Acta Chimica Sinica ›› 2026, Vol. 84 ›› Issue (3): 275-279.DOI: 10.6023/A26010002 Previous Articles Next Articles
Communication
王翔宇a, 夏天骐a, 冷雪冰a, 王东阳a, 邓亮a,b,*( )
)
投稿日期:2026-01-05
发布日期:2026-02-10
基金资助:
Wang Xiangyua, Xia Tianqia, Leng Xuebinga, Wang Dongyanga, Deng Lianga,b,*( )
)
Received:2026-01-05
Published:2026-02-10
Contact:
* E-mail: deng@sioc.ac.cn
Supported by:Share
Wang Xiangyu, Xia Tianqi, Leng Xuebing, Wang Dongyang, Deng Liang. Mononuclear Palladium(I) Halides Supported by tert-Butyl-Substituted Xantphos Ligand[J]. Acta Chimica Sinica, 2026, 84(3): 275-279.
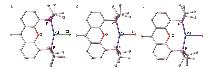
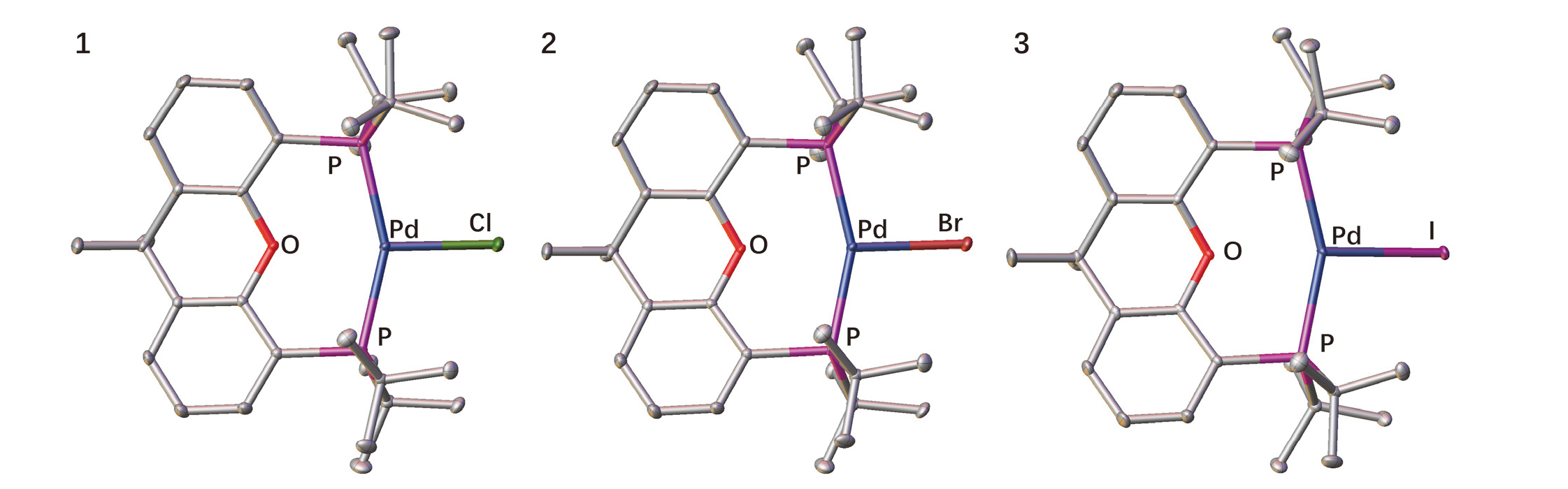
| 键长/nm与键角/(°) | Pd—X | Pd···O | Pd—P | P—Pd—P | P—Pd—X |
|---|---|---|---|---|---|
| [(tBuXantphos)PdCl] (1) | 0.2469(2) | 0.2472(2) | 0.2326(1) 0.2337(1) | 153.5(1) | 101.6(1) 104.6(1) |
| [(tBuXantphos)PdCl] (1') a | 0.24717(7) | 0.24803(18) | 0.23115(8) 0.23098(7) | 153.22(2) | 101.61(2) 104.75(2) |
| [(dtbpf)PdCl] b | 0.2360(2) | 0.2319(2) 0.2315(2) | 107.1(1) | 118.4(1) 134.5(1) | |
| [(tBuXantphos)PdBr] (2) | 0.2591(1) | 0.2465(2) | 0.2328(1) 0.2340(1) | 153.7(1) | 101.3(1) 104.7(1) |
| [(tBuXantphos)PdBr] (2') a | 0.25813(3) | 0.2516 | 0.23256(8) 0.23256(8) | 152.81(2) | 101.853(17) 105.288(17) |
| [(dtbpf)PdBr] b | 0.2466(1) | 0.2310(2) 0.2319(2) | 107.5(1) | 124.2(1) 128.3(1) | |
| [(tBuXantphos)PdI] (3) | 0.2739(1) | 0.2468(2) | 0.2333(1) 0.2346(1) | 153.4(1) | 101.6(1) 104.6(1) |
| [(dtbpf)PdI] b | 0.2608(2) | 0.2318(2) 0.2312(2) | 108.0(1) | 123.8(1) 128.2(1) |
| 键长/nm与键角/(°) | Pd—X | Pd···O | Pd—P | P—Pd—P | P—Pd—X |
|---|---|---|---|---|---|
| [(tBuXantphos)PdCl] (1) | 0.2469(2) | 0.2472(2) | 0.2326(1) 0.2337(1) | 153.5(1) | 101.6(1) 104.6(1) |
