Chinese Journal of Organic Chemistry ›› 2026, Vol. 46 ›› Issue (3): 1000-1007.DOI: 10.6023/cjoc202509013 Previous Articles Next Articles
ARTICLES
梁新宇a,†, 米春春a,†, 郭福军a, 谢文斌a,*( ), 史钦钦a,*(
), 史钦钦a,*( ), 黄辉a,b,*(
), 黄辉a,b,*( )
)
收稿日期:2025-09-09
修回日期:2025-11-06
发布日期:2025-12-09
作者简介:†共同第一作者
基金资助:
Xinyu Lianga, Chunchun Mia, Fujun Guoa, Wenbin Xiea,*( ), Qinqin Shia,*(
), Qinqin Shia,*( ), Hui Huanga,b,*(
), Hui Huanga,b,*( )
)
Received:2025-09-09
Revised:2025-11-06
Published:2025-12-09
Contact:
*E-mail: About author:†These authors contributed equally to this work
Supported by:Share
Xinyu Liang, Chunchun Mi, Fujun Guo, Wenbin Xie, Qinqin Shi, Hui Huang. Desulfinative Cross-Coupling of Aryl Sodium Sulfinate with Aryl Sulfonium Salts[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 1000-1007.

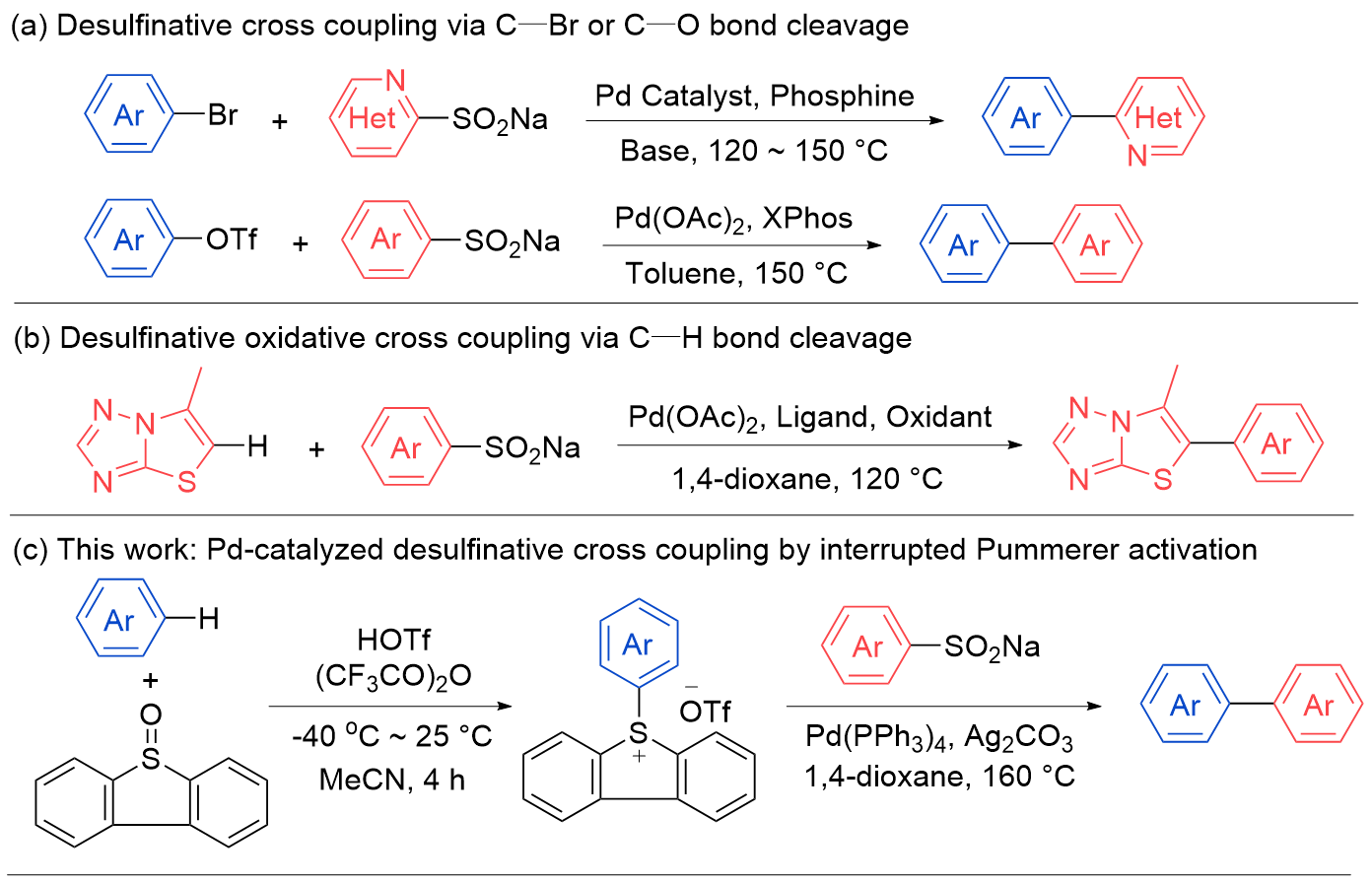
| Entry | [Pd] (10 mol%) | Ligand (20 mol%) | Additive (2.0 equiv.) | Solvent | Temperature/℃ | Yieldb/% |
|---|---|---|---|---|---|---|
| 1 | Pd(OAc)2 | PCy3 | K2CO3 | 1,4-dioxane | 160 | 67 |
| 2 | Pd(OAc)2 | P(t-Bu)3H+BF4- | K2CO3 | 1,4-dioxane | 160 | 42 |
| 3 | Pd(PPh3)4 | — | K2CO3 | 1,4-dioxane | 160 | 72 |
| 4 | Pd(OAc)2 | dtbpf | K2CO3 | 1,4-dioxane | 160 | 63 |
| 5 | Pd(OAc)2 | dppf | K2CO3 | 1,4-dioxane | 160 | 61 |
| 6 | Pd(PPh3)4 | — | KOt-Bu | 1,4-dioxane | 160 | 16 |
| 7 | Pd(PPh3)4 | — | KF | 1,4-dioxane | 160 | 71 |
| 8 | Pd(PPh3)4 | — | Cs2CO3 | 1,4-dioxane | 160 | 16 |
| 9 | Pd(PPh3)4 | — | AgOAc | 1,4-dioxane | 160 | 68 |
| 10 | Pd(PPh3)4 | — | Ag2CO3 | 1,4-dioxane | 160 | 78 (72)c |
| 11 | Pd(PPh3)4 | — | Ag2CO3 | 1,4-dioxane | 110 | 31 |
| 12 | Pd(PPh3)4 | — | Ag2CO3 | DMSO | 160 | 72 |
| Entry | [Pd] (10 mol%) | Ligand (20 mol%) | Additive (2.0 equiv.) | Solvent | Temperature/℃ | Yieldb/% |
|---|---|---|---|---|---|---|
| 1 | Pd(OAc)2 | PCy3 | K2CO3 | 1,4-dioxane | 160 | 67 |
| 2 | Pd(OAc)2 | P(t-Bu)3H+BF4- | K2CO3 | 1,4-dioxane | 160 | 42 |
| 3 | Pd(PPh3)4 | — | K2CO3 | 1,4-dioxane | 160 | 72 |
| 4 | Pd(OAc)2 | dtbpf | K2CO3 | 1,4-dioxane | 160 | 63 |
