | 1.19×10−5 (694 ℃) 2.59×10−5 (730 ℃) 3.19×10−5 (762 ℃) |
| Groult[ | 2011 | n(LiF):n(KF):n(ZrF4)= (26:37:37) | C | Zr(IV)→ZrC | — |
| Xu[ | 2017 | K2ZrF6 | Mo | Zr(IV)→Zr(II) Zr(IV)/Zr(II)→Zr | 8.14×10−6~1.91×10−5 (750 ℃) |
| Quaranta[ | 2018 | ZrF4 | Ag | Zr(IV)→Zr | 1.21×10−5~1.35×10−5 (750 ℃) |
| FLiNaK (LiF:NaF:KF) | |||||
|---|---|---|---|---|---|
| Author | Time | Zr(IV) Ion Source | Electrode | Electroreduction behavior of Zr(IV) | D(Zr4+)/(cm2•s−1) |
| Mellors[ | 1966 | ZrF4 & K2ZrF6 | Mo | Zr(IV)→Zr | — |
| Li[ | 2023 | K2ZrF6 | Cu | | — |
| Zuo[ | 2025 | ZrF4 | W | | 3.85×10−6 (600 ℃) |
| LiF-CaF2 | |||||
| Gibilaro[ | 2013 | ZrF4 | Ta, Cu, Ni | Zr(IV)→Zr | 9.96×10−6 (840 ℃) |
| Fabian[ | 2022 | ZrF4 | Mo, Ni | Zr(IV)→Zr0(sol)→Zr | 2.6×10−5 (840 ℃) |
| LiF-KF | |||||
| Mellors[ | 1966 | ZrF4 & K2ZrF6 | Mo | Zr(IV)→Zr | — |
| Park[ | 2013 | ZrF4 | Mo | Zr(IV)→Zr | — |
| Xu[ | 2016 | ZrF4 | Mo | Step 1: Zr4+→Zr2+ Step 2: Zr4+, Zr2+→Zr+ Step 3: Zr+, Zr2+, Zr4+→Zr | 1.32×10−6~1.53×10−5 (600 ℃) |
| LiF-NaF | |||||
| Groult[ | 2007 | ZrF4 | W, Mo, C, Ni | Zr(IV)→Zr | 1.19×10−5 (694 ℃) 2.59×10−5 (730 ℃) 3.19×10−5 (762 ℃) |
| Groult[ | 2011 | n(LiF):n(KF):n(ZrF4)= (26:37:37) | C | Zr(IV)→ZrC | — |
| Xu[ | 2017 | K2ZrF6 | Mo | Zr(IV)→Zr(II) Zr(IV)/Zr(II)→Zr | 8.14×10−6~1.91×10−5 (750 ℃) |
| Quaranta[ | 2018 | ZrF4 | Ag | Zr(IV)→Zr | 1.21×10−5~1.35×10−5 (750 ℃) |

| Author | Year | Technique/Method | Electrolyte | Electroreduction behavior of Zr(IV) | Temp.a/℃ |
|---|---|---|---|---|---|
| Mellors[ | 1966 | ED b | NaCl-KCl-ZrCl4 NaCl-KCl-K2ZrF6 | ZrCl2 forms in electroreduction | 600~900 |
| Swaroop[ | 1966 | EFM | NaCl-KCl-ZrCl2 NaCl-KCl-ZrCl2-ZrCl3 NaCl-KCl-ZrCl3-ZrCl4 | Zr(IV)→Zr(III)→Zr(II)→Zr(0) | 670~740 |
| Sakakura[ | 1976 | CP | NaCl-KCl-ZrCl2 NaCl-KCl-ZrCl4 | Zr(IV)→Low Valency→Zr(0) | 700~900 |
| Polyakova[ | 1982 | LSV | NaCl-KCl-ZrCl4 | Zr(IV)→Zr(II)→Zr(0) | 735 |
| NaCl-KCl-K2ZrF6 | | 805 | |||
| Guang-Sen[ | 1990 | LSV and CV | NaCl-KCl-ZrCl4 | | 760 |
| Wu[ | 2011 | CV and SWV | NaCl-KCl-K2ZrF6 | Zr(IV)→Zr(II)→Zr(0) | 750 |
| Ueda[ | 2015 | CV | AlCl3-NaCl-KCl-ZrCl4 | Zr(IV)→Zr(II)→Zr(0) | 175 |