| [(tBuXantphos)PdCl] (1') a | 0.24717(7) | 0.24803(18) | 0.23115(8) 0.23098(7) | 153.22(2) | 101.61(2) 104.75(2) |
| [(dtbpf)PdCl] b | 0.2360(2) | 0.2319(2) 0.2315(2) | 107.1(1) | 118.4(1) 134.5(1) | |
| [(tBuXantphos)PdBr] (2) | 0.2591(1) | 0.2465(2) | 0.2328(1) 0.2340(1) | 153.7(1) | 101.3(1) 104.7(1) |
| [(tBuXantphos)PdBr] (2') a | 0.25813(3) | 0.2516 | 0.23256(8) 0.23256(8) | 152.81(2) | 101.853(17) 105.288(17) |
| [(dtbpf)PdBr] b | 0.2466(1) | 0.2310(2) 0.2319(2) | 107.5(1) | 124.2(1) 128.3(1) | |
| [(tBuXantphos)PdI] (3) | 0.2739(1) | 0.2468(2) | 0.2333(1) 0.2346(1) | 153.4(1) | 101.6(1) 104.6(1) |
| [(dtbpf)PdI] b | 0.2608(2) | 0.2318(2) 0.2312(2) | 108.0(1) | 123.8(1) 128.2(1) |
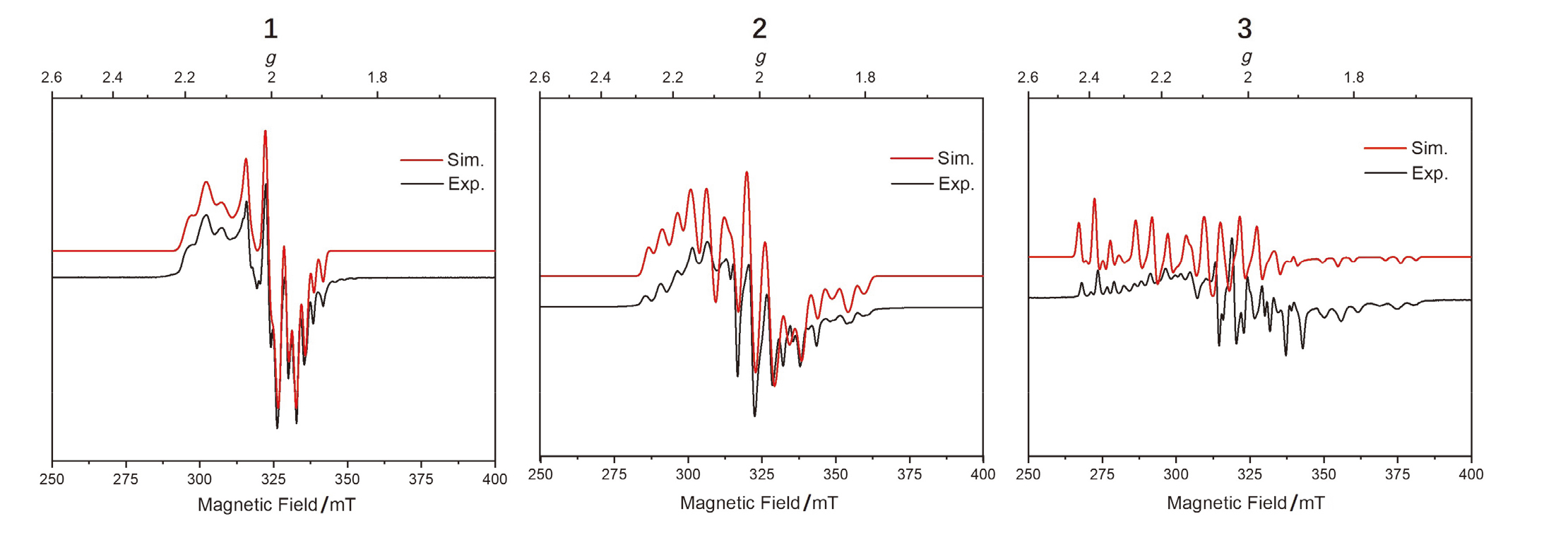
| g因子与超精细耦合常数/MHz | g | A(31P) | A(X) |
|---|---|---|---|
| [(tBuXantphos)PdCl] (1) | [2.231, 2.079, 2.034] | [151, 188, 153] [180, 189, 177] | [33, 27, 90] |
| [(tBuXantphos)PdCl] (1') a | [2.241, 2.079, 2.037] | [168, 135, 177] [169, 134, 181] | [50, 37, 87] |
| [(dtbpf)PdCl] b | [2.167, 2.045, 2.020] | [663, 324, 318] [531, 508, 311] | [<10, <10, <13] |
| [(tBuXantphos)PdBr] (2) | [2.195, 2.081, 2.035] | [164, 144, 140] [109, 166, 165] | [312, 75, 425] |
| [(tBuXantphos)PdBr] (2') a | [2.206, 2.081, 2.038] | [160, 140, 150] [126, 160, 162] | [328, 68, 398] |
| [(dtbpf)PdBr] b | [2.178, 2.051, 2.035] | [575, 446, 278] [513, 413, 394] | [112, 47, 37] |
| [(tBuXantphos)PdI] (3) | [2.201, 2.129, 2.074] | [180, 150, 150] [180, 150, 150] | [360, 150, 600] |
| [(dtbpf)PdI] b | [2.201, 2.060, 2.050] | [553, 445, 262] [508, 437, 315] | [150, 140, 28] |
| g因子与超精细耦合常数/MHz | g | A(31P) | A(X) |
|---|---|---|---|
| [(tBuXantphos)PdCl] (1) | [2.231, 2.079, 2.034] | [151, 188, 153] [180, 189, 177] | [33, 27, 90] |
