Chinese Journal of Organic Chemistry ›› 2022, Vol. 42 ›› Issue (11): 3562-3587.DOI: 10.6023/cjoc202204060 Previous Articles Next Articles
Special Issue: 有机氟化学虚拟合辑
REVIEWS
郭檬檬a, 于子伦a, 陈玉兰a, 葛丹华a,*( ), 马猛涛b, 沈志良a, 褚雪强a,*(
), 马猛涛b, 沈志良a, 褚雪强a,*( )
)
收稿日期:2022-04-25
修回日期:2022-06-24
发布日期:2022-07-13
通讯作者:
葛丹华, 褚雪强
作者简介:基金资助:
Mengmeng Guoa, Zilun Yua, Yulan Chena, Danhua Gea( ), Mengtao Mab, Zhiliang Shena, Xueqiang Chua(
), Mengtao Mab, Zhiliang Shena, Xueqiang Chua( )
)
Received:2022-04-25
Revised:2022-06-24
Published:2022-07-13
Contact:
Danhua Ge, Xueqiang Chu
About author:Supported by:Share
Mengmeng Guo, Zilun Yu, Yulan Chen, Danhua Ge, Mengtao Ma, Zhiliang Shen, Xueqiang Chu. Difluorinated Silyl Enol Ethers as Fluorine-Containing Building Blocks for the Synthesis of Organofluorine Compounds[J]. Chinese Journal of Organic Chemistry, 2022, 42(11): 3562-3587.
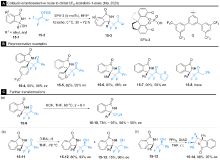
| [1] |
(a) Sorochinsky, A. E.; Fustero, S.; Soloshonok, V. A.; Liu, H. Chem. Rev. 2014, 114, 2432.
doi: 10.1021/cr4002879 pmid: 25078138 |
|
(b) Prchalová, E.; Štěpánek, O.; Smrček, S.; Kotora, M. Future Med. Chem. 2014, 6, 1201.
doi: 10.4155/fmc.14.53 pmid: 25078138 |
|
| [2] |
Hu, J.; Ding, K. Acta Chim. Sinica 2018, 76, 905. (in Chinese)
doi: 10.6023/A1812E001 |
|
( 胡金波, 丁奎岭, 化学学报 2018, 76, 905.)
doi: 10.6023/A1812E001 |
|
| [3] |
(a) Ge, D.; Chu, X.-Q. Org. Chem. Front. 2022, 9, 2013.
doi: 10.1039/D1QO01749G |
|
(b) Chen, B.; Vicic, D. A. Top. Organomet. Chem. 2014, 52, 113.
|
|
| [4] |
(a) Schirlin, D.; Tarnus, C.; Baltzer, S.; Remy, J. M. Bioorg. Med. Chem. Lett. 1992, 2, 651.
doi: 10.1016/S0960-894X(00)80383-2 pmid: 8551523 |
|
(b) Dreyer, G. B.; Metcalf, B. W. Tetrahedron Lett. 1988, 29, 6885.
doi: 10.1016/S0040-4039(00)88466-X pmid: 8551523 |
|
|
(c) Nikolaou, A.; Kokotou, M. G.; Vasilakaki, S.; Kokotos, G. Biochim. Biophys. Acta, Mol. Cell Biol. Lipids 2019, 1864, 941.
doi: 10.1016/j.bbalip.2018.08.009 pmid: 8551523 |
|
|
(d) Cregge, R. J.; Durham, S. L.; Farr, R. A.; Gallion, S. L.; Hare, C. M.; Hoffman, R. V.; Janusz, M. J.; Kim, H.-O.; Koehl, J. R.; Mehdi, S.; Metz, W. A.; Peet, N. P.; Pelton, J. T.; Schreuder, H. A.; Sunder, S.; Tardif, C. J. Med. Chem. 1998, 41, 2461.
pmid: 8551523 |
|
|
(e) Silva, A. M.; Cachau, R. E.; Sham, H. L.; Erickson, J. W. J. Mol. Biol. 1996, 255, 321.
