Chinese Journal of Organic Chemistry ›› 2024, Vol. 44 ›› Issue (1): 180-194.DOI: 10.6023/cjoc202306016 Previous Articles Next Articles
ARTICLES
杨维清a, 葛宴兵a, 陈元元a, 刘萍a, 付海燕b, 马梦林a,b,*( )
)
收稿日期:2023-06-19
修回日期:2023-09-04
发布日期:2023-09-21
基金资助:
Weiqing Yanga, Yanbing Gea, Yuanyuan Chena, Ping Liua, Haiyan Fub, Menglin Maa,b( )
)
Received:2023-06-19
Revised:2023-09-04
Published:2023-09-21
Contact:
*E-mail: Supported by:Share
Weiqing Yang, Yanbing Ge, Yuanyuan Chen, Ping Liu, Haiyan Fu, Menglin Ma. Design and Synthesis of Fluorescent 1,8-Napthalimide Derivatives and Their Identification of Cysteine[J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 180-194.
| Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) | Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) | Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) | Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 4a | 353 | 10.78 | 5a | 436 | 13.60 | 6a | 338 | 17.20 | 6a+Cys | 348 | 16.25 |
| 4b | 352 | 10.86 | 5b | 436 | 21.15 | 6b | 346 | 16.10 | 6b+Cys | 366 | 9.60 |
| 4c | 352 | 7.64 | 5c | 434 | 11.40 | 6c | 338 | 17.85 | 6c+Cys | 344 | 15.90 |
| 4d | 350 | 11.29 | 5d | 437 | 13.70 | 6d | 353 | 9.00 | 6d+Cys | 351 | 7.45 |
| 4e | 354 | 14.85 | 5e | 441 | 10.15 | 6e | 354 | 9.75 | 6e+Cys | 355 | 8.55 |
| 4f | 352 | 13.70 | 5f | 436 | 10.55 | 6f | 352 | 7.10 | 6f+Cys | 351 | 5.70 |
| 4g | 354 | 13.50 | 5g | 437 | 8.10 | 6g | 344 | 9.55 | 6g+Cys | 348 | 10.60 |
| 4h | 355 | 12.60 | 5h | 440 | 18.40 | 6h | 346 | 7.30 | 6h+Cys | 344 | 8.20 |
| 4i | 354 | 4.45 | 5i | 434 | 10.85 | 6i | 347 | 10.80 | 6i+Cys | 347 | 8.30 |
| 4j | 354 | 8.90 | 5j | 436 | 12.10 | 6j | 344 | 12.25 | 6j+Cys | 349 | 13.30 |
| 4k | 354 | 11.95 | 5k | 437 | 9.65 | 6k | 344 | 15.85 | 6k+Cys | 348 | 14.60 |
| 4l | 356 | 12.70 | 5l | 438 | 10.75 | 6l | 348 | 13.20 | 6l+Cys | 349 | 12.10 |
| 4m | 354 | 11.45 | 5m | 437 | 8.80 | 6m | 364 | 15.05 | 6m+Cys | 364 | 15.90 |
| 4n | 353 | 13.45 | 5n | 437 | 12.15 | 6n | 348 | 14.25 | 6n+Cys | 354 | 14.20 |
| 4o | 357 | 14.15 | 5o | 438 | 15.15 | 6o | 345 | 13.35 | 6o+Cys | 359 | 13.10 |
| 4p | 357 | 13.80 | 5p | 437 | 19.25 | 6p | 345 | 12.40 | 6p+Cys | 345 | 11.15 |
| 4q | 359 | 11.90 | 5q | 437 | 17.10 | 6q | 345 | 10.45 | 6q+Cys | 365 | 9.70 |
| 4r | 352 | 8.75 | 5r | 437 | 30.25 | 6r | 342 | 12.15 | 6r+Cys | 359 | 10.55 |
| 4s | 353 | 5.10 | 5s | 436 | 7.55 | 6s | 341 | 9.05 | 6s+Cys | 340 | 8.65 |
| Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) | Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) | Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) | Compd. | λmax/nm | ε/(103 L• mol-¹•cm-¹) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 4a | 353 | 10.78 | 5a | 436 | 13.60 | 6a | 338 | 17.20 | 6a+Cys | 348 | 16.25 |
