有机化学 ›› 2026, Vol. 46 ›› Issue (3): 907-914.DOI: 10.6023/cjoc202510007 上一篇 下一篇
研究论文
刘轩宇a, 姚嘉欣a, 陈宇翔a, 张天宇a, 江雨晴a, 胡智学a, 孙宏顺a,*( ), 褚雪强a,b,*(
), 褚雪强a,b,*( )
)
收稿日期:2025-10-12
修回日期:2025-11-19
发布日期:2026-01-06
基金资助:
Xuanyu Liua, Jiaxin Yaoa, Yuxiang Chena, Tianyu Zhanga, Yuqing Jianga, Zhixue Hua, Hongshun Suna,*( ), Xueqiang Chua,b,*(
), Xueqiang Chua,b,*( )
)
Received:2025-10-12
Revised:2025-11-19
Published:2026-01-06
Contact:
*E-mail: Supported by:文章分享
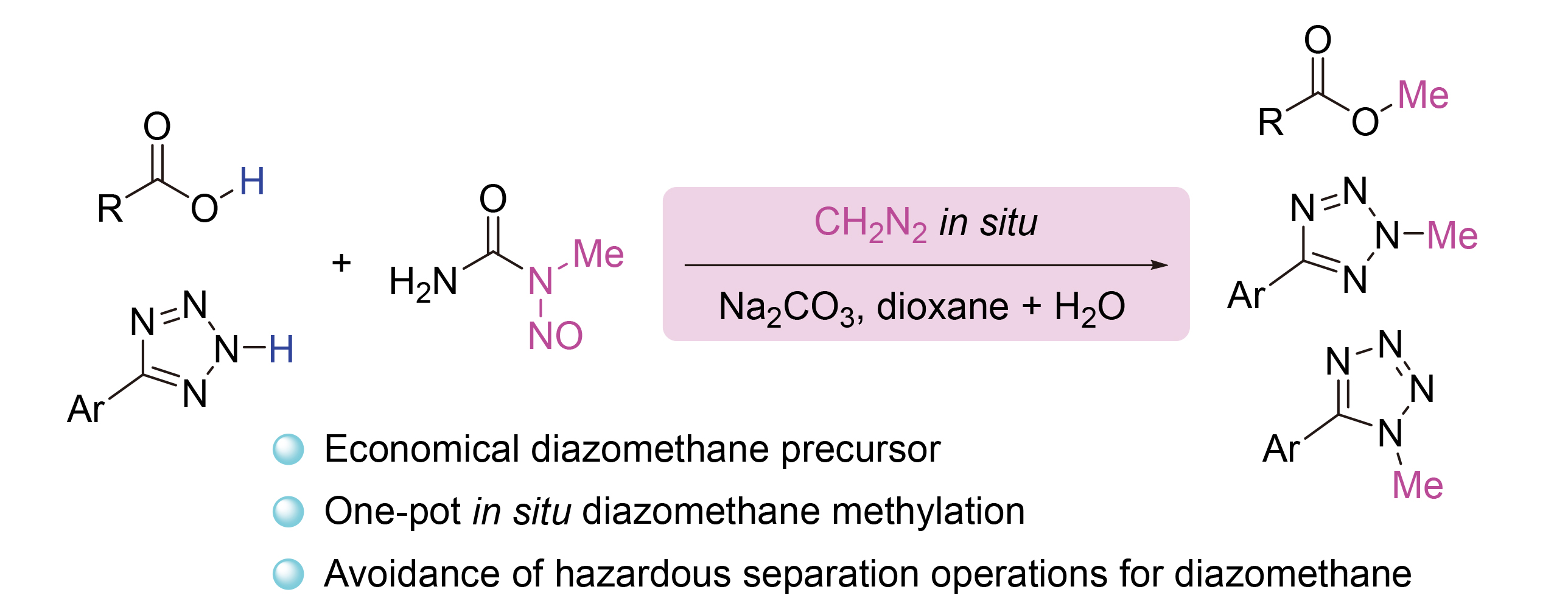
以N-甲基-N-亚硝基脲(MNU)作为经济、安全的重氮甲烷前体, 通过“一锅法”原位生成重氮甲烷, 实现了羧酸的效选择性甲基化反应. 该方法避免了高危险性重氮甲烷的分离和转移操作, 显著提升了实验安全性. 反应条件温和, 具备良好的官能团耐受性和广泛的底物适应性, 适用于多种羧酸、氨基酸、多肽及药物分子, 以中等至优良的收率获得相应的甲酯产物. 此外, 该策略还可成功应用于羧酸生物电子等排体苯基四唑的N-甲基化反应, 进一步展示了其在复杂分子后期修饰及药物化学合成中的潜在应用价值.