pmid: 8551523 |
|
|
(f) Fah, C.; Hardegger, L. A.; Baitsch, L.; Schweizer, W. B.; Meyer, S.; Bur, D.; Diederich, F. Org. Biomol. Chem. 2009, 7, 3947.
doi: 10.1039/b908489d pmid: 8551523 |
|
| [5] |
(a) Mei, H.; Liu, J.; Fustero, S.; Román, R.; Ruzziconi, R.; Soloshonok, V. A.; Han, J. Org. Biomol. Chem. 2019, 17, 762.
doi: 10.1039/C8OB02843E |
|
(b) Gong, Y.; Yu, J.-S.; Hao, Y.-J.; Zhou, Y.; Zhou, J. Asian J. Org. Chem. 2019, 8, 610.
doi: 10.1002/ajoc.201900071 |
|
|
(c) Dong, D.-Q.; Yang, H.; Shi, J.-L.; Si, W.-J.; Wang, Z.-L.; Xu, X.-M. Org. Chem. Front. 2020, 7, 2538.
doi: 10.1039/D0QO00567C |
|
|
(d) Belhomme, M.-C.; Besset, T.; Poisson, T.; Pannecoucke, X. Chem. Eur. J. 2015, 21, 12836.
doi: 10.1002/chem.201501475 |
|
|
(e) Rong, J.; Ni, C.; Hu, J. Asian J. Org. Chem. 2017, 6, 139.
doi: 10.1002/ajoc.201600509 |
|
| [6] |
Yamana, M.; Ishihara, T.; Ando, T. Tetrahedron Lett. 1983, 24, 507.
doi: 10.1016/S0040-4039(00)81449-5 |
| [7] |
(a) Liu, Y.-L.; Zhou, J. Chem. Commun. 2012, 48, 1919.
doi: 10.1039/c2cc17140f |
|
(b) Liu, Y.-L.; Zeng, X.-P.; Zhou, J. Acta Chim. Sinica 2012, 70, 1451 (in Chinese)
doi: 10.6023/A12040145 |
|
|
( 刘运林, 周剑, 化学学报 2012, 70, 1451.)
doi: 10.6023/A12040145 |
|
|
(c) Liu, Y.-L.; Liao, F.-M.; Niu, Y.-F.; Zhao, X.-L.; Zhou, J. Org. Chem. Front. 2014, 1, 742.
doi: 10.1039/C4QO00126E |
|
|
(d) Liao, F.-M.; Liu, Y.-L.; Yu, J.-S.; Zhou, F.; Zhou, J. Org. Biomol. Chem. 2015, 13, 8906.
doi: 10.1039/C5OB01125F |
|
|
(e) Liao, F.-M.; Gao, X.-T.; Hu, X.-S.; Xie, S.-L.; Zhou, J. Sci. Bull. 2017, 62, 1504.
doi: 10.1016/j.scib.2017.10.016 |
|
| [8] |
(a) Yu, J.-S.; Zhou, J. Org. Biomol. Chem. 2015, 13, 10968.
doi: 10.1039/C5OB01895A |
|
(b) Yu, J.-S.; Zhou, J. Org. Chem. Front. 2016, 3, 298.
doi: 10.1039/C5QO00407A |
|
|
(c) Hu, X.-S.; Du, Y.; Yu, J.-S.; Liao, F.-M.; Ding, P.-G.; Zhou, J. Synlett 2017, 28, 2194.
doi: 10.1055/s-0036-1588475 |
|
|
(d) Hu, X.-S.; Yu, J.-S.; Gong, Y.; Zhou, J. J. Fluorine Chem. 2019, 219, 106.
doi: 10.1016/j.jfluchem.2019.01.003 |
|
| [9] |
(a) Uneyama, K.; Tanaka, H.; Kobayashi, S.; Shioyama, M.; Amii, H. Org. Lett. 2004, 6, 2733.
pmid: 15281756 |
|
(b) Guo, Y.; Shreeve, J. M. Chem. Commun. 2007, 3583.
pmid: 15281756 |
|
|
(c) Guo, Y.; Twamley, B.; Shreeve, J. M. Org. Biomol. Chem. 2009, 7, 1716.