| 4b | 352 | 10.86 | 5b | 436 | 21.15 | 6b | 346 | 16.10 | 6b+Cys | 366 | 9.60 |
| 4c | 352 | 7.64 | 5c | 434 | 11.40 | 6c | 338 | 17.85 | 6c+Cys | 344 | 15.90 |
| 4d | 350 | 11.29 | 5d | 437 | 13.70 | 6d | 353 | 9.00 | 6d+Cys | 351 | 7.45 |
| 4e | 354 | 14.85 | 5e | 441 | 10.15 | 6e | 354 | 9.75 | 6e+Cys | 355 | 8.55 |
| 4f | 352 | 13.70 | 5f | 436 | 10.55 | 6f | 352 | 7.10 | 6f+Cys | 351 | 5.70 |
| 4g | 354 | 13.50 | 5g | 437 | 8.10 | 6g | 344 | 9.55 | 6g+Cys | 348 | 10.60 |
| 4h | 355 | 12.60 | 5h | 440 | 18.40 | 6h | 346 | 7.30 | 6h+Cys | 344 | 8.20 |
| 4i | 354 | 4.45 | 5i | 434 | 10.85 | 6i | 347 | 10.80 | 6i+Cys | 347 | 8.30 |
| 4j | 354 | 8.90 | 5j | 436 | 12.10 | 6j | 344 | 12.25 | 6j+Cys | 349 | 13.30 |
| 4k | 354 | 11.95 | 5k | 437 | 9.65 | 6k | 344 | 15.85 | 6k+Cys | 348 | 14.60 |
| 4l | 356 | 12.70 | 5l | 438 | 10.75 | 6l | 348 | 13.20 | 6l+Cys | 349 | 12.10 |
| 4m | 354 | 11.45 | 5m | 437 | 8.80 | 6m | 364 | 15.05 | 6m+Cys | 364 | 15.90 |
| 4n | 353 | 13.45 | 5n | 437 | 12.15 | 6n | 348 | 14.25 | 6n+Cys | 354 | 14.20 |
| 4o | 357 | 14.15 | 5o | 438 | 15.15 | 6o | 345 | 13.35 | 6o+Cys | 359 | 13.10 |
| 4p | 357 | 13.80 | 5p | 437 | 19.25 | 6p | 345 | 12.40 | 6p+Cys | 345 | 11.15 |
| 4q | 359 | 11.90 | 5q | 437 | 17.10 | 6q | 345 | 10.45 | 6q+Cys | 365 | 9.70 |
| 4r | 352 | 8.75 | 5r | 437 | 30.25 | 6r | 342 | 12.15 | 6r+Cys | 359 | 10.55 |
| 4s | 353 | 5.10 | 5s | 436 | 7.55 | 6s | 341 | 9.05 | 6s+Cys | 340 | 8.65 |
| Compd. | λEx/nm | λEm/nm | Hightb | Compd. | λEx/nm | λEm/nm | Hightb | Compd. | λEx/nm | λEm/nm | ΦFa/% |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 5a | 440 | 537 | 214.2 | 6a | 274 | 328 | 67.6 | 6a+Cys | 378 | 468 | 9.06 |
| 5b | 440 | 537 | 200.7 | 6b | 272 | 328 | 171.0 | 6b+Cys | 372 | 472 | —c |
| 5c | 276 | 327 | 167.0 | 6c | 273 | 326 | 182.0 | 6c+Cys | 372 | 470 | —c |
| 5d | 440 | 536 | 213.5 | 6d | 280 | 310 | 28.7 | 6d+Cys | 378 | 470 | 11.36 |
| 5e | 440 | 537 | 191.8 | 6e | 280 | 310 | 33.9 | 6e+Cys | 370 | 467 | 12.56 |
| 5f | 440 | 538 | 149.6 | 6f | 370 | 472 | 66.6 | 6f+Cys | 375 | 468 | 16.32 |
| 5g | 270 | 327 | 75.62 | 6g | 270 | 324 | 60.2 | 6g+Cys | 370 | 465 | —c |
| 5h | 257 | 340 | 209.0 | 6h | 280 | 310 | 36.7 | 6h+Cys | 370 | 468 | 0.18 |
| 5i | 440 | 539 | 83.35 | 6i | 273 | 325 | 72.3 | 6i+Cys | 371 | 468 | —c |
| 5j | 438 | 540 | 135.7 | 6j | 280 | 310 | 33.1 | 6j+Cys | 380 | 469 | 1.52 |
| 5k | 440 | 538 | 119.6 | 6k | 280 | 310 | 29.6 | 6k+Cys | 370 | 471 | 1.50 |