| Wang[ | 2016 | CV, CP and ED b | NaCl-KCl-K2ZrF6 | Pathway 1: Zr(IV)→Zr(III)→Zr(II)→Zr(0) Pathway 2: Zr(IV)→Zr(0) | 750 |
| Tekeda[ | 2018 | CV and SWV | NaCl-KCl-ZrCl4 | Pathway 1: Zr(IV)→Zr(II)→Zr(0) Pathway 2: Zr(IV)→Zr(0) | 800 |
| NaCl-KCl-K2ZrF6 | | ||||
| Zhang[ | 2022 | CV | NaCl-KCl-K2ZrF6 | Zr(IV)→Zr(II)→Zr(0) | 750 |
| Wang[ | 2023 | CV | NaCl-KCl-K2ZrF6 | Pathway 1: Zr(IV)→Zr(III)→Zr(II)→Zr(0) Pathway 2: Zr(IV)→Zr(0) | 750 |
| Author | Year | Technique/Method | Electrolyte | Electroreduction behavior of Zr(IV) | Temp.a/℃ |
|---|---|---|---|---|---|
| Mellors[ | 1966 | ED b | NaCl-KCl-ZrCl4 NaCl-KCl-K2ZrF6 | ZrCl2 forms in electroreduction | 600~900 |
| Swaroop[ | 1966 | EFM | NaCl-KCl-ZrCl2 NaCl-KCl-ZrCl2-ZrCl3 NaCl-KCl-ZrCl3-ZrCl4 | Zr(IV)→Zr(III)→Zr(II)→Zr(0) | 670~740 |
| Sakakura[ | 1976 | CP | NaCl-KCl-ZrCl2 NaCl-KCl-ZrCl4 | Zr(IV)→Low Valency→Zr(0) | 700~900 |
| Polyakova[ | 1982 | LSV | NaCl-KCl-ZrCl4 | Zr(IV)→Zr(II)→Zr(0) | 735 |
| NaCl-KCl-K2ZrF6 | | 805 | |||
| Guang-Sen[ | 1990 | LSV and CV | NaCl-KCl-ZrCl4 | | 760 |
| Wu[ | 2011 | CV and SWV | NaCl-KCl-K2ZrF6 | Zr(IV)→Zr(II)→Zr(0) | 750 |
| Ueda[ | 2015 | CV | AlCl3-NaCl-KCl-ZrCl4 | Zr(IV)→Zr(II)→Zr(0) | 175 |
| Wang[ | 2016 | CV, CP and ED b | NaCl-KCl-K2ZrF6 | Pathway 1: Zr(IV)→Zr(III)→Zr(II)→Zr(0) Pathway 2: Zr(IV)→Zr(0) | 750 |
| Tekeda[ | 2018 | CV and SWV | NaCl-KCl-ZrCl4 | Pathway 1: Zr(IV)→Zr(II)→Zr(0) Pathway 2: Zr(IV)→Zr(0) | 800 |
| NaCl-KCl-K2ZrF6 | | ||||
| Zhang[ | 2022 | CV | NaCl-KCl-K2ZrF6 | Zr(IV)→Zr(II)→Zr(0) | 750 |
| Wang[ | 2023 | CV | NaCl-KCl-K2ZrF6 | Pathway 1: Zr(IV)→Zr(III)→Zr(II)→Zr(0) Pathway 2: Zr(IV)→Zr(0) | 750 |

| [1] |
doi: 10.1007/BF02843474 |
| [2] |
|
|
(熊炳昆, 冶金工业出版社, 锆铪冶金, 北京, 2002, pp. 15-38.)
|
|
| [3] |
|
| [4] |
doi: 10.1016/0261-3069(85)90165-7 |
| [5] |
|
| [6] |
doi: 10.1016/j.jnucmat.2017.12.043 |
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
doi: 10.1179/cmq.1985.24.3.273 |
| [11] |
|
| [12] |
doi: 10.1016/j.jnucmat.2008.10.016 |
| [13] |
|
| [14] |
doi: 10.1016/j.nucengdes.2005.09.024 |
| [15] |
doi: 10.1039/D2EE02010F |
| [16] |
|
|
(王挺, 恽迪, 杜沛南, 伍晓勇, 材料导报, 2026, 40, 25040205.)