doi: 10.1039/b900311h pmid: 15281756 |
|
|
(d) Guo, Y.; Tao, G.-H.; Blumenfeld, A.; Shreeve, J. M. Organometallics 2010, 29, 1818.
doi: 10.1021/om1000259 pmid: 15281756 |
|
| [10] |
(a) Lefebvre, O.; Brigaud, T.; Portella, C. J. Org. Chem. 2001, 66, 4348.
pmid: 11397175 |
|
(b) Belanger, E.; Cantin, K.; Messe, O.; Tremblay, M.; Paquin, J.-F. J. Am. Chem. Soc. 2007, 129, 1034.
doi: 10.1021/ja067501q pmid: 11397175 |
|
| [11] |
(a) Poisson, T.; Gembus, V.; Dalla, V.; Oudeyer, S.; Levacher, V. J. Org. Chem. 2010, 75, 7704.
doi: 10.1021/jo101585t pmid: 20958068 |
|
(b) Liao, K.; Hu, X.-S.; Zhu, R.-Y.; Rao, R.-H.; Yu, J.-S.; Zhou, F.; Zhou, J. Chin. J. Chem. 2019, 37, 799.
doi: 10.1002/cjoc.201900198 pmid: 20958068 |
|
| [12] |
Higashiya, S.; Chung, W. J.; Lim, D. S.; Ngo, S. C.; Kelly IV, W. H.; Toscano, P. J.; Welch, J. T. J. Org. Chem. 2004, 69, 6323.
doi: 10.1021/jo049551o |
| [13] |
(a) Yu, J.-S.; Liao, F.-M.; Gao, W.-M.; Liao, K.; Zuo, R.-L.; Zhou, J. Angew. Chem., Int. Ed. 2015, 54, 7381.
doi: 10.1002/anie.201501747 |
|
(b) Hao, Y.-J.; Hu, X.-S.; Yu, J.-S.; Zhou, F.; Zhou, Y.; Zhou, J. Tetrahedron 2018, 74, 7395.
doi: 10.1016/j.tet.2018.11.017 |
|
| [14] |
(a) Liao, F.-M.; Cao, Z.-Y.; Yu, J.-S.; Zhou, J. Angew. Chem., Int. Ed. 2017, 56, 2459.
doi: 10.1002/anie.201611625 |
|
(b) Liao, F.-M.; Du, Y.; Zhou, F.; Zhou, J. Acta Chim. Sinica 2018, 76, 862. (in Chinese)
doi: 10.6023/A18060238 |
|
|
( 廖富民, 杜溢, 周锋, 周剑, 化学学报 2018, 76, 862.)
doi: 10.6023/A18060238 |
|
| [15] |
Hu, X.-S.; Yu, J.-S.; Zhou, J. Chem. Commun. 2019, 55, 13638.
doi: 10.1039/C9CC07677H |
| [16] |
Chu, X.-Q.; Ge, D.; Cui, Y.-Y.; Shen, Z.-L.; Li, C.-J. Chem. Rev. 2021, 121, 12548.
doi: 10.1021/acs.chemrev.1c00084 |
| [17] |
Yu, J.; Liu, Y.; Tang, J.; Wang X.; Zhou, J. Angew. Chem., Int. Ed. 2014, 53, 9512.
doi: 10.1002/anie.201404432 |
| [18] |
(a) Müller, K.; Faeh, C.; Diederich, F. Science 2007, 317, 1881.
doi: 10.1126/science.1131943 pmid: 17901324 |
|
(b) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
doi: 10.1039/B610213C pmid: 17901324 |
|
|
(c) O’Hagan, D. Chem. Soc. Rev. 2008, 37, 308.
doi: 10.1039/B711844A pmid: 17901324 |
|
| [19] |
Tian, Y.-P.; Gong, Y.; Hu, X.-S.; Yu, J.-S.; Zhou, Y.; Zhou, J. Org. Biomol. Chem. 2019, 17, 9430.