| 5 | Pd(OAc)2 | dppf | K2CO3 | 1,4-dioxane | 160 | 61 |
| 6 | Pd(PPh3)4 | — | KOt-Bu | 1,4-dioxane | 160 | 16 |
| 7 | Pd(PPh3)4 | — | KF | 1,4-dioxane | 160 | 71 |
| 8 | Pd(PPh3)4 | — | Cs2CO3 | 1,4-dioxane | 160 | 16 |
| 9 | Pd(PPh3)4 | — | AgOAc | 1,4-dioxane | 160 | 68 |
| 10 | Pd(PPh3)4 | — | Ag2CO3 | 1,4-dioxane | 160 | 78 (72)c |
| 11 | Pd(PPh3)4 | — | Ag2CO3 | 1,4-dioxane | 110 | 31 |
| 12 | Pd(PPh3)4 | — | Ag2CO3 | DMSO | 160 | 72 |
| [1] |
(a)
doi: 10.1021/jacs.5c07642 |
|
(b)
doi: 10.1038/s41467-025-62092-3 |
|
|
(c)
doi: 10.1002/anie.v60.39 |
|
| [2] |
(a)
doi: 10.1021/acscatal.5b00448 pmid: 21618370 |
|
(b)
doi: 10.1002/anie.201107017 pmid: 21618370 |
|
|
(c)
doi: 10.1021/cs300082f pmid: 21618370 |
|
|
(d)
doi: 10.1002/anie.201101379 pmid: 21618370 |
|
| [3] |
doi: 10.1021/acs.orglett.7b02424 |
| [4] |
doi: 10.1021/acs.jmedchem.0c01786 |
| [5] |
(a)
doi: 10.1021/ja303401s pmid: 22679903 |
|
(b)
doi: 10.1021/ja400881n pmid: 22679903 |
|
| [6] |
(a)
doi: 10.1021/jacs.6b03283 |
|
(b)
doi: 10.1021/jacs.7b07444 |
|
| [7] |
doi: 10.1007/s00244-004-0105-1 |
| [8] |
doi: 10.1021/acs.chemrev.5b00393 pmid: 26555044 |
| [9] |
(a)
doi: 10.1039/C1CS15127D |
|
(b)
doi: 10.1002/chem.v24.31 |
|
| [10] |
doi: 10.1039/c1cs15093f |
| [11] |
doi: 10.1002/ejoc.v2016.3 |
| [12] |
(a)
doi: 10.1021/jacs.8b09595 pmid: 30412397 |
|
(b)
doi: 10.1002/anie.v60.41 pmid: 30412397 |
|
|
(c)
doi: 10.1021/jo302005s pmid: 30412397 |
|
| [13] |
doi: 10.1055/s-00000083 |
| [14] |
doi: 10.1021/cr500431s |
| [15] |
(a)
doi: 10.1038/s41586-019-0982-0 |
|
(b)
doi: 10.1021/acs.orglett.1c01322 |
|
|
(c)
|
|
|
(d)
doi: 10.1021/acscatal.3c03096 |
|
|
(e)
doi: 10.1021/acscatal.5c00082 |
|
|
(f)
|
|
|
(g)
doi: 10.1039/D2SC01241C |
|
| [16] |
(a)
doi: 10.1039/c7sc00675f pmid: 28936330 |
|
(b)
doi: 10.1021/jacs.9b13260 pmid: 28936330 |
|
| [17] |
(a)
doi: 10.1021/ja00155a007 |
|
(b)
doi: 10.1038/s41467-018-03718-7 |
|
| [18] |
(a)
doi: 10.1021/acs.chemrev.2c00478 pmid: 37134187 |
|
(b)
doi: 10.1021/acs.chemrev.2c00881 pmid: 37134187 |
|
| [19] |