|
|
| [17] |
doi: 10.1007/s11669-013-0270-x |
| [18] |
doi: 10.1016/0022-3115(88)90127-4 |
| [19] |
doi: 10.1016/j.jnucmat.2022.153523 |
| [20] |
doi: 10.1007/BF02745341 |
| [21] |
doi: 10.1016/j.jnucmat.2009.03.007 |
| [22] |
doi: 10.1016/j.jnucmat.2012.03.045 |
| [23] |
doi: 10.1080/00295450.2017.1323535 |
| [24] |
doi: 10.13182/NT99-A2975 |
| [25] |
doi: 10.1016/j.pnucene.2021.104037 |
| [26] |
|
| [27] |
doi: 10.1080/00295450.2019.1623617 |
| [28] |
doi: 10.1007/BF02647233 |
| [29] |
doi: 10.1007/s10512-009-9092-7 |
| [30] |
doi: 10.1016/j.resconrec.2025.108708 |
| [31] |
|
| [32] |
doi: 10.1016/j.jnucmat.2015.07.010 |
| [33] |
|
| [34] |
|
| [35] |
doi: 10.1149/1945-7111/ac0996 |
| [36] |
|
| [37] |
|
|
(刘辉, 王化军, 许文, 徐国栋, 刘璐, 王俊莲, 中国材料进展, 2023, 42, 335.)
|
|
| [38] |
|
| [39] |
|
| [40] |
doi: 10.1021/cr60005a001 |
| [41] |
|
| [42] |
doi: 10.1002/qua.v111.3 |
| [43] |
doi: 10.1002/zaac.v255:1/3 |
| [44] |
|
| [45] |
doi: 10.1007/BF02345549 |
| [46] |
doi: 10.1021/ja01589a010 |
| [47] |
doi: 10.1080/15422119.2014.920876 |
| [48] |
doi: 10.1016/j.seppur.2021.119779 |
| [49] |
doi: 10.1016/j.seppur.2024.128539 |
| [50] |
doi: 10.1016/j.cej.2013.03.127 |
| [51] |
doi: 10.1021/acsami.5c02082 |
| [52] |
doi: 10.1016/j.seppur.2025.132620 |
| [53] |
|
| [54] |
doi: 10.3390/en80910354 |
| [55] |
doi: 10.1134/S003602952470160X |
| [56] |
|
| [57] |
doi: 10.1515/HTMP.1999.18.4.213 |
| [58] |
doi: 10.1021/je00020a019 |
| [59] |
|
| [60] |
doi: 10.1021/je800299y |
| [61] |
doi: 10.1080/01496395.2016.1170852 |
| [62] |
|
| [63] |
doi: 10.1007/BF02652514 |
| [64] |
|
| [65] |
|
| [66] |
|
| [67] |
|
| [68] |
doi: 10.1007/BF01012474 |
| [69] |
doi: 10.1002/zaac.v148:1 |
| [70] |
doi: 10.1149/1.3071683 |
| [71] |
doi: 10.1016/0016-0032(55)90727-4 |
| [72] |
doi: 10.1149/1.2773819 |
| [73] |
doi: 10.1007/BF03398172 |
| [74] |
|
| [75] |
|
|
(吴延科, 陈松, 徐志高, 王力军, 稀有金属, 2009, 33, 62.)
|
|
| [76] |
|
| [77] |
doi: 10.1149/1.2423865 |
| [78] |
doi: 10.1149/1.2781210 |
| [79] |
doi: 10.1149/1.2430009 |
| [80] |
doi: 10.1016/0013-4686(82)80092-3 |
| [81] |
doi: 10.1007/s12598-011-0187-7 |
| [82] |
|
| [83] |
doi: 10.1016/j.ijhydene.2021.12.038 |
| [84] |
doi: 10.1007/BF02642064 |
| [85] |
doi: 10.1149/1.2114020 |
| [86] |
doi: 10.1016/j.jmst.2019.10.043 |
| [87] |
doi: 10.1016/j.electacta.2018.01.213 |
| [88] |
doi: 10.1007/s10008-015-2861-4 |
| [89] |
doi: 10.1149/1945-7111/ad02c3 |
| [90] |
|
| [91] |
|
| [92] |
doi: 10.1007/s11663-004-0024-6 |
| [93] |
doi: 10.20964/2018.05.23 |
| [94] |
|
| [95] |
|
|
(李晴宇, 杜继红, 奚正平, 稀有金属材料与工程, 2007, 36, 390.)