doi: 10.1039/C9OB02129A |
| [20] |
Yang, J.; Liu, S.; Hong, P.; Li, J.; Wang, Z.; Ren, J. J. Org. Chem. 2022, 87, 1144.
doi: 10.1021/acs.joc.1c02504 |
| [21] |
(a) Purser, S.; Moore, P. R.; Swallow, S.; Gouverneur, V. Chem. Soc. Rev. 2008, 37, 320.
doi: 10.1039/B610213C |
|
(b) Gillis, E. P.; Eastman, K. J.; Hill, M. D.; Donnelly, D. J.; Meanwell, N. A. Synthesis 2013, 45, 1.
doi: 10.1055/s-0032-1317575 |
|
| [22] |
Liu, S.; Li, Y.; Wang, F.; Ma, C.; Yang, G.; Yang, J.; Ren, J. Synthesis 2022, 54, 161.
doi: 10.1055/a-1581-2408 |
| [23] |
(a) Jonet, S.; Cherouvrier, F.; Brigaud, T.; Portella, C. Eur. J. Org. Chem. 2005, 4304.
|
|
(b) Yuan, Z.; Wei, Y.; Shi, M. Chin. J. Chem. 2010, 28, 1709.
doi: 10.1002/cjoc.201090289 |
|
|
(c) Chu, L.; Zhang, X.; Qing, F.-L. Org. Lett. 2009, 11, 2197.
doi: 10.1021/ol900541n |
|
|
(d) Wu, Y.-B.; Wan, L.; Lu, G.-P.; Cai, C. Eur. J. Org. Chem. 2017, 3438.
|
|
| [24] |
Hu, X.-S.; Ding, P.-G.; Yu, J.-S.; Zhou, J. Org. Chem. Front. 2019, 6, 2500.
doi: 10.1039/C9QO00577C |
| [25] |
(a) Yoshimura, A.; Zhdankin, V. V. Chem. Rev. 2016, 116, 3328.
doi: 10.1021/acs.chemrev.5b00547 pmid: 18986207 |
|
(b) Zhdankin, V. V.; Stang, P. J. Chem. Rev. 2008, 108, 5299.
doi: 10.1021/cr800332c pmid: 18986207 |
|
| [26] |
Huang, X.; Zhang, Y.; Zhang, C.; Zhang, L.; Xu, Y.; Kong, L.; Wang, Z.-X.; Peng, B. Angew. Chem., Int. Ed. 2019, 58, 5956.
doi: 10.1002/anie.201900745 |
| [27] |
(a) Roche, S. P.; Porco Jr, J. A. Angew. Chem., Int. Ed. 2011, 50, 4068.
doi: 10.1002/anie.201006017 pmid: 30839047 |
|
(b) Zheng, C.; You, S.-L. Nat. Prod. Rep. 2019, 36, 1589.
doi: 10.1039/c8np00098k pmid: 30839047 |
|
|
(c) Ding, Q.; Ye, Y.; Fan, R. Synthesis 2013, 45, 1.
doi: 10.1055/s-0032-1317575 pmid: 30839047 |
|
| [28] |
Huang, X.; Zhang, Y.; Liang, W.; Zhang, Q.; Zhan, Y.; Kong, L.; Peng, B. Chem. Sci. 2020, 11, 3048.
doi: 10.1039/d0sc00244e pmid: 34122809 |
| [29] |
(a) Ilardi, E. A.; Vitaku, E.; Njardarson, J. T. J. Med. Chem. 2014, 57, 2832.
doi: 10.1021/jm401375q |
|
(b) Feng, M.; Tang, B.; Liang, S. H.; Jiang, X. Curr. Top. Med. Chem. 2016, 16, 1200.
doi: 10.2174/1568026615666150915111741 |
|
| [30] |
Huang, X.; Zhao, W.; Liang, Y.; Wang, M.; Zhan, Y.; Zhang, Y.; Kong, L.; Wang, Z.-X.; Peng, B. Org. Chem. Front. 2021, 8, 1280.
doi: 10.1039/D0QO01513J |
| [31] |
Gao, X.; Cheng, R.; Xiao, Y.-L.; Wan, X.-L.; Zhang, X. Chem 2019, 5, 2987.