doi: 10.1021/jacs.1c09884 pmid: 34871503 |
| [20] |
doi: 10.1021/ja902046m |
| [21] |
doi: 10.1002/chem.v17.22 |
| [22] |
doi: 10.1021/acs.orglett.9b04474 |
| [23] |
doi: 10.1021/acs.orglett.9b00942 pmid: 30908055 |
| [24] |
|
| [25] |
doi: 10.1039/C7GC02804K |
| [26] |
doi: 10.1002/anie.v54.40 |
| [27] |
|
| [28] |
doi: 10.1021/jo301384r |
| [29] |
doi: 10.1021/jo101709n |
| [30] |
doi: 10.1002/anie.201605584 pmid: 27456275 |
| [31] |
doi: 10.1002/chem.v25.9 |
| [32] |
doi: 10.1007/s11426-019-9652-x |
| [33] |
doi: 10.1039/D1RA04947J |
| [34] |
doi: 10.1021/acscatal.1c04533 |
| [1] | Cui Wei, Jinqiu Liu, Can Deng, Ning Zou, Wenjun Zhou. Recent Advances on the Synthesis of Spirofluorene-Based Compounds [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 817-839. |
| [2] | Yingjie Liu, Laisheng Min, Ruirong Yang, Dongxue Song, Rui Peng, Deqiang Liang. CuI-Catalyzed C—C Bond Coupling Reaction for the Construction of 2-Carbonyl-1,4-diketones [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 603-611. |
| [3] | Yiling Zeng, Fangpeng Liang, Hui Li, Rongrong Liu, Shiqing Li. Multi-component C—H Annulation of α-Oxocarboxylic Acids, Allynes and Primary Amines to Yield Isoquinolinium Skeletons [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 554-563. |
| [4] | Lanxing Ren, A'na Xu, Xilin Xiao, Hengzhi You, Lijuan Song. Theoretical Study on the Solvent Effect on the Enantioselectivity of Iridium-Catalyzed Asymmetric Hydrogenation Reactions [J]. Chinese Journal of Organic Chemistry, 2025, 45(8): 2904-2912. |
| [5] | Ming Chen, Jing Zhang. Recent Progress in Traceless Directed Functionalization Reactions of Arylcarboxylic Acids [J]. Chinese Journal of Organic Chemistry, 2025, 45(8): 2660-2676. |
| [6] | Xinghui Tao, Kaiyue Guo, Jitan Zhang, Meihua Xie, Jiaping Wu. Synthesis of Functionalized Azepinones and Phenanthridines via a Pd-Catalyzed ortho-C—H Amination/C—H Arylation Cyclization Cascade of Aryl Bromides [J]. Chinese Journal of Organic Chemistry, 2025, 45(8): 2825-2835. |
