化学学报 ›› 2026, Vol. 84 ›› Issue (4): 490-497.DOI: 10.6023/A25120391 上一篇 下一篇
研究论文
雷晨a, 赵鑫a, 董婧a, 林文军a, 张蕊a, 王传军a,*( ), 王国强b,*(
), 王国强b,*( )
)
投稿日期:2025-12-02
发布日期:2026-02-14
通讯作者:
王传军, 王国强
基金资助:
Chen Leia, Xin Zhaoa, Jing Donga, Wenjun Lina, Rui Zhanga, Chuanjun Wanga,*( ), Guoqiang Wangb,*(
), Guoqiang Wangb,*( )
)
Received:2025-12-02
Published:2026-02-14
Contact:
Chuanjun Wang, Guoqiang Wang
Supported by:文章分享
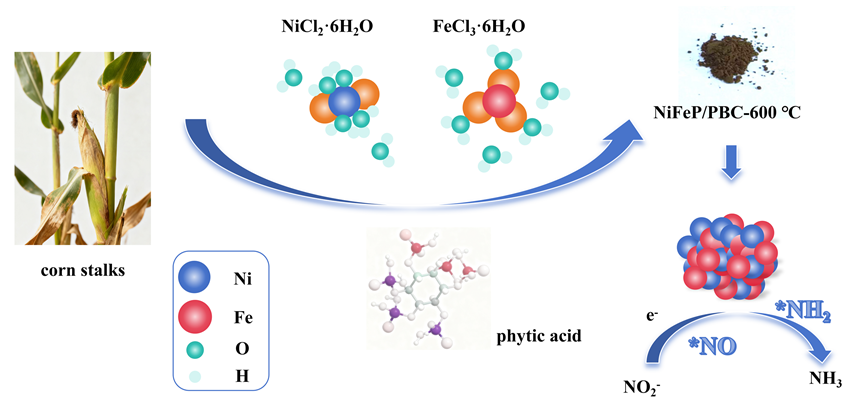
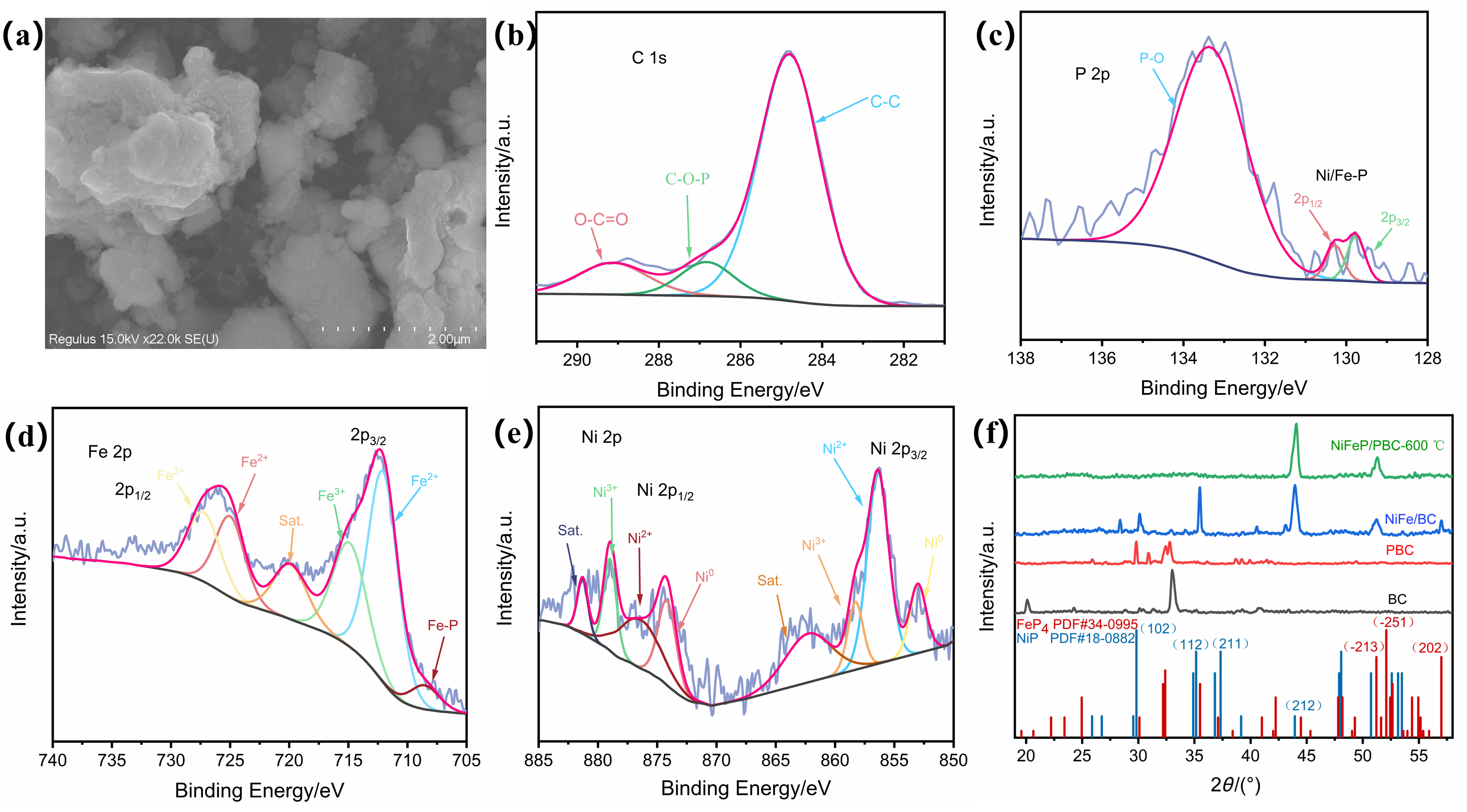
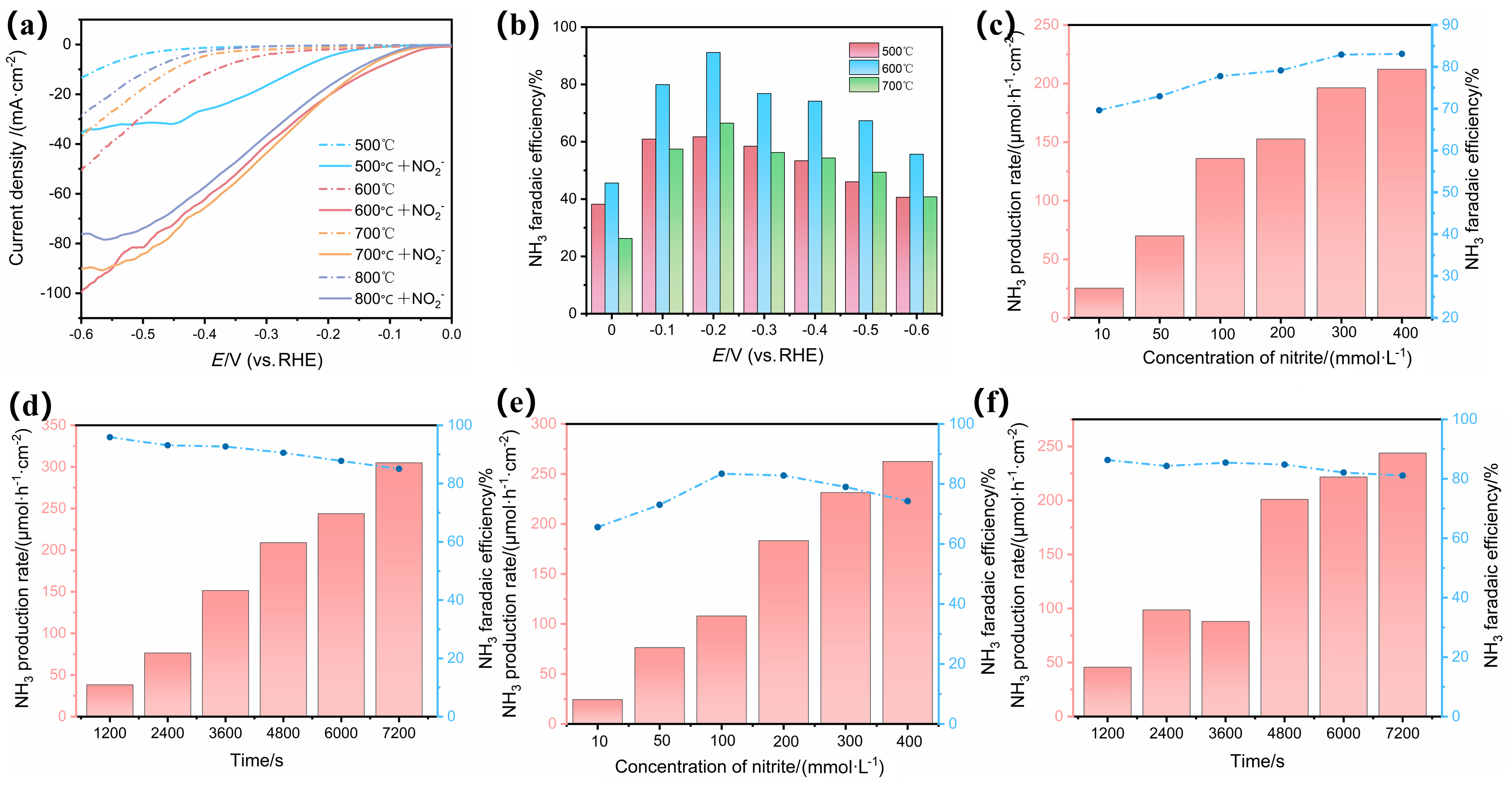