doi: 10.1016/j.chempr.2019.09.012 |
| [32] |
Jiang, X.; Meyer, D.; Baran, D.; Cortes Gonzalez, M. A.; Szabo, K. J. J. Org. Chem. 2020, 85, 8311.
doi: 10.1021/acs.joc.0c01030 |
| [33] |
(a) Speck, K.; Magauer, T. J. Org. Chem. 2013, 9, 2048.
|
|
(b) Bhatia, R. K. Curr. Top. Med. Chem. 2016, 17, 189.
doi: 10.2174/1568026616666160530154100 |
|
| [34] |
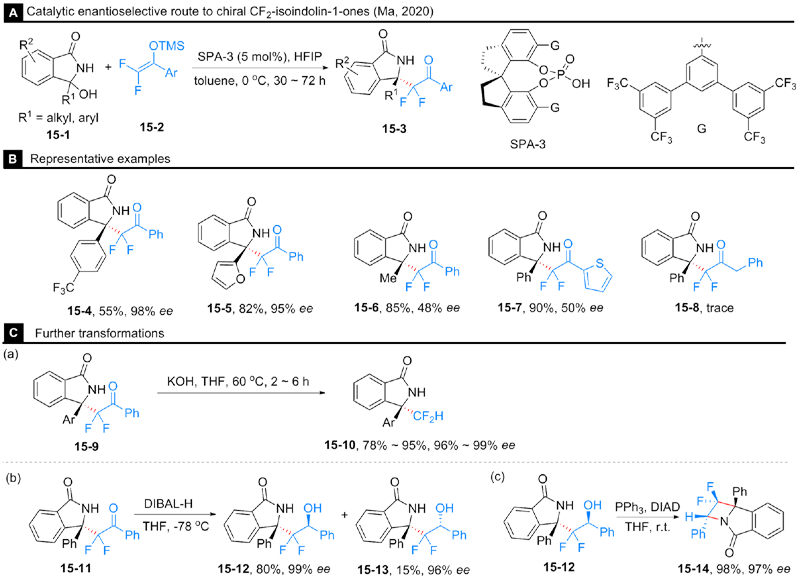
Rong, M.-Y.; Li, J.-S.; Zhou, Y.; Zhang, F.-G.; Ma, J.-A. Org. Lett. 2020, 22, 9010.
doi: 10.1021/acs.orglett.0c03406 |
| [35] |
Hao, Y.-J.; Gong, Y.; Zhou, Y.; Zhou, J.; Yu, J.-S. Org. Lett. 2020, 22, 8516.
doi: 10.1021/acs.orglett.0c03123 |
| [36] |
(a) Wencel-Delord, J.; Colobert, F. Org. Chem. Front. 2016, 3, 394.
doi: 10.1039/C5QO00398A |
|
(b) Colomer, I.; Chamberlain, A. E. R.; Haughey, M. B.; Donohoe, T. J. Nat. Rev. Chem. 2017, 1, 0088.
doi: 10.1038/s41570-017-0088 |
|
| [37] |
Li, J.; Xi, W.; Zhong, R.; Yang, J.; Wang, L.; Ding, H.; Wang, Z. Chem. Commun. 2021, 57, 1050.
doi: 10.1039/D0CC06980A |
| [38] |
(a) Zhang, K. F.; Bian, K. J.; Li, C.; Sheng, J.; Li, Y.; Wang, X. S. Angew. Chem., Int. Ed. 2019, 58, 5069.
doi: 10.1002/anie.201813184 pmid: 31964875 |
|
(b) Taj Muhammad, M.; Jiao, Y.; Ye, C.; Chiou, M.-F.; Israr, M.; Zhu, X.; Li, Y.; Wen, Z.; Studer, A.; Bao, H. Nat. Commun. 2020, 11, 416.
doi: 10.1038/s41467-019-14254-3 pmid: 31964875 |
|
| [39] |
Li, J.; Xi, W.; Liu, S.; Ruan, C.; Zheng, X.; Yang, J.; Wang, L.; Wang, Z. Org. Lett. 2021, 23, 7264.