|
|
| [96] |
doi: 10.1021/cr200305x pmid: 23339568 |
| [97] |
doi: 10.1016/S0364-5916(02)00027-5 |
| [98] |
doi: 10.1179/026708304225012242 |
| [99] |
doi: 10.1007/s11663-006-9016-z |
| [100] |
doi: 10.1021/cm802237v |
| [101] |
doi: 10.1021/cm902073g |
| [102] |
|
| [103] |
doi: 10.1007/s10800-010-0242-3 |
| [104] |
|
| [105] |
doi: 10.1134/S0031918X1413016X |
| [106] |
doi: 10.1149/07515.0391ecst |
| [107] |
doi: 10.1149/1945-7111/acf8fc |
| [108] |
doi: 10.3390/met13020408 |
| [109] |
doi: 10.1557/jmr.2006.0268 |
| [110] |
|
| [111] |
doi: 10.1007/s40831-018-0199-8 |
| [112] |
doi: 10.1007/s11663-021-02255-x |
| [113] |
doi: 10.1016/j.molliq.2024.126831 |
| [114] |
doi: 10.1016/j.seppur.2021.118803 |
| [115] |
doi: 10.3390/ma18112634 |
| [116] |
|
| [117] |
doi: 10.1016/j.seppur.2020.118096 |
| [118] |
doi: 10.1016/j.proche.2012.10.013 |
| [119] |
doi: 10.1016/j.pnucene.2023.105026 |
| [120] |
|
| [121] |
|
| [122] |
|
| [123] |
doi: 10.1080/00295450.2020.1757961 |
| [124] |
doi: 10.1016/j.jnucmat.2023.154339 |
| [125] |
|
| [126] |
doi: 10.1016/j.radphyschem.2022.110732 |
| [127] |
doi: 10.1016/j.nucengdes.2014.04.035 |
| [128] |
doi: 10.1002/er.v45.8 |
| [129] |
|
| [130] |
|
| [131] |
doi: 10.1016/0022-3115(92)90515-M |
| [132] |
doi: 10.1007/s11426-016-0321-x |
| [133] |
|
| [134] |
doi: 10.1016/j.electacta.2013.02.022 |
| [135] |
doi: 10.1007/s11595-023-2804-3 |
| [136] |
doi: 10.1016/j.jnucmat.2013.01.310 |
| [137] |
doi: 10.1016/j.jnucmat.2020.152219 |
| [138] |
doi: 10.1021/jp411781n |
| [139] |
doi: 10.1149/1945-7111/ac4b1a |
| [140] |
doi: 10.1149/1945-7111/add41f |
| [141] |
|
| [142] |
doi: 10.1016/j.jnucmat.2016.04.037 |
| [143] |
|
| [144] |
doi: 10.1149/1.2423405 |
| [145] |
doi: 10.5796/kogyobutsurikagaku.39.864 |
| [146] |
|
| [147] |
doi: 10.13182/NT01-A3162 |
| [148] |
doi: 10.1016/j.anucene.2021.108924 |
| [149] |
doi: 10.1016/j.jpcs.2004.06.049 |
| [150] |
doi: 10.1524/zpch.2006.220.10.1247 |
| [151] |
|
| [152] |
doi: 10.1039/C6RA17102H |
| [153] |
|
| [154] |
doi: 10.1016/j.jfluchem.2011.06.040 |
| [155] |
doi: 10.1016/j.jnucmat.2017.03.028 |
| [156] |
|
| [157] |
doi: 10.1007/BF00616685 |
| [158] |
|
| [159] |
|
| [160] |
doi: 10.1016/j.jallcom.2007.05.008 |
| [161] |
doi: 10.1016/j.jallcom.2010.10.048 |
| [162] |
doi: 10.1016/j.seppur.2019.115965 |
| [163] |
doi: 10.1021/acssuschemeng.1c06931 |
| [164] |
doi: 10.1016/j.seppur.2021.119683 |
| [165] |
|
| [166] |
doi: 10.1016/j.jelechem.2008.12.011 |
| [167] |
|
| [168] |
doi: 10.1016/j.electacta.2015.02.078 |
| [169] |
doi: 10.1016/j.jnucmat.2021.153330 |
| [170] |
|
|
(肖益群, 王有群, 林如山, 贾艳虹, 何辉, 核化学与放射化学, 2018, 40, 100.)