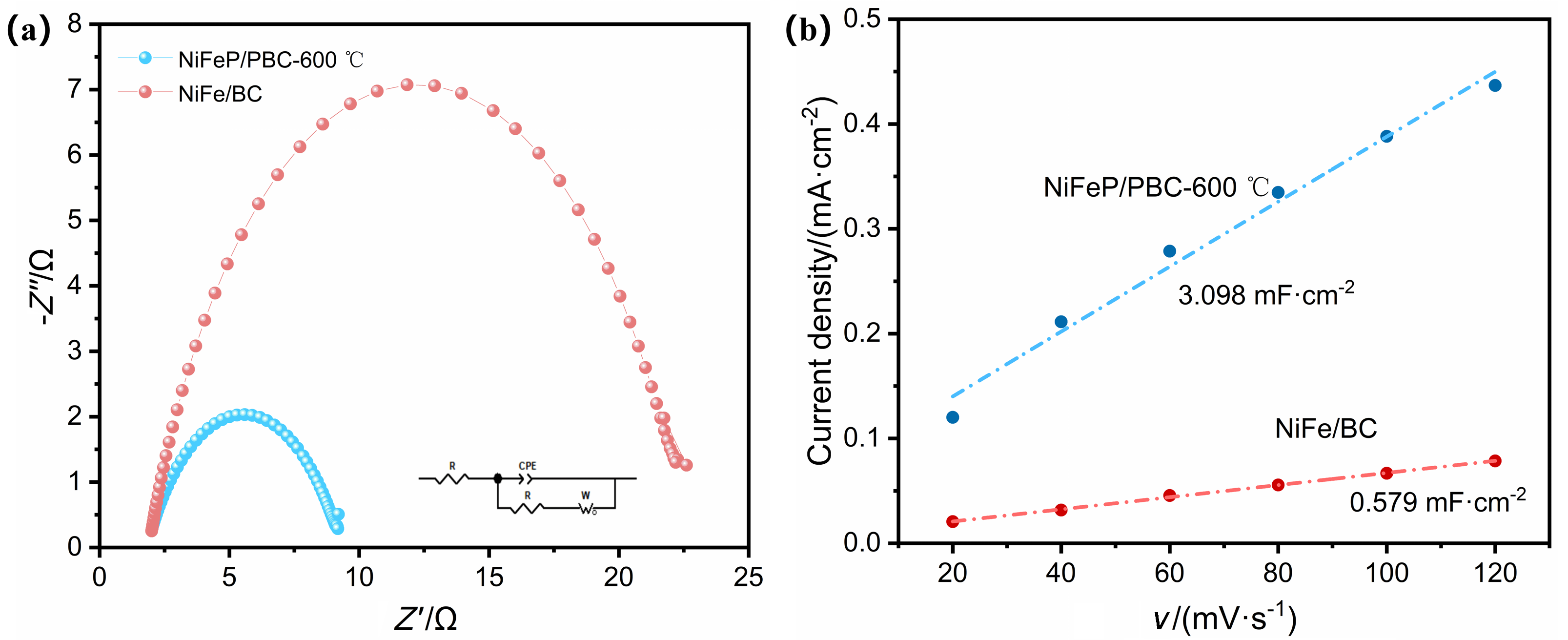
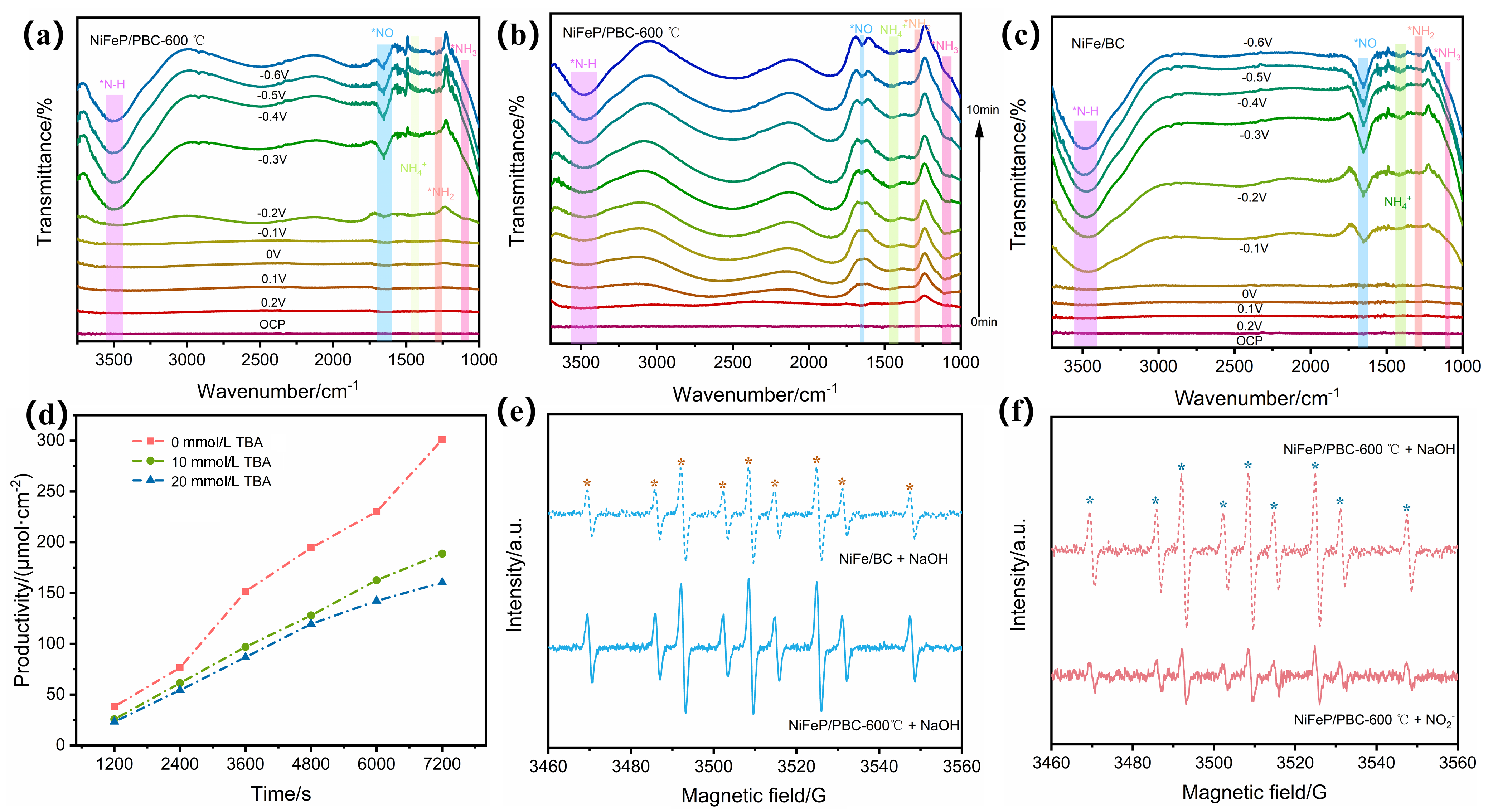
氨在农业、工业生产上有着重要的用途, 传统的哈伯法将氢气和氮气直接转化为氨, 消耗大量能源的同时对环境造成严重污染. 而电催化具有反应条件温和、选择性高、绿色环保等优势成为研究的热点. 其中开发有效选择性合成氨的廉价催化剂是关键. 本研究以废弃玉米秸秆为炭源, 植酸为磷源, 采用一步煅烧法成功制备出镍铁双金属磷化物负载的生物质炭催化剂(NiFeP/PBC), 表现出优异的电催化亚硝酸盐还原(eNO2RR)合成氨性能, 在-0.2 V (vs. RHE) 电压下法拉第效率高达95.9%, 氨产率达到304.9 μmol•h−1•cm−2, 并具备良好的稳定性. 电化学原位红外研究表明, 催化剂在反应过程中生成NO、NH2和NH3等关键中间体, 且活性氢(*H)在还原过程中起重要作用. 该工作为废弃生物质的高值化利用和绿色电合成氨提供了新策略, 具有显著的环境和能源意义.
雷晨, 赵鑫, 董婧, 林文军, 张蕊, 王传军, 王国强. 生物质衍生炭载镍铁磷化物催化剂用于含氮污染物电催化合成氨[J]. 化学学报, 2026, 84(4): 490-497.
Chen Lei, Xin Zhao, Jing Dong, Wenjun Lin, Rui Zhang, Chuanjun Wang, Guoqiang Wang. Biomass-derived Carbon-supported Nickel-iron Phosphide Catalyst for Electrocatalytic Synthesis of Ammonia from Nitrogen-containing Pollutants[J]. Acta Chimica Sinica, 2026, 84(4): 490-497.