doi: 10.1021/acs.orglett.1c02659 |
| [40] |
(a) Hu, X.-S.; He, J.-X.; Dong, S.-Z.; Zhao, Q.-H.; Yu, J.-S.; Zhou, J. Nat. Commun. 2020, 11, 5500.
doi: 10.1038/s41467-020-19387-4 |
|
(b) Chen, G.; Liu, Y. Chin. J. Org. Chem. 2021, 41, 869. (in Chinese)
doi: 10.6023/cjoc202100015 |
|
|
( 陈国术, 刘运林, 有机化学 2021, 41, 869.)
doi: 10.6023/cjoc202100015 |
|
| [41] |
Li, J.; Liu, S.; Zhong, R.; Yang, Y.; He, Y.; Yang, J.; Ma, Y.; Wang, Z. Org. Lett. 2021, 23, 5859.
doi: 10.1021/acs.orglett.1c01993 |
| [42] |
(a) Hari, D. P.; Schroll, P.; Konig, B. J. Am. Chem. Soc. 2012, 134, 2958.
doi: 10.1021/ja212099r |
|
(b) Bu, M. J.; Niu, T. F.; Cai, C. Catal. Sci. Technol. 2015, 5, 830.
doi: 10.1039/C4CY01523A |
|
| [43] |
Wu, Y.-B.; Lu, G.-P.; Zhou, B.-J.; Bu, M.-J.; Wan, L.; Cai, C. Chem. Commun. 2016, 52, 5965.
doi: 10.1039/C6CC00177G |
| [44] |
Klauck, F. J. R.; James, M. J.; Glorius, F. Angew. Chem., Int. Ed. 2017, 56, 12336.
doi: 10.1002/anie.201706896 |
| [45] |
(a) Huang, Y.; Jia, J.; Huang, Q.-P.; Zhao, L.; Wang, P.; Gu, J.; He, C.-Y. Chem. Commun. 2020, 56, 14247.
doi: 10.1039/D0CC05725H |
|
(b) Huang, Q.-P.; Huang, Y.; Wang, A.-J.; Zhao, L.; Jia, J.; Yu, Y.; Tong, J.; Gu, J.; He, C.-Y. Org. Chem. Front. 2021, 8, 4438.
doi: 10.1039/D1QO00507C |
|
| [46] |
He, F.-S.; Yao, Y.; Xie, W.; Wu, J. Chem. Commun. 2020, 56, 9469.
doi: 10.1039/D0CC03591B |
| [47] |
Song, H.; Cheng, R.; Min, Q.-Q.; Zhang, X. Org. Lett. 2020, 22, 7747.
doi: 10.1021/acs.orglett.0c02997 |
| [48] |
Zhu, X.; Huang, Y.; Xu, X.; Qing, F. Chin. Chem. Lett. 2022, 33, 817.
doi: 10.1016/j.cclet.2021.07.030 |
| [49] |
(a) Borth, P. W.; J. Econ. Entomol. 1986, 79, 1632.
doi: 10.1093/jee/79.6.1632 |
|
(b) Medebielle, M.; Ait-Mohand, S.; Burkhloder, C.; Dolbier, W. R.; Laumond, G.; Aubertin, A. M. J. Fluorine Chem. 2005, 126, 533.
doi: 10.1016/j.jfluchem.2004.12.016 |
|
| [50] |
(a) Zou, J.-Y.; Wang, Y.-Z.; Sun, W.-H.; Lin, W.-J.; Liu, X.-Y. Org. Biomol. Chem. 2021, 19, 8696.
doi: 10.1039/D1OB01530C |
|
(b) Huang, Q.-P.; Li, W.-P.; Li, R.; Zhao, L.; Wang, H.-Y.; Li, X.; Wang, P.; He, C.-Y. Tetrahedron Lett. 2022, 97, 153782.
doi: 10.1016/j.tetlet.2022.153782 |
|
| [51] |
Shi, Y.; Pan, B.-W.; He, J.-X.; Zhou, Y.; Zhou, J.; Yu, J.-S. J. Org. Chem. 2021, 86, 7797.