| 5l | 440 | 536 | 167.8 | 6l | 370 | 466 | 48.7 | 6l+Cys | 370 | 468 | 19.25 |
| 5m | 442 | 540 | 97.3 | 6m | 280 | 310 | 34.8 | 6m+Cys | 370 | 459 | —c |
| 5n | 440 | 538 | 85.3 | 6n | 280 | 310 | 32.6 | 6n+Cys | 370 | 468 | 11.89 |
| 5o | 280 | 311 | 36.3 | 6o | 280 | 311 | 34.1 | 6o+Cys | 370 | 470 | 8.26 |
| 5p | 440 | 542 | 105.9 | 6p | 280 | 310 | 35.2 | 6p+Cys | 372 | 466 | 6.71 |
| 5q | 440 | 540 | 161.4 | 6q | 280 | 310 | 35.2 | 6q+Cys | 370 | 470 | 17.89 |
| 5r | 440 | 538 | 98.6 | 6r | 280 | 310 | 36.8 | 6r+Cys | 370 | 462 | —c |
| 5s | 440 | 542 | 35.0 | 6s | 280 | 310 | 36.3 | 6s+Cys | 370 | 309 | 0.13 |
| Compd. | λEx/nm | λEm/nm | Hightb | Compd. | λEx/nm | λEm/nm | Hightb | Compd. | λEx/nm | λEm/nm | ΦFa/% |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 5a | 440 | 537 | 214.2 | 6a | 274 | 328 | 67.6 | 6a+Cys | 378 | 468 | 9.06 |
| 5b | 440 | 537 | 200.7 | 6b | 272 | 328 | 171.0 | 6b+Cys | 372 | 472 | —c |
| 5c | 276 | 327 | 167.0 | 6c | 273 | 326 | 182.0 | 6c+Cys | 372 | 470 | —c |
| 5d | 440 | 536 | 213.5 | 6d | 280 | 310 | 28.7 | 6d+Cys | 378 | 470 | 11.36 |
| 5e | 440 | 537 | 191.8 | 6e | 280 | 310 | 33.9 | 6e+Cys | 370 | 467 | 12.56 |
| 5f | 440 | 538 | 149.6 | 6f | 370 | 472 | 66.6 | 6f+Cys | 375 | 468 | 16.32 |
| 5g | 270 | 327 | 75.62 | 6g | 270 | 324 | 60.2 | 6g+Cys | 370 | 465 | —c |
| 5h | 257 | 340 | 209.0 | 6h | 280 | 310 | 36.7 | 6h+Cys | 370 | 468 | 0.18 |
| 5i | 440 | 539 | 83.35 | 6i | 273 | 325 | 72.3 | 6i+Cys | 371 | 468 | —c |
| 5j | 438 | 540 | 135.7 | 6j | 280 | 310 | 33.1 | 6j+Cys | 380 | 469 | 1.52 |
| 5k | 440 | 538 | 119.6 | 6k | 280 | 310 | 29.6 | 6k+Cys | 370 | 471 | 1.50 |
| 5l | 440 | 536 | 167.8 | 6l | 370 | 466 | 48.7 | 6l+Cys | 370 | 468 | 19.25 |
| 5m | 442 | 540 | 97.3 | 6m | 280 | 310 | 34.8 | 6m+Cys | 370 | 459 | —c |
| 5n | 440 | 538 | 85.3 | 6n | 280 | 310 | 32.6 | 6n+Cys | 370 | 468 | 11.89 |
| 5o | 280 | 311 | 36.3 | 6o | 280 | 311 | 34.1 | 6o+Cys | 370 | 470 | 8.26 |
| 5p | 440 | 542 | 105.9 | 6p | 280 | 310 | 35.2 | 6p+Cys | 372 | 466 | 6.71 |
| 5q | 440 | 540 | 161.4 | 6q | 280 | 310 | 35.2 | 6q+Cys | 370 | 470 | 17.89 |
| 5r | 440 | 538 | 98.6 | 6r | 280 | 310 | 36.8 | 6r+Cys | 370 | 462 | —c |
| 5s | 440 | 542 | 35.0 | 6s | 280 | 310 | 36.3 | 6s+Cys | 370 | 309 | 0.13 |
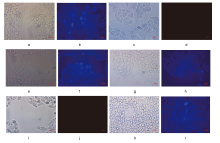

| [1] |
(a) Sun, Y.; Fu, M. L; Bian, M. L; Zhu, Q. Biotechnol Bioeng. 2023, 120, 7.
doi: 10.1002/bit.v120.1 |
|
(b) Ahmed, N.; Zareen, W.; Ye, Y. Chin. Chem. Lett. 2022, 33, 2765.
doi: 10.1016/j.cclet.2021.12.092 |
|
|
(c) Liu, F. R.; Jing, J.; Zhang, X. L. Chin. J. Org. Chem. 2023, 43, 2053 (in Chinese).