| 催化剂 | 电解质 | 氨产率 | 法拉第效率/% | 电压a | 参考文献 |
|---|---|---|---|---|---|
| Cu@Cu2O MSs | 0.1 mol•L−1 Na2SO4 | 0.3 mol•h−1•g−1 | 80.6 | -1.10 V | [ |
| Cu/Ti3C2 | 0.1 mol•L−1 KOH | 3.0 μmol•h−1•cm−2 | 7.3 | -0.50 V | [ |
| NiFe-LDH | 0.25 mol•L−1 Li2SO4 | 0.1 mmol•h−1•cm−2 | 82 | -0.70 V | [ |
| Cu10Fe1 纳米合金 | 0.5 mol•L−1 Na2SO4 | 0.2 mmol•h−1•cm−2 | 93.7 | -0.35 V | [ |
| CoP | 0.2 mol•L−1 Na2SO4 | 47.2 μmol•h−1•cm−2 | 88.3 | -0.20 V | [ |
| CoS1-x | 0.2 mol•L−1 Na2SO4 | 44.7 μmol•h−1•cm−2 | 53.6 | -0.40 V | [ |
| NiCoP | 0.1 mol•L−1 Na2SO4 | 5553.4 mg•h−1•cm−2 | 91.3 | -1.20 V | [ |
| 竹笋NC-800 | 0.1 mol•L−1 HCl | 16.28 μg•h−1•mg−1 | 27.5 | -0.35 V | [ |
| 木棉纤维O-KFCNTs | 0.1 mol•L−1 HCl | 25.12 μg•h−1•mg−1 | 9.1 | -0.80 V | [ |
| NiFeP/PBC-600 ℃ | 0.1 mol•L−1 NaNO2 | 304.9 μmol•h−1•cm−2 | 95.9 | -0.20 V | 本工作 |
| 催化剂 | 电解质 | 氨产率 | 法拉第效率/% | 电压a | 参考文献 |
|---|---|---|---|---|---|
| Cu@Cu2O MSs | 0.1 mol•L−1 Na2SO4 | 0.3 mol•h−1•g−1 | 80.6 | -1.10 V | [ |
| Cu/Ti3C2 | 0.1 mol•L−1 KOH | 3.0 μmol•h−1•cm−2 | 7.3 | -0.50 V | [ |
| NiFe-LDH | 0.25 mol•L−1 Li2SO4 | 0.1 mmol•h−1•cm−2 | 82 | -0.70 V | [ |
| Cu10Fe1 纳米合金 | 0.5 mol•L−1 Na2SO4 | 0.2 mmol•h−1•cm−2 | 93.7 | -0.35 V | [ |
| CoP | 0.2 mol•L−1 Na2SO4 | 47.2 μmol•h−1•cm−2 | 88.3 | -0.20 V | [ |
| CoS1-x | 0.2 mol•L−1 Na2SO4 | 44.7 μmol•h−1•cm−2 | 53.6 | -0.40 V | [ |
| NiCoP | 0.1 mol•L−1 Na2SO4 | 5553.4 mg•h−1•cm−2 | 91.3 | -1.20 V | [ |
| 竹笋NC-800 | 0.1 mol•L−1 HCl | 16.28 μg•h−1•mg−1 | 27.5 | -0.35 V | [ |
| 木棉纤维O-KFCNTs | 0.1 mol•L−1 HCl | 25.12 μg•h−1•mg−1 | 9.1 | -0.80 V | [ |
| NiFeP/PBC-600 ℃ | 0.1 mol•L−1 NaNO2 | 304.9 μmol•h−1•cm−2 | 95.9 | -0.20 V | 本工作 |
| [1] |
doi: 10.1021/acs.chemrev.9b00538 pmid: 32501681 |
| [2] |
|
| [3] |
doi: 10.1016/j.jece.2024.112892 |
| [4] |
|
| [5] |
doi: 10.1016/j.nanoen.2024.109499 |
| [6] |
doi: 10.1007/s10311-025-01848-1 |
| [7] |
|
| [8] |
doi: 10.6023/A25030091 |
|
(郭超凡, 苏进展, 郭烈锦, 化学学报, 2025, 83, 716.)
