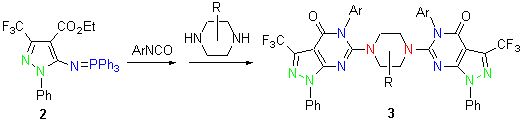
| [1] Holla, B. S.; Mahalinga, M.; Karthikeyan, M. S.; Akberali, P. M.; Shetty, N. S. Bioorg. Med. Chem. 2006, 14, 2040. [2] He, H. Y.; Zhao, J. N.; Jia, R.; Zhao, Y. L. Molecules 2011, 16, 10685. [3] Bakavoli, M.; Bagherzadeh, G.; Vaseghifar, M.; Shiri, A.; Pordel, M. Eur. J. Med. Chem. 2010, 45, 647. [4] Zhao, Y.; Zhai, X.; Chen, J.; Guo, S.; Gong, P. Chem. Res. Chin. Univ. 2006, 22, 468. [5] Allerton, C. M.; Barber, C. G.; Beaumont, K. C.; Brown, D. G.; Cole, S. M. J. Med. Chem. 2006, 49, 3581. [6] Ali, T. E. Eur. J. Med. Chem. 2009, 44, 4385. [7] Westrip, S. P. J. Appl. Cryst. 2010, 43, 920. [8] Wang, H.-Q.; Liu, H.; Liu, Z.-J. Chin. J. Org. Chem. 2004, 24, 1563 (in Chinese). (王宏青, 刘惠, 刘钊杰, 有机化学, 2004, 24, 1563.)[9] Diaa, A.; Ibrahim, A. M.; Al-Arab, E. E. Arkivoc 2009, (vii), 12. [10] Zhang, X. H.; Lin, Q. L.; Zhong, P. Molecules 2010, 15, 3079. [11] Dömling, A. Chem. Rev. 2006, 106, 17. [12] Dömling, A; Wang, W.; Wang, K. Chem. Rev. 2012, 112, 3083. [13] Isambert, N.; Lavilla, R. Chem. Eur. J. 2008, 14, 8444. [14] Sunderhaus, J. D.; Martin, S. F. Chem. Eur. J. 2009, 15, 1300. [15] Ganem, B. Acc. Chem. Res. 2009, 42, 463. [16] Palacios, F.; Alonso, C.; Aparicio, D. T.; Rubiales, G.; Santos, J. Tetrahedron 2007, 63, 523. [17] Carraro, F.; Naldini, A.; Pucci, A.; Locatelli, G. A.; Maga, G.; Schenone, S.; Bruno, O.; Ranise, A.; Bondavalli, F. J. Med. Chem. 2006, 49, 1549. [18] Yang, X. H.; Wu, M. H.; Sun, S. F.; Ding, M. W. J. Heterocycl. Chem. 2008, 45, 1365. [19] Yang, X.-H.; Wu, M.-H.; Ding, M.-W. Chin. J. Org. Chem. 2010, 30, 1032 (in Chinese). (杨绪红, 吴鸣虎, 丁明武, 有机化学, 2010, 30, 1032.)[20] Ding, M.-W. Sci. China: Chem. 2010, 40, 869 (in Chinese). (丁明武, 中国科学(化学) 2010, 40, 869.)[21] Parmar, K.; Suthar, B.; Suthar, A.; Maheta, A. J. Heterocycl. Chem. 2009, 46, 975. [22] Hafez, H. N.; EI-Gazzar, A. B. A. Bioorg. Med. Chem. Lett. 2008, 18, 5222. [23] Fang, Z.-D.; Liu, J.-Q. Chin. J. Org. Chem. 2011, 31, 2102 (in Chinese). (方正东, 吕鉴全, 有机化学, 2011, 31, 2102.)[24] Kirk, K. L. Org. Process Res. Dev. 2008, 12, 305. [25] Tomashenko, O. A.; Grushin, V. V. Chem. Rev. 2011, 111, 4475.[26] Katsuta, H.; Kitashima, T.; Sato, K. EP 1524271, 2005 [Chem. Abstr. 2005, 148, 588755]. |