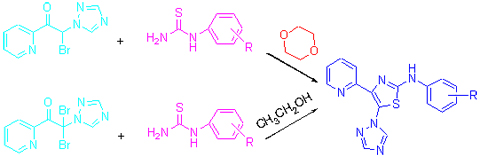
| [1] Zhou, Z. Z.; Chen, Q.; Yang, G. F. Chin. J. Org. Chem. 2008, 28, 1385 (in Chinese). (周中振, 陈琼, 杨光富, 有机化学, 2008, 28, 1385.)[2] Thomae, D.; Perspicace, E.; Hesse, S.; Kirsch, G.; Seck, P. Tetrahedron 2008, 64, 9309. [3] Yan, M.; Shi, D. Q.; Xiao, L. X. Chin. J. Org. Chem. 2008, 28, 1385 (in Chinese). (严曼, 石德清, 肖琳霞, 有机化学, 2008, 28, 1012.)[4] Zhou, X.; Jin, Z.; Liu, J. B.; Dai, H.; Fang, J. X. Heteroat. Chem. 2007, 18, 55. [5] Dzurilla, M.; Ružinský, M.; Kutschy, P. Colletc. Czech. Chem. Commun. 1999, 64, 1448. [6] Jung, K. Y.; Kim, S. K.; Gao, Z. G.; Gross, A. S.; Melman, N.; Jacobson, K. A.; Kim, Y. C. Bioorg. Med. Chem. 2004, 12, 613. [7] Zhang, W. T.; Ruan, J. L.; Wu, P. F.; Jiang, F. C.; Zhang, L. N.; Fang, W.; Chen, X. L.; Wang, Y.; Cao, B. S.; Chen, G. Y.; Zhu, Y. J.; Gu, J.; Chen, J. G. J. Med. Chem. 2009, 52, 718. [8] Öğretir, C.; Demirayak, S.; Duran, M. J. Chem. Eng. Data 2010, 55, 1137. [9] Hu, A. X.; He, L. M.; Xia, L.; Xu, J. J. Chin. J. Med. Chem. 2006, 16, 229 (in Chinese). (胡艾希, 贺丽敏, 夏林, 徐娟娟, 中国药物化学杂志. 2006, 16, 229.)[10] Wang, D. M.; Sun, J. Y.; Shang, Y. J. Chin. J. Org. Chem. 2011, 31, 1136 (in Chinese). (王冬梅, 孙军勇, 商永嘉, 有机化学, 2011, 31, 1136.) [11] Wang, F.; Li, W. H.; Li, D.; Fan, Z.; Li, Z. Chin. J. Org. Chem. 2012, 32, 601 (in Chinese). (王锋, 李稳宏, 李冬, 范峥, 李珍, 有机化学, 2012, 32, 601.) [12] Chu, C. H.; Hui, X. P.; Sun, L.; Zhang, Y.; Zhang, Z.; Li, Z. C.; Liao, R. A. J. Chin. Chem. Soc. 2001, 8, 121. [13] Takano, H.; Oguri, Y.; Kato, T. J. Pestic. Sci. 1983, 8, 572. [14] Izumi, K.; Yamaguchi, I.; wada, A.; Oshio, H.; Takahashi, N. Plant Cell Phys. 1984, 25, 611. [15] Singh, P. K. J. Agric. Food Chem. 2012, 60, 5813. [16] Dong, F. S.; Li, J.; Chankvetadze, B.; Cheng, Y. P.; Xu, J.; Liu, X. G.; Li, Y. B.; Chen, X.; Bertucci, C.; Tedsco, D.; Zanasi, R.; Zheng, Y. Q. Environ. Sci. Technol. 2013, 47, 3386. [17] Hantzsch, A.; Weber, J. H. Ber Dtsch. Chem. Ges. 1887, 20, 3118. [18] Rudolph, J. Tetrahedron 2000, 56, 3161. [19] Dahiya, R.; Pujari, H. K. Indian J. Chem. 1986, 25B, 966. [20] Ralhan, N. K.; Sandhu, G. S.; Sachdev, H. S.; Narang, K. S. J. Indian Chem. Soc. 1960, 37, 773. [21] Bariana, D. S.; Sachdev H. S.; Narang, K. S. J. Indian Chem. Soc. 1955, 32, 427. [22] Fang, J. X.; Jin, Z.; Liu, T. Liu, W. Appl. Organoment. Chem. 2003, 17, 145. [23] Liu, J. B.; Tao, W. F.; Dai, H.; Jin, Z.; Fang, J. X. Heteroat. Chem. 2007, 18, 376. [24] Wang, M.; Zhang, M. G.; Bruce, M. H.; Kathy, M. D.; George, S. W. Bioorg. Med. Chem. 2006, 14, 8599. |