doi: 10.7538/hhx.2018.YX.2017034 |
|
| [171] |
|
| [172] |
doi: 10.1016/j.elecom.2017.10.010 |
| [173] |
doi: 10.1149/1945-7111/ab69f3 |
| [174] |
doi: 10.1016/0013-4686(62)80035-8 |
| [175] |
doi: 10.1139/v65-410 |
| [176] |
doi: 10.1139/v66-031 |
| [177] |
doi: 10.5796/jesj.35.2.75 |
| [178] |
doi: 10.1149/06404.0609ecst |
| [179] |
|
| [180] |
|
| [181] |
doi: 10.1007/BF00615261 |
| [182] |
doi: 10.1021/j100630a014 |
| [183] |
|
| [184] |
doi: 10.1016/j.molliq.2025.127254 |
| [185] |
doi: 10.3103/S1067821209050101 |
| [186] |
|
| [1] | 罗雅玲, 庄展洋, 范峰滔, 李灿. Al掺杂SrTiO3的合成调控及其光催化全分解水性能[J]. 化学学报, 2025, 83(6): 608-615. |
| [2] | 韩逸之, 蓝建慧, 刘学, 石伟群. 基于机器学习势函数的熔盐体系分子动力学研究进展[J]. 化学学报, 2023, 81(11): 1663-1672. |
| [3] | 樊小勇, 张帅, 朱永强, 敬茂森, 王凯鑫, 张露露, 李巨龙, 许磊, 苟蕾, 李东林. 三维多孔铜和锌镀层协同构筑无枝晶锂金属电极[J]. 化学学报, 2022, 80(4): 517-525. |
| [4] | 刘毅川, 刘雅兰, 姜仕林, 李梅, 石伟群. 氯化物熔盐体系中铀的化学种态研究进展[J]. 化学学报, 2021, 79(12): 1425-1437. |
| [5] | 林伟芬, 陈念嘉, 游乐星, 周顺桂. 希瓦氏菌MR-1影响玻碳表面电沉积Cd的机理[J]. 化学学报, 2018, 76(7): 543-548. |
| [6] | 陶熊新, 李莉, 齐学强, 魏子栋. Pt-Ni-P/Ti蜂窝状电极的制备及其催化甲醇氧化性能研究[J]. 化学学报, 2017, 75(2): 237-240. |
| [7] | 冷明浩, 陈仕谋, 张军玲, 郎海燕, 康艳红, 张锁江. 含羰基有机添加剂对AlCl3-[Emim]Cl电沉积铝的影响[J]. 化学学报, 2015, 73(5): 403-408. |
| [8] | 范存华, 杨逸群, 赵伟, 肖宇, 罗静, 刘晓亚. 双亲聚合物分子印迹自组装胶束电化学传感器研究[J]. 化学学报, 2013, 71(06): 934-940. |
| [9] | 寇东星, 刘伟庆, 胡林华, 陈双宏, 黄阳, 戴松元. 电沉积表面修饰对染料敏化太阳电池微观性能影响机理研究[J]. 化学学报, 2013, 71(05): 777-781. |
| [10] | 史国玉, 王宗花, 夏建飞, 张菲菲, 夏延致, 李延辉. 电化学合成PtCo/石墨烯复合催化剂及对甲醇的电催化氧化[J]. 化学学报, 2013, 71(02): 227-233. |
| [11] | 林青含, 邱丽琴, 程璇, 周健. 电沉积Bi2Te3 基薄膜的电化学阻抗谱研究[J]. 化学学报, 2012, 70(10): 1173-1178. |
| [12] | 左晓希, 李奇, 刘建生, 肖信, 范成杰, 南俊民. LiPF6/三氟乙酰胺室温熔盐的制备及在碳-碳电容器中的性能[J]. 化学学报, 2012, 70(04): 367-371. |
| [13] | 郑精武, 周杰, 郑飚, 乔梁, 姜力强, 张诚. 三乙醇胺对羟基亚乙基二膦酸镀铜液的影响研究[J]. 化学学报, 2011, 69(24): 2921-2928. |
| [14] | 周蓉, 张红梅, 杜玉扣, 杨平. 电沉积Pt-Au双金属催化剂及其对甲酸的电催化研究[J]. 化学学报, 2011, 69(13): 1533-1539. |
| [15] | 张红梅, 周卫强, 杜玉扣, 杨平, 徐景坤. 电化学合成Pt-Au复合催化剂及其对甲醇的电催化氧化[J]. 化学学报, 2010, 68(24): 2529-2534. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||