doi: 10.6023/A25030091 |
|
| [9] |
|
| [10] |
doi: 10.1016/j.jechem.2024.10.054 |
| [11] |
doi: 10.1016/j.apenergy.2017.01.071 |
| [12] |
doi: 10.1007/s10311-022-01402-3 |
| [13] |
doi: 10.1021/acs.energyfuels.9b02924 |
| [14] |
|
| [15] |
doi: 10.1016/j.matchemphys.2024.129165 |
| [16] |
|
| [17] |
doi: 10.1016/j.jpba.2022.115102 |
| [18] |
doi: 10.1007/s12649-020-01109-y |
| [19] |
doi: 10.1021/acsomega.0c02673 |
| [20] |
doi: 10.1186/s40643-023-00671-2 |
| [21] |
|
| [22] |
|
| [23] |
doi: 10.1002/cjoc.v42.24 |
| [24] |
|
| [25] |
doi: 10.1016/j.nanoen.2023.108415 |
| [26] |
doi: 10.1002/adfm.v32.35 |
| [27] |
doi: 10.1016/j.jeurceramsoc.2023.09.086 |
| [28] |
doi: 10.1007/s12598-020-01513-2 |
| [29] |
|
| [30] |
doi: 10.1002/cplu.v86.1 |
| [31] |
doi: 10.1039/D2CC02463B |
| [32] |
doi: 10.1016/j.nanoen.2023.108840 |
| [33] |
doi: 10.1039/D2QI00002D |
| [34] |
doi: 10.1021/acs.inorgchem.2c01112 |
| [35] |
doi: 10.1039/D5RA00391A |
| [36] |
doi: 10.3390/catal10030353 |
| [37] |
doi: 10.1039/C9CC90001B |
| [1] | 甘芮弋, 罗俊淋, 陈瑞, 陈颖, 杨梦, 韦佳雪, 李思雨, 李凯航, 王磊, 杨娜. 活性污泥基生物质炭在锂硫电池正极材料中的应用及性能研究★[J]. 化学学报, 2025, 83(8): 827-832. |
| [2] | 刘珊珊, 董微微, 李珍珍, 张瑶瑶, 李超, 焦林郁. 离子液体调控的串联脱氢环化反应多样性合成生物质基氮杂环[J]. 化学学报, 2025, 83(5): 479-487. |
| [3] | 郭建荣, 张书玉, 贺军辉, 任世学. 基于生物质可降解薄膜的制备与应用[J]. 化学学报, 2024, 82(2): 242-256. |
| [4] | 鞠嘉浩, 徐吉磊, 王康军, 黄家辉. 果糖一步法制备5-甲氧基甲基-2-呋喃甲醛及树脂催化剂再生方法探究[J]. 化学学报, 2024, 82(12): 1216-1225. |
| [5] | 刘露杰, 张建, 王亮, 肖丰收. 生物质基多元醇的多相催化选择性氢解★[J]. 化学学报, 2023, 81(5): 533-547. |
| [6] | 于璐瑶, 任祯, 杨宇森, 卫敏. 生物基聚酯单体的定向催化制备[J]. 化学学报, 2023, 81(2): 175-190. |
| [7] | 田钊炜, 达伟民, 王雷, 杨宇森, 卫敏. 第二代生物柴油制备的多相催化剂的结构设计及研究进展[J]. 化学学报, 2022, 80(9): 1322-1337. |
| [8] | 舒恒, 包义德日根, 那永. CdS基纳米管光催化氧化5-羟甲基糠醛选择性生成2,5-呋喃二甲醛[J]. 化学学报, 2022, 80(5): 607-613. |
| [9] | 王文彬, 温群磊, 刘友文, 翟天佑. 表界面化学调控二维材料电催化生物质转化的研究进展[J]. 化学学报, 2020, 78(11): 1185-1199. |
| [10] | 李翠, 张琪, 傅尧. 过渡金属催化的醇类脱氧脱水反应[J]. 化学学报, 2018, 76(7): 501-514. |
| [11] | 赵婧, 龚俊伟, 李一举, 程魁, 叶克, 朱凯, 闫俊, 曹殿学, 王贵领. 自掺杂氮多孔交联碳纳米片在超级电容器中的应用[J]. 化学学报, 2018, 76(2): 107-112. |
| [12] | 王玉珏, 胡敏, 王渝, 秦艳红, 陈红阳, 曾立民, 雷建容, 黄晓锋, 何凌燕, 张瑞芹, 吴志军. 秸秆燃烧排放PM2.5特征及影响因素研究[J]. 化学学报, 2016, 74(4): 356-362. |
| [13] | 李江, 黄耀兵, 郭庆祥, 傅尧. 水热条件下酸催化氧化木质纤维生物质制取乙酸[J]. 化学学报, 2014, 72(12): 1223-1227. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||