| [(tBuXantphos)PdCl] (1') a | [2.241, 2.079, 2.037] | [168, 135, 177] [169, 134, 181] | [50, 37, 87] |
| [(dtbpf)PdCl] b | [2.167, 2.045, 2.020] | [663, 324, 318] [531, 508, 311] | [<10, <10, <13] |
| [(tBuXantphos)PdBr] (2) | [2.195, 2.081, 2.035] | [164, 144, 140] [109, 166, 165] | [312, 75, 425] |
| [(tBuXantphos)PdBr] (2') a | [2.206, 2.081, 2.038] | [160, 140, 150] [126, 160, 162] | [328, 68, 398] |
| [(dtbpf)PdBr] b | [2.178, 2.051, 2.035] | [575, 446, 278] [513, 413, 394] | [112, 47, 37] |
| [(tBuXantphos)PdI] (3) | [2.201, 2.129, 2.074] | [180, 150, 150] [180, 150, 150] | [360, 150, 600] |
| [(dtbpf)PdI] b | [2.201, 2.060, 2.050] | [553, 445, 262] [508, 437, 315] | [150, 140, 28] |
| [1] |
doi: 10.1021/acs.organomet.5b00567 |
| [2] |
doi: 10.1021/acs.organomet.9b00110 |
| [3] |
doi: 10.6023/A23070336 |
|
(韩叶强, 史炳锋, 化学学报, 2023, 81, 1522.)
doi: 10.6023/A23070336 |
|
| [4] |
doi: 10.6023/A23100472 |
|
(高炜洋, 邓伟超, 高扬, 梁仁校, 贾义霞, 化学学报, 2024, 82, 1.)
doi: 10.6023/A23100472 |
|
| [5] |
doi: 10.6023/A21120597 |
|
(葛懿修, 邱早早, 谢作伟, 化学学报, 2022, 80, 432.)
doi: 10.6023/A21120597 |
|
| [6] |
doi: 10.6023/A23080395 |
|
(张大伟, 赵海洋, 冯笑甜, 顾玉诚, 张新刚, 化学学报, 2024, 82, 105.)
doi: 10.6023/A23080395 |
|
| [7] |
doi: 10.1002/cjoc.v41.15 |
| [8] |
doi: 10.1021/jacs.6b01628 pmid: 27149524 |
| [9] |
doi: 10.1126/science.aba5901 |
| [10] |
doi: 10.1021/acscatal.4c06913 pmid: 39839849 |
| [11] |
doi: 10.1021/ja00832a048 |
| [12] |
doi: 10.1016/S0040-4020(01)96431-2 |
| [13] |
|
| [14] |
doi: 10.1021/jacs.5c04407 pmid: 40440468 |
| [15] |
doi: 10.1002/anie.v55.11 |
| [16] |
doi: 10.1016/j.chempr.2016.11.007 |
| [17] |
doi: 10.1021/jacs.2c08765 |
| [18] |
doi: 10.1021/jacs.3c04167 |