刘轩宇, 姚嘉欣, 陈宇翔, 张天宇, 江雨晴, 胡智学, 孙宏顺, 褚雪强. N-甲基-N-亚硝基脲(MNU)作为重氮甲烷前体参与的“一锅法”选择性甲基化反应研究[J]. 有机化学, 2026, 46(3): 907-914.
Xuanyu Liu, Jiaxin Yao, Yuxiang Chen, Tianyu Zhang, Yuqing Jiang, Zhixue Hu, Hongshun Sun, Xueqiang Chu. One-Pot Chemoselective Methylation Using N-Methyl-N-nitrosourea (MNU) as Diazomethane Precursor[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 907-914.
| Entry | Base (Dosage/equiv.) | Solvent | Yieldb/% |
|---|---|---|---|
| 1 | KOH (5) | Dioxane/H2O | 43 |
| 2 | NaOH (5) | Dioxane/H2O | 37 |
| 3 | NaHCO3 (5) | Dioxane/H2O | 82 |
| 4 | Na2CO3 (5) | Dioxane/H2O | 87 |
| 5 | K2CO3 (5) | Dioxane/H2O | 83 |
| 6 | K3PO4 (5) | Dioxane/H2O | 79 |
| 7 | Et3N (5) | Dioxane/H2O | 10 |
| 8 | DBU (5) | Dioxane/H2O | 23 |
| 9 | None | Dioxane/H2O | |
| 10 | Na2CO3 (5) | CH3CN/H2O | 52 |
| 11 | Na2CO3 (5) | THF/H2O | 35 |
| 12 | Na2CO3 (5) | CH2Cl2/H2O | 21 |
| 13 | Na2CO3 (5) | CH2Cl2 | 8 |
| 14 | Na2CO3 (5) | Dioxane | 48 |
| 15 | Na2CO3 (5) | H2O | 6 |
| 16c | Na2CO3 (5) | Dioxane/H2O | 65 |
| 17 | Na2CO3 (3) | Dioxane/H2O | 91 |
| 18 | Na2CO3 (2) | Dioxane/H2O | 98 |
| 19d | Na2CO3 (2) | Dioxane/H2O | 98 |
| 20e | Na2CO3 (2) | Dioxane/H2O | 40 |
| 21f | Na2CO3 (2) | Dioxane/H2O | 53 |
| 22g | Na2CO3 (2) | Dioxane/H2O | 98 |
| Entry | Base (Dosage/equiv.) | Solvent | Yieldb/% |
|---|---|---|---|
| 1 | KOH (5) | Dioxane/H2O | 43 |
| 2 | NaOH (5) | Dioxane/H2O | 37 |
| 3 | NaHCO3 (5) | Dioxane/H2O | 82 |
| 4 | Na2CO3 (5) | Dioxane/H2O | 87 |
| 5 | K2CO3 (5) | Dioxane/H2O | 83 |
| 6 | K3PO4 (5) | Dioxane/H2O | 79 |
| 7 | Et3N (5) | Dioxane/H2O | 10 |
| 8 | DBU (5) | Dioxane/H2O | 23 |
| 9 | None | Dioxane/H2O | |
| 10 | Na2CO3 (5) | CH3CN/H2O | 52 |
| 11 | Na2CO3 (5) | THF/H2O | 35 |
| 12 | Na2CO3 (5) | CH2Cl2/H2O | 21 |
| 13 | Na2CO3 (5) | CH2Cl2 | 8 |
| 14 | Na2CO3 (5) | Dioxane | 48 |
| 15 | Na2CO3 (5) | H2O | 6 |
| 16c | Na2CO3 (5) | Dioxane/H2O | 65 |
| 17 | Na2CO3 (3) | Dioxane/H2O | 91 |
| 18 | Na2CO3 (2) | Dioxane/H2O | 98 |
| 19d | Na2CO3 (2) | Dioxane/H2O | 98 |
| 20e | Na2CO3 (2) | Dioxane/H2O | 40 |
| 21f | Na2CO3 (2) | Dioxane/H2O | 53 |
| 22g | Na2CO3 (2) | Dioxane/H2O | 98 |
| [1] |
(a)
doi: 10.1002/anie.v52.47 pmid: 21631125 |
|
(b)
doi: 10.1021/cr200060g pmid: 21631125 |
|
| [2] |
doi: 10.1007/s10867-012-9268-6 |
| [3] |
(a)
doi: 10.1002/chem.v25.14 |
|
(b)
doi: 10.6023/cjoc202502038 |
|
|
(赵宇含, 马惠敏, 戈书林, 孔令斌, 有机化学, 2025, 45, 3469.)
