Chinese Journal of Organic Chemistry ›› 2026, Vol. 46 ›› Issue (4): 1205-1221.DOI: 10.6023/cjoc202601016 Previous Articles Next Articles
REVIEWS
收稿日期:2026-01-13
修回日期:2026-03-10
发布日期:2026-04-13
通讯作者:
舒超
作者简介:† 共同第一作者
基金资助:
Xinran Hana, Hengyu Huoa, Xuepan Songb, Chao Shua,*( )
)
Received:2026-01-13
Revised:2026-03-10
Published:2026-04-13
Contact:
Chao Shu
About author:†These authors contributed equally to this work
Supported by:Share
Xinran Han, Hengyu Huo, Xuepan Song, Chao Shu. Recent Advances for the Synthesis of α-Alkylidene-γ-lactones[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1205-1221.
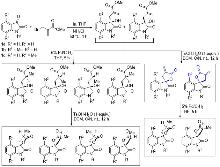
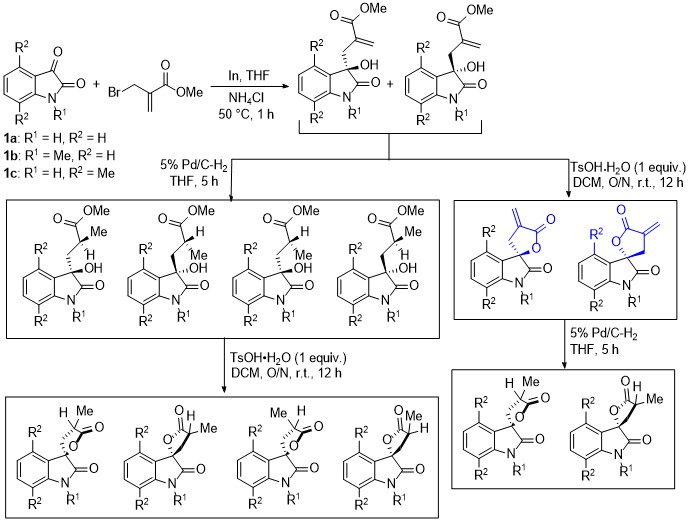

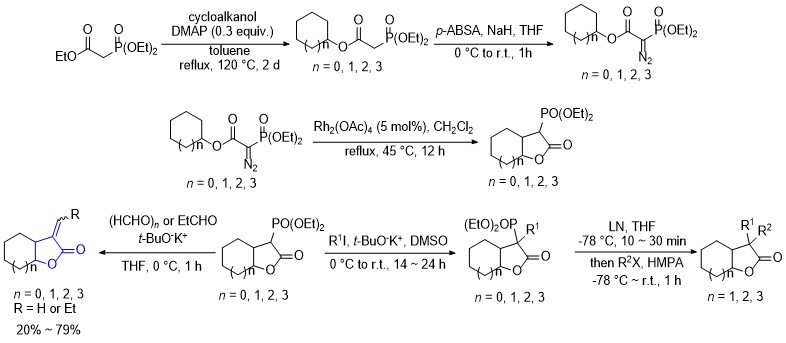
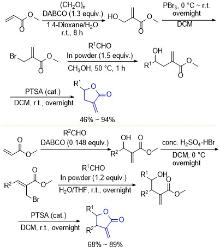
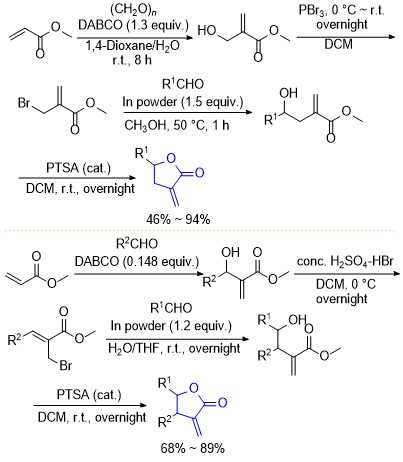
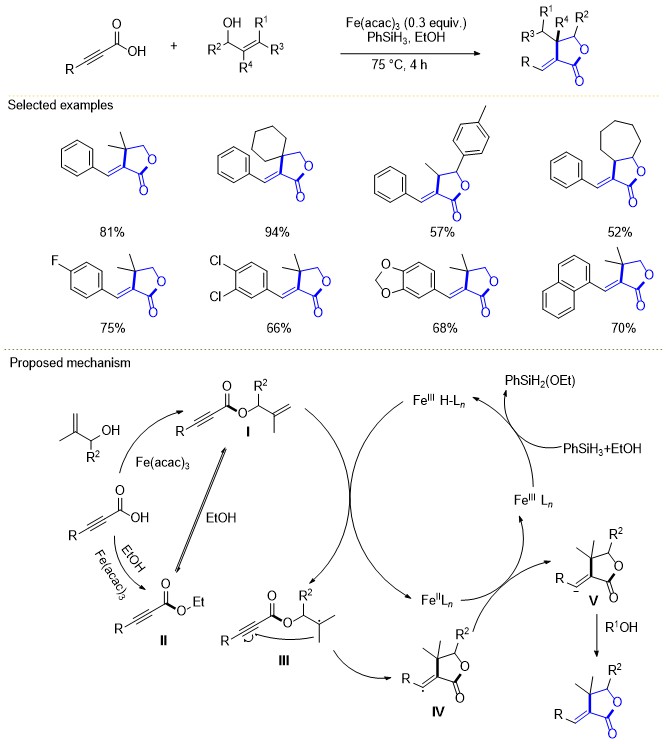
| [1] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
| [2] |
|
| [3] |
|
| [4] |
|
| [5] |
(a)
|
|
(b)
|
|
| [6] |
|
| [7] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [8] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
| [9] |
(a)
|
|
(b)
|
|
|
(c)
|
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
(a)
|
|
(b)
|
|
| [16] |
|
| [17] |
|
| [18] |
(a)
|
|
(b)
|
|
| [19] |
(a)
|
|
(b)
|
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
(a)
|
|
(b)
|
|
| [41] |
|
| [42] |
(a)
|
|
(b)
|
|
| [43] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
| [44] |
(a)
|
|
(b)
|
|
| [45] |
(a)
|
|
(b)
|
|
| [46] |
|
| [47] |
|
| [48] |
|
| [49] |
|
|
(杨政, 黄丹凤, 文岚, 王娟娟, 王克虎, 胡雨来, 有机化学, 2018, 38, 1725.)