| [7] | Chunjie Qian, Boyang He, Xingyu Liu, Ping Wang, Guangchun Gao, Shihui Liu. Copper/Selectfluor Co-catalyzed Ritter-Type Benzylic C—H Amination [J]. Chinese Journal of Organic Chemistry, 2025, 45(6): 2181-2188. |
| [8] | Shuang Wang, Yangjie Mao, Shaojie Lou, Danqian Xu. Recent Advances in Asymmetric C—H Bond Functionalization Using Oxidizing Directing Groups [J]. Chinese Journal of Organic Chemistry, 2025, 45(6): 1961-1994. |
| [9] | Xun Tian, Guogang Deng, Xiaodong Yang. Progress on Cobalt-Catalyzed C(sp2)—H Activation for the Construction of Nitrogen-Containing Benzo Heterocycles [J]. Chinese Journal of Organic Chemistry, 2025, 45(2): 655-667. |
| [10] | Zeshui Liu, Zhenzhen Guo, Junlong Niu. Recent Progress in the Construction of Silacycles by Transition- Metal-Catalyzed C—H Silylation [J]. Chinese Journal of Organic Chemistry, 2025, 45(2): 423-447. |
| [11] | Mingshun Mei, Yanghui Zhang. Palladium-Catalyzed Intermolecular Functionalization of Unactivated Methylene C(sp3)—H Bonds [J]. Chinese Journal of Organic Chemistry, 2025, 45(2): 620-640. |
| [12] | Yu Zou, Weicong Guo, Jun Wang. Application of Planar Chiral Cyclopentadienyl Rhodium Catalysts without Chiral Substituents in Asymmetric C—H Activation [J]. Chinese Journal of Organic Chemistry, 2025, 45(2): 466-476. |
| [13] | Xuan Wang, Maochen Liu, Yao Zhong, Renjie Song. Advances in Design of Covalent Organic Frameworks and Their Application in Catalytic C—H Activation [J]. Chinese Journal of Organic Chemistry, 2025, 45(2): 448-465. |
| [14] | Fan Yang, Xiaomeng Fan, Xuejing Yao, Ruijie Mi, Songjie Yu, Xingwei Li, Jian Xiao. Rhodium(III)-Catalyzed Annulative Coupling between Sulfoxonium Ylides and Diazo Compounds [J]. Chinese Journal of Organic Chemistry, 2025, 45(1): 331-342. |
| [15] | Mengke Wen, Yiyue Li, Haokang Du, Zhangpei Chen, Xifa Yang. Synthesis of Spiropyrans via Rh(III)-Catalyzed [3+3] Cyclization of 3-Aryl-2H-1,4-benzoxazine and Diazonaphthalene-2H-ones [J]. Chinese Journal of Organic Chemistry, 2024, 44(7): 2223-2232. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||