| [19] |
doi: 10.1021/acs.inorgchem.3c02878 |
| [20] |
doi: 10.1021/jacs.1c04965 |
| [21] |
doi: 10.31635/ccschem.024.202404464 |
| [22] |
|
| [23] |
doi: 10.1002/anie.v60.7 |
| [24] |
doi: 10.1021/ja0731571 |
| [25] |
doi: 10.1021/jacs.5c17683 |
| [26] |
doi: 10.1021/jacs.5c14709 |
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [1] | Helin Wang, Qingle Zeng. Cascade Addition and Cyclization of Indoloalkynes with Phosphine Oxides for the Synthesis of Diarylphosphinoyl Indolo[1,2-a]quinolines [J]. Acta Chimica Sinica, 2026, 84(2): 189-195. |
| [2] | Qi Gao, Lirong Chen, Jinyi Qian, Ruifeng Fan, Weiqing Sun, Yafei Guo, Baomin Fan. Asymmetric Hydrophosphination of Diarylphosphine Oxides to α,β-Unsaturated Bifunctional Compounds Catalyzed by Chiral Oxazaborolidine [J]. Acta Chimica Sinica, 2024, 82(7): 742-747. |
| [3] | Hangqing Lin, Ruoru Ma, Yilan Jiang, Murong Xu, Yangpeng Lin, Kezhao Du. Research Progress of Materials Used for Elemental Halogen Capture [J]. Acta Chimica Sinica, 2024, 82(1): 62-74. |
| [4] | Yandong Zhang, Shoufei Zhu. Perspective for Phosphine Ligands with Cyclopropane Backbone★ [J]. Acta Chimica Sinica, 2023, 81(7): 777-783. |
| [5] | Jie Yang, Lin Ling, Yuxue Li, Long Lu. Density Functional Theory Study on Thermal Decomposition Mechanisms of Ammonium Perchlorate [J]. Acta Chimica Sinica, 2023, 81(4): 328-337. |
| [6] | Yunfang Xu, Yang Li, Zitong Fu, Shaoyan Lin, Jie Zhu, Lei Wu. Palladium-catalyzed Stereoselective Synthesis of (Z)-[3]Dendralenes [J]. Acta Chimica Sinica, 2022, 80(10): 1369-1375. |
| [7] | Zhu Ren-Yi, Liao Kui, Yu Jin-Sheng, Zhou Jian. Recent Advances in Catalytic Asymmetric Synthesis of P-Chiral Phosphine Oxides [J]. Acta Chimica Sinica, 2020, 78(3): 193-216. |