|
|
| [50] |
|
| [51] |
|
| [52] |
|
| [53] |
|
| [54] |
|
| [55] |
|
| [56] |
|
| [57] |
|
|
(李建晓, 张振明, 李春生, 罗维, 杨少容, 有机化学, 2015, 35, 2199.)
|
|
| [58] |
|
| [61] |
|
| [59] |
|
| [60] |
|
| [1] | Zhenlei Zou, Yifan Li, Chen Chen, Wangzhe Chen, Weigang Zhang, Yi Wang. Recent Advances in Sulfoxide Synthesis via Sulfinyl Building Blocks [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1635-1658. |
| [2] | Yuqing Tang, Zixian Yang, Bing Yang, Yi Wang, Bingnan Du. Research Progress in Mechanically Driven Radical Reactions under Ball Milling Conditions [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1111-1145. |
| [3] | Lingxu Meng, Lin Shen, Xiangling Luo, Yumei Lin, Lei Gong. Design of Photoactive Cr- and Co-Based Complexes and Their Applications in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1513-1528. |
| [4] | Chao Ma, Xiaoling Xu, Xiang Luo, Ruijuan Lu, Guoqi Yu, Tao Cai. Electrochemical Synthesis of 4-Sulfenylisocoumarins via Radical Cascade Annulation of o-Alkynylbenzoates with Thiophenols [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 1039-1049. |
| [5] | Shunli Zhou, Ji Liu, Ting Lei, Binquan Yang, Shizhi Jiang. Advances in Chiral Synthesis of 2-Aryl-2H-chromene Derivatives [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 773-785. |
| [6] | Xueqian Wang, Baozhong Duan, Hongchang Tian, Lei Zhang. Research Progress in Synthesis of Indole Alkaloid Catharanthine [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 725-742. |
| [7] | Wenfu Cheng, Minghao Zhao, Yuchen Yang, Lijia Wang. The First Total Synthesis of N-Methyl Pratensilin B [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 670-677. |
| [8] | Weigang He, Mingyue Duan, Shuai Yan, Weijie Ye, Xueyuan Xu, Xianyu Sun. Progress in the Total Synthesis of the Kopsia Alkaloids Arboridinine and Arborisidine [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 443-454. |
| [9] | Mina Zhao, Jiayi Tang. Progress in the Synthesis of Imidazole Compounds [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 455-474. |
| [10] | Junfeng Guo, Chunying Li, Ronggui Hu, Haojie Rong, Jiangwei Li, Yongmei Du, Yue Qin, Jian* Lü, Dao'an Sun. Advances in the Construction Methods of Cyclopropane Skeleton and Their Applications [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 379-398. |
| [11] | Lingyan Tian, Shoufeng Wang, Wei Zeng. Progress in the Synthesis and Biological Studies of S-1-Propenyl-L-cysteine from Garlic [J]. Chinese Journal of Organic Chemistry, 2026, 46(1): 74-86. |
| [12] | Lilin Zhao, Zhaoqi Li, Liping Zhong, Long Min. Recent Progress in the Total Synthesis of abeo-Steroids [J]. Chinese Journal of Organic Chemistry, 2026, 46(1): 21-38. |
| [13] | Xinyi Cui, Lifan Guo, Congxuan Ma, Yun Li, Jianhua Liang. Structural Modifications, Structure-Activity Relationships, and Total Synthesis Advances in Erythromycin Analogs against Resistant Pathogens [J]. Chinese Journal of Organic Chemistry, 2026, 46(1): 39-73. |
| [14] | Qianqian Zhao, Peiyao Wei, Sundian Liu, Boxin Zhang, Chengyuan Liang. Applications of Palladium-Catalyzed Cascade Reactions in Total Synthesis of Complex Natural Products with Quaternary Carbon Centers [J]. Chinese Journal of Organic Chemistry, 2025, 45(9): 3289-3300. |
| [15] | Liping Guan, Zhiqi Lin, Chaohua Fang, Qiushi Wang, Zhiyang Fu, Bin Zhang. Advances in Synthesis Methods of Quinoline and Isoquinoline Derivatives [J]. Chinese Journal of Organic Chemistry, 2025, 45(9): 3255-3288. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||