doi: 10.6023/cjoc202209005 |
|
|
(刘飞冉, 敬静, 张小玲, 有机化学, 2023, 43, 2053.)
|
|
| [2] |
(a) Kolinska, J.; Grzelakowska, A. Spectrochim. Acta, A Mol. Biomol. Spectrosc. 2021, 262, 120151.
doi: 10.1016/j.saa.2021.120151 |
|
(b) Zhang, Y. L; Shao, X. M; Wang, Y.; Pan, F. C.; Kang, R. X.; Peng, F. F.; Huang, Z. T.; Zhang, W. J; Zhao, W. L. Chem. Commun. 2015, 51, 4245.
doi: 10.1039/C4CC08687B |
|
|
(c) Ge, C. P.; Wang, H.; Zhang, B. X.; Yao, J.; Li, X. M; Feng, W. M; Zhou, P. P.; Wang, Y. W; Fang, J. G Chem. Commun. 2015, 51, 14913.
|
|
| [3] |
(a) Xu, Z. Y.; Zhao, X. F.; Zhou, M.; Zhang, Z. J; Qin, T. Y; Wang, D.; Wang, L.; Peng, X. J.; Liu, B. Sens. Actuators, B 2021, 345, 130367.
doi: 10.1016/j.snb.2021.130367 |
|
(b) Xu, Z. Y.; Si, S. F.; Zhang, Z. J; Tan, H. Y; Qin, T. Y; Wang, Z. L; Wang, D.; Wang, L.; Liu, B. Anal. Chim. Acta 2021, 1176, 338763.
doi: 10.1016/j.aca.2021.338763 |
|
| [4] |
(a) Anbu, S.; Paul, A.; Surendranath, K.; Sidali, A.; Pombeiro, A. J. L. J. Inorg. Biochem. 2021, 220, 111466.
doi: 10.1016/j.jinorgbio.2021.111466 |
|
(b) Dimov, S. M.; Georgiev, N. I.; Asiri, A. M.; Bojinov, V. B J. Fluoresc. 2014, 24, 1621.
|
|
|
(c) Georgiev, N. I.; Dimov, S. M.; Asiri, A. M.; Alamry, K. A.; Obaid, A.Y.; Bojinov, V. B. J. Lumin. 2014, 149, 325.
doi: 10.1016/j.jlumin.2014.01.028 |
|
| [5] |
Qu, L. J.; Yin, C. X; Huo, F. J.; Li, J. F; Chao, J. B; Zhang, Y. B. Sens. Actuators, B 2014, 195, 246.
doi: 10.1016/j.snb.2014.01.026 |
| [6] |
Vasilev, A. A.; Baluschev, S.; Cheshmedzhieva, D.; Ilieva, S.; Castano, O. D.; Vaquero, J. J.; Angelova, S. E.; Landfester, K. Aus. J. Chem. 2015, 68, 1399.
doi: 10.1071/CH15139 |
| [7] |
Girouard, S.; Houle, M. H.; Grandbois, A.; Keillor, J. W.; Michnick, S. W. J. Am. Chem. Soc. 2005, 127, 559.
doi: 10.1021/ja045742x |
| [8] |
Khosravi, A.; Moradian, S.; Gharanjig, K.; Afshar, F. T. J. Chin. Chem. Soc. 2005, 52, 495.
doi: 10.1002/jccs.v52.3 |
| [9] |
Alexiou, M. S.; Tyman, J. H. P. J. Chem. Res. Miniprint 2000, 5, 632.
|
| [10] |
Costi, M. P.; Tondi, D.; Rinaldi, M.; Barlocco, D.; Cignarella, G.; Santi, D. V.; Musiu, C.; Pudu, I.; Vacca, G.; Colla, P. L. Eur. J. Med. Chem. 1996, 31, 1011.
doi: 10.1016/S0223-5234(97)86180-6 |
| [11] |
Cui, L.; Peng, Z. X.; Ji, C. F.; Huang, J. H. Huang, D. T.; Ma, J.; Zhang, S. P.; Qian, X. H.; Xu, Y. F. Chem. Commun. 2014, 50, 1485.
doi: 10.1039/C3CC48304E |
| [12] |
Zhou, J; Fang, C. L; Liu, Y; Zhao, Y; Zhang, N; Liu, X. J.; Wang, F. Y.; Shangguan, D. H. Org. Biomol. Chem. 2015, 13, 3931.
doi: 10.1039/C5OB00302D |
| [13] |
Wang, J. F; Jin, S.; Akay, S.; Wang, B. H. Eur. J. Med. Chem., 2007, 13, 2091.