doi: 10.6023/cjoc202502038 |
|
| [4] |
(a)
doi: 10.1021/ja406484v |
|
(b)
doi: 10.1002/anie.v50.29 |
|
|
(c)
|
|
| [5] |
(a)
doi: 10.1055/s-00000084 pmid: 23998269 |
|
(b)
doi: 10.1021/jo4016387 pmid: 23998269 |
|
| [6] |
(a)
doi: 10.1021/jo401941v pmid: 24138535 |
|
(b)
doi: 10.1080/17518253.2012.706647 pmid: 24138535 |
|
| [7] |
(a)
doi: 10.1002/cber.v27:2 |
|
(b)
|
|
| [8] |
doi: 10.2174/1385272822666180910140543 |
| [9] |
(a)
doi: 10.1021/jo035733r |
|
(b) Anonymous Dangerous Prop. Ind. Mater. Rep. 1992, 12, 530.
|
|
|
(c)
|
|
|
(d)
doi: 10.6023/cjoc201307030 |
|
|
(蔡少君, 肖龙强, 廖立琼, 刘立建, 有机化学, 2013, 33, 2602.)
doi: 10.6023/cjoc201307030 |
|
| [10] |
(a)
doi: 10.1021/acs.orglett.7b02866 |
|
(b)
doi: 10.1002/anie.v46:37 |
|
|
(c)
doi: 10.1002/(ISSN)1521-3897 |
|
| [11] |
(a)
doi: 10.1021/acs.oprd.9b00422 |
|
(b) Diazomethane (MAK Value Documentation, 1999) In The MAK-Collection for Occupational Health and Safety 2012, 1, 142.
|
|
|
(c)
|
|
| [12] |
(a)
doi: 10.1002/hlca.v63:4 |
|
(b)
doi: 10.1016/S0040-4020(97)00018-5 |
|
| [13] |
(a)
doi: 10.1021/ja01185a516 |
|
(b)
|
|
|
(c)
doi: 10.1016/0006-291X(60)90064-4 |
|
| [14] |
doi: 10.1002/jlcr.v57.12 |
| [15] |
(a)
doi: 10.1021/jo01314a036 |
|
(b)
doi: 10.1021/ac60335a020 |
|
| [16] |
doi: 10.1016/j.tetlet.2023.154554 |
| [17] |
(a)
doi: 10.6023/cjoc202307005 pmid: 24128181 |
|
(冯康博, 陈炯, 古双喜, 王海峰, 陈芬儿, 有机化学, 2024, 44, 378.)
doi: 10.6023/cjoc202307005 pmid: 24128181 |
|
|
(b)
doi: 10.1021/ol4027914 pmid: 24128181 |
|
|
(c)
doi: 10.1021/ar500359m pmid: 24128181 |
|
|
(d)
doi: 10.1002/anie.v50.5 pmid: 24128181 |
|
|
(e)
doi: 10.6023/cjoc201712029 pmid: 24128181 |
|
|
(郜云鹏, 王剑波, 有机化学, 2018, 38, 1275.)
doi: 10.6023/cjoc201712029 pmid: 24128181 |
|
| [18] |
(a)
doi: 10.1016/j.isci.2022.105005 |
|
(b)
doi: 10.6023/cjoc202401030 |
|
|
(刘蒙金, 肖燕, 周锴, 李子成, 黄文才, 有机化学, 2024, 44, 2251.)
doi: 10.6023/cjoc202401030 |
|
|
(c)
doi: 10.6023/cjoc202304005 |
|
|
(景智霞, 杜建喜, 蒋平, 阿布拉江•克依木, 有机化学, 2023, 43, 3930.)