| [8] | Zhang Ronghua, Xu Bing, Zhang Zhanming, Zhang Junliang. Ming-Phos/Copper(I)-Catalyzed Asymmetric[3+2] Cycloaddition of Azomethine Ylides with Nitroalkenes [J]. Acta Chimica Sinica, 2020, 78(3): 245-249. |
| [9] | Zhao Xiaochun, Ding Tianqi, Jiang Lüqi, Yi Wenbin. One-Pot Synthesis of Monofluoromethoxy Arenes from Aryl Halides, Arylboronic Acids and Arenes [J]. Acta Chimica Sinica, 2019, 77(12): 1263-1267. |
| [10] | Li Xin, Zhang Taiyang, Wang Tian, Zhao Yixin. Recent Progress of Photocatalysis Based on Metal Halide Perovskites [J]. Acta Chimica Sinica, 2019, 77(11): 1075-1088. |
| [11] | He Shijiang, Pi Jingjing, Li Yan, Lu Xi, Fu Yao. Nickel-Catalyzed Suzuki-Type Cross Coupling of Fluorinated Alkenyl Boronates with Alkyl Halides [J]. Acta Chim. Sinica, 2018, 76(12): 956-961. |
| [12] | Wu Miao Miao, Liu Shiqiang, Chen Hao, Wei Xuehu, Li Mingyang, Yang Zhibin, Ma Xiangdong. Superhalogen Substitutions in Cubic Halide Perovskite Materials for Solar Cells:A First-principles Investigation [J]. Acta Chim. Sinica, 2018, 76(1): 49-54. |
| [13] | Zhang Yongling, Wang Min, Cao Peng, Liao Jian. Copper-Catalyzed Enantioselective Aminoboration of Styrenes with Chiral Sulfoxide Phosphine Ligand [J]. Acta Chim. Sinica, 2017, 75(8): 794-797. |
| [14] | Li Ran, Xu Wenyuan, Zhao Jinqin, Yu Xin, Wang Wenguang, Tung Chen-Ho. Azo-bridged New Diiron Carbonyl Complex: Synthesis of Fe2(NR)2-(CO)6-x(PR3)x and the Derivatives [J]. Acta Chim. Sinica, 2017, 75(1): 92-98. |
| [15] | Yang Lijun, Ma Junan. New Advances on Nucleophilic Phosphine-Triggered Annulation Reactions of Allenoates [J]. Acta Chim. Sinica, 2016, 74(2): 130-148. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||