|
| [14] |
Yuan, D. W; Brown, R. G.; Hepworth, J. D.; Alexiou, M. S.; Tyman, J. H. P. J. Heterocycl. Chem. 2008, 45, 397.
doi: 10.1002/jhet.v45:2 |
| [15] |
Li, X. M; Zheng, Y. J; Tong, H. J; Qian, R.; Zhou, L.; Liu, G. X; Tang, Y.; Li, H.; Lou, K. Y.; Wang, W. Chem.-Eur. J. 2016, 27, 9247.
|
| [1] | Penghui Li, Qingyang Xie, Fuxian Wan, Yuanhong Zhang, Lin Jiang. Synthesis and Fungicidal Activity of Novel Substituted Pyrimidine-5-carboxamides Bearing Cyclopropyl Moiety [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 650-656. |
| [2] | Fakai Zou, Nengzhong Wang, Hui Yao, Hui Wang, Mingguo Liu, Nianyu Huang. Regio- and Stereo-selective Synthesis of 1β-/3R-Aryl Thiosugar [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 593-604. |
| [3] | Luyao Li, Zhongwen He, Zhenguo Zhang, Zhenhua Jia, Teck-Peng Loh. Application of Triaryl Carbenium in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 421-437. |
| [4] | Qinggang Mei, Qinghan Li. Recent Progress of Visible Light-Induced the Synthesis of C(3) (Hetero)arylthio Indole Compounds [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 398-408. |
| [5] | Qianfan Zhao, Yongzheng Chen, Shiming Zhang. Application and Mechanism Study of Carbon-Based Metal-Free Catalysts in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 137-147. |
| [6] | Shan Chen, Zhilin Chen, Qiong Hu, Yanshuang Meng, Yue Huang, Pingfang Tao, Liru Lu, Guobao Huang. Recognition of Bis-thiourea Tweezers to Neutral Molecules in Non-Polar Solvent [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 277-281. |
| [7] | Huakun Wang, Xiaolong Ren, Yining Xuan. Study of the Halide Salt Catalyzed [3+2] Cycloaddition of α,β-Epoxy Carboxylate with Isocyanate [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 251-258. |
| [8] | Yukun Jin, Baoyi Ren, Fushun Liang. Visible Light-Mediated Selective C—F Bond Cleavage of Trifluoromethyl Groups and Its Application in Synthesizing gem-Difluoro-Containing Compounds [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 85-110. |
| [9] | Cuiyun Ma, Hailan Luo, Fuhua Zhang, Dan Guo, Shuxing Chen, Fei Wang. Green Biosynthesis, Photophysical Properties and Application of 3-Pyrrolyl BODIPY [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 216-223. |
| [10] | Bozhen Wang, Jie Zhang, Chunhui Nian, Mingming Jin, Miaomiao Kong, Wulan Li, Wenfei He, Jianzhang Wu. Synthesis and Antitumor Activity of 3,4-Dichlorophenyl Amides [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 232-241. |
| [11] | Yingzhen Zhang, Dandan Jiang, Juanhua Li, Jingjing Wang, Kunming Liu, Jinbiao Liu. Construction Strategy and Imaging of Highly Selective Selenocysteine Fluorescent Probes [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 41-53. |
| [12] | Shihang Yu, Jiawei Liu, Biyu An, Qinghua Bian, Min Wang, Jiangchun Zhong. Asymmetric Synthesis of the Contact Sex Pheromone of Neoclytus acuminatus acuminatus (Fabricius) [J]. Chinese Journal of Organic Chemistry, 2024, 44(1): 301-308. |
| [13] | Yang Li, Jinding Yuan, Di Zhao. Deep Eutectic Solvent of 1,3-Dimethylurea/L-(+)-Tartaric Acid for the Green Synthesis of (E)-2-Styrylquinoline-3-carboxylic Acid Derivatives [J]. Chinese Journal of Organic Chemistry, 2023, 43(9): 3268-3276. |
| [14] | Dandan Sui, Nannan Cen, Ruoqu Gong, Yang Chen, Wenbo Chen. Supporting-Electrolyte-Free Electrochemical Synthesis of Trifluoromethylated Oxindoles in Continuous Flow [J]. Chinese Journal of Organic Chemistry, 2023, 43(9): 3239-3245. |
| [15] | Ruixia Cao, Yuping Jia. Synthesis and Biological Activity of Novel Pyrrolo[2,3-d]pyrimidine Derivatives Containing Coumarin [J]. Chinese Journal of Organic Chemistry, 2023, 43(9): 3304-3311. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||