doi: 10.6023/cjoc202304005 |
|
| [19] |
(a)
doi: 10.1126/science.1218781 pmid: 22442479 |
|
(b)
doi: 10.1002/anie.v59.5 pmid: 22442479 |
|
|
(c)
doi: 10.1039/D2OB00578F pmid: 22442479 |
|
| [20] |
(a)
doi: 10.1002/anie.v62.37 |
|
(b)
doi: 10.1002/cctc.v12.22 |
|
|
(c)
doi: 10.1055/a-1828-7504 |
|
|
(d)
doi: 10.1002/adsc.v368.3 |
|
| [21] |
(a)
doi: 10.1021/acs.chemrev.5b00121 |
|
(b)
doi: 10.1021/acs.oprd.7b00302 |
|
|
(c)
doi: 10.1002/cjoc.v43.4 |
|
| [22] |
doi: 10.1002/anie.201508804 pmid: 26663740 |
| [23] |
(a)
|
|
(b)
doi: 10.1016/j.tetlet.2015.11.005 |
|
| [24] |
doi: 10.1021/acs.orglett.4c00392 |
| [25] |
doi: 10.1002/chem.v26.27 |
| [26] |
|
| [27] |
doi: 10.1002/adsc.v365.11 |
| [28] |
doi: 10.1039/c3cc42596g |
| [29] |
doi: 10.1016/j.tet.2017.04.007 |
| [30] |
doi: 10.1039/C5OB01586C |
| [31] |
doi: 10.1021/acs.orglett.9b02617 |
| [32] |
doi: 10.1002/anie.201611570 pmid: 28145614 |
| [33] |
doi: 10.1016/j.tetlet.2009.06.104 |
| [34] |
doi: 10.1039/D5OB00057B |
| [35] |
doi: 10.1002/anie.v57.31 |
| [36] |
doi: 10.1002/jhet.v52.5 |
| [1] | 韩璐瑶, 胡硕真, 郭庆春, 郭红永, 高照群, 许颖, 张新胜. 二苯甲酮腙电氧化制备二苯重氮甲烷的研究[J]. 有机化学, 2024, 44(3): 951-965. |
| [2] | 陈任宏, 吴桂贞, 杨凯, 叶斌, 陈庆凤, 汪朝阳. 一锅法合成N-呋喃酮基磺酰腙类化合物[J]. 有机化学, 2021, 41(7): 2750-2759. |
| [3] | 朱文庆, 许婷怡, 韩文勇. 二氟甲基重氮甲烷作为含氟砌块的应用研究进展[J]. 有机化学, 2021, 41(4): 1275-1287. |
| [4] | 胡智宇, 姜国芳, 祝志强, 龚伯桢, 谢宗波, 乐长高. 深共融溶剂促进的亨利-傅克烷基化串联反应[J]. 有机化学, 2021, 41(1): 325-332. |
| [5] | 王帅, 杨成, 孙硕, 孙晗力, 王剑波. 钯催化芳香溴化物与三甲基硅基重氮甲烷的还原偶联及其在芳香化合物甲基化中的应用[J]. 有机化学, 2020, 40(11): 3881-3888. |
| [6] | 马会芳, 李文博, 阿布拉江·克依木. Amberlyst-15催化下的二氢[1,2,4]三唑并[1,5-a]嘧啶类及二氢苯并[4,5]咪唑并[1,2-a]嘧啶类衍生物的一锅法合成[J]. 有机化学, 2019, 39(7): 1945-1952. |
| [7] | 常青, 康娟, 张为钢, 王娟娟, 黄丹凤, 王克虎, 苏瀛鹏, 胡雨来. 锡粉促进下α-酰肼基-γ-戊烯酸乙酯的合成[J]. 有机化学, 2016, 36(12): 2920-2927. |
| [8] | 廖本仁, 洪镛裕, 徐军, 刘波. 应用Arndt-Eistert 反应合成手性非天然N-Fmoc-β-氨基酸(III)[J]. 有机化学, 2004, 24(1): 63-66. |
| [9] | 刘波,洪镛裕,廖本仁,熊飞,陶克美. 从N-Fmoc-L-α-氨基酸合成对应的同系物N--Fmoc-L-β-氨基酸(I)[J]. 有机化学, 2000, 20(2): 184-188. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||