doi: 10.1021/acs.joc.1c00570 |
| [52] |
((a) Orsi, D. L.; Altman, R. A. Chem. Commun. 2017, 53, 7168.
doi: 10.1039/C7CC02341C |
| [53] |
((a) Hao, Y.-J..; Yu, J.-S..; Zhou, Y..; Wang, X..; Zhou, J.. Acta Chim. Sinica 2018, 76, 925. (in Chinese)
doi: 10.6023/A18080360 |
|
( 郝永佳, 余金生, 周英, 王欣, 周剑, 化学学报 2018, 76, 925.)
doi: 10.6023/A18080360 |
|
| [54] |
He, J.-X.; Zhang, Z.-H.; Mu, B.-S.; Cui, X.-Y.; Zhou, J.; Yu, J.-S. J. Org. Chem. 2021, 86, 9206.
doi: 10.1021/acs.joc.1c00754 |
| [55] |
Li, J.; Liu, S.; Zhong, R.; Yang, Y.; Xu, J.; Yang, J.; Ding, H.; Wang, Z. Org. Lett. 2021, 23, 9526.
doi: 10.1021/acs.orglett.1c03745 |
| [56] |
Li, J.; Xi, W.; Liu, S.; Yang, Y.; Yang, J.; Ding, H.; Wang, Z. Chin. Chem. Lett. 2022, 3007.
|
| [57] |
Rios, R. Chem. Soc. Rev. 2012, 41, 1060.
doi: 10.1039/C1CS15156H |
| [58] |
Wu, H.; Hong, P.; Xi, W.; Li, J. Org. Lett. 2022, 24, 2488.
doi: 10.1021/acs.orglett.2c00550 |
| [1] | Yingke Feng, He Wang, Mengxing Cui, Ran Sun, Xin Wang, Yang Chen, Lei Li. Visible-Light-Induced Difluoroalkylated Cyclization of Novel Functionalized Aromatic Isocyanides [J]. Chinese Journal of Organic Chemistry, 2023, 43(8): 2913-2925. |
| [2] | Jinxiao Zhao, Tonghui Wei, Sen Ke, Yi Li. Visible Light-Catalyzed Synthesis of Difluoroalkylated Polycyclic Indoles [J]. Chinese Journal of Organic Chemistry, 2023, 43(3): 1102-1114. |
| [3] | Zhi Tu, Jinsheng Yu, Jian Zhou. Synthesis of (Bromodifluoromethyl)trimethylsilane and Its Applications in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2023, 43(10): 3491-3507. |
| [4] | Qi Sun, Zeying Sun, Ze Yu, Guangwei Wang. Nickel-Catalyzed Stereoselective Aryl-Difluoroalkylation of Alkynes [J]. Chinese Journal of Organic Chemistry, 2022, 42(8): 2515-2520. |
| [5] | Buqing Cheng, Danhua Ge, Xin Wang, Xueqiang Chu. Perfluoroalkyl Halides as Fluorine-Containing Building Blocks for the Synthesis of Fluoroalkylated Heterocycles [J]. Chinese Journal of Organic Chemistry, 2021, 41(5): 1925-1938. |
| [6] | Jun Pan, Jingjing Wu, Fanhong Wu. Progress in Fluoroalkylation of Multicomponent [J]. Chinese Journal of Organic Chemistry, 2021, 41(3): 983-1001. |
| [7] | Zhang Ke, Xu Xiuhua, Qing Fengling. Recent Advances of Direct Trifluoromethylthiolation [J]. Chin. J. Org. Chem., 2015, 35(3): 556-569. |
| [8] | Qing Feng-Ling. Recent Advances of Trifluoromethylation [J]. Chin. J. Org. Chem., 2012, 32(05): 815-824. |
| [9] | CHEN, Yanga ; SHUI, Sheng-Xiaa; FANG, Xianga; WU, Fang-Hong*,a,b. Application of β-Alkoxyvinyl Trifluoromethyl Ketones to the Synthesis of Heterocycles [J]. Chin. J. Org. Chem., 2008, 28(12): 2023-2038. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||