Chinese Journal of Organic Chemistry ›› 2026, Vol. 46 ›› Issue (3): 1027-1038.DOI: 10.6023/cjoc202508021 Previous Articles Next Articles
ARTICLES
韦娜娜, 郭婉真, 鲁星, 任志强, 马豪杰, 张玉琦, 王记江, 韩波*( )
)
收稿日期:2025-08-25
修回日期:2025-10-26
发布日期:2025-11-27
基金资助:
Nana Wei, Wanzhen Guo, Xing Lu, Zhiqiang Ren, Haojie Ma, Yuqi Zhang, Jijiang Wang, Bo Han*( )
)
Received:2025-08-25
Revised:2025-10-26
Published:2025-11-27
Contact:
*E-mail: Supported by:Share
Nana Wei, Wanzhen Guo, Xing Lu, Zhiqiang Ren, Haojie Ma, Yuqi Zhang, Jijiang Wang, Bo Han. Magnesium-Promoted Selectively Reduction of Unprotected Indoles and Quinoxalines[J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 1027-1038.
| Entry | Catalyst (mol%) | AB/equiv. | Solvent | Temp./℃ | 2a/% |
|---|---|---|---|---|---|
| 1 | MgCl2 (10) | 2.0 | Toluene | 80 | 49 |
| 2 | MgSO4 (10) | 2.0 | Toluene | 80 | 52 |
| 3 | Mg(OTf)2 (10) | 2.0 | Toluene | 80 | 40 |
| 4 | MgI2 (10) | 2.0 | Toluene | 80 | 37 |
| 5 | Mg (50) | 2.0 | Toluene | 80 | 75 |
| 6 | Zn (50) | 2.0 | Toluene | 80 | 50 |
| 7 | Al (50) | 2.0 | Toluene | 80 | 40 |
| 8 | Mn (50) | 2.0 | Toluene | 80 | 45 |
| 9 | Mg (50) | 2.0 | Et2O | 80 | 24 |
| 10 | Mg (50) | 2.0 | THF | 80 | ndb |
| 11 | Mg (50) | 2.0 | CH2Cl2 | 80 | 43 |
| 12 | Mg (50) | 2.0 | CH3CN | 80 | Trace |
| 13 | Mg (50) | 2.0 | Toluene | 100 | 77 |
| 14 | Mg (50) | 2.0 | Toluene | 120 | 78 |
| 15 | Mg (50) | 3.0 | Toluene | 80 | 85 |
| 16 | Mg (50) | 4.0 | Toluene | 80 | 83 |
| 17c | Mg (50) | 3.0 | Toluene | 80 | 71 |
| 18 | Mg (40) | 3.0 | Toluene | 80 | 80 |
| 19 | Mg (30) | 3.0 | Toluene | 80 | 77 |
| 20 | — | 3.0 | Toluene | 80 | 14 |
| Entry | Catalyst (mol%) | AB/equiv. | Solvent | Temp./℃ | 2a/% |
|---|---|---|---|---|---|
| 1 | MgCl2 (10) | 2.0 | Toluene | 80 | 49 |
| 2 | MgSO4 (10) | 2.0 | Toluene | 80 | 52 |
| 3 | Mg(OTf)2 (10) | 2.0 | Toluene | 80 | 40 |
| 4 | MgI2 (10) | 2.0 | Toluene | 80 | 37 |
| 5 | Mg (50) | 2.0 | Toluene | 80 | 75 |
| 6 | Zn (50) | 2.0 | Toluene | 80 | 50 |
| 7 | Al (50) | 2.0 | Toluene | 80 | 40 |
| 8 | Mn (50) | 2.0 | Toluene | 80 | 45 |
| 9 | Mg (50) | 2.0 | Et2O | 80 | 24 |
| 10 | Mg (50) | 2.0 | THF | 80 | ndb |
| 11 | Mg (50) | 2.0 | CH2Cl2 | 80 | 43 |
| 12 | Mg (50) | 2.0 | CH3CN | 80 | Trace |
| 13 | Mg (50) | 2.0 | Toluene | 100 | 77 |
| 14 | Mg (50) | 2.0 | Toluene | 120 | 78 |
| 15 | Mg (50) | 3.0 | Toluene | 80 | 85 |
| 16 | Mg (50) | 4.0 | Toluene | 80 | 83 |
| 17c | Mg (50) | 3.0 | Toluene | 80 | 71 |
| 18 | Mg (40) | 3.0 | Toluene | 80 | 80 |
| 19 | Mg (30) | 3.0 | Toluene | 80 | 77 |
| 20 | — | 3.0 | Toluene | 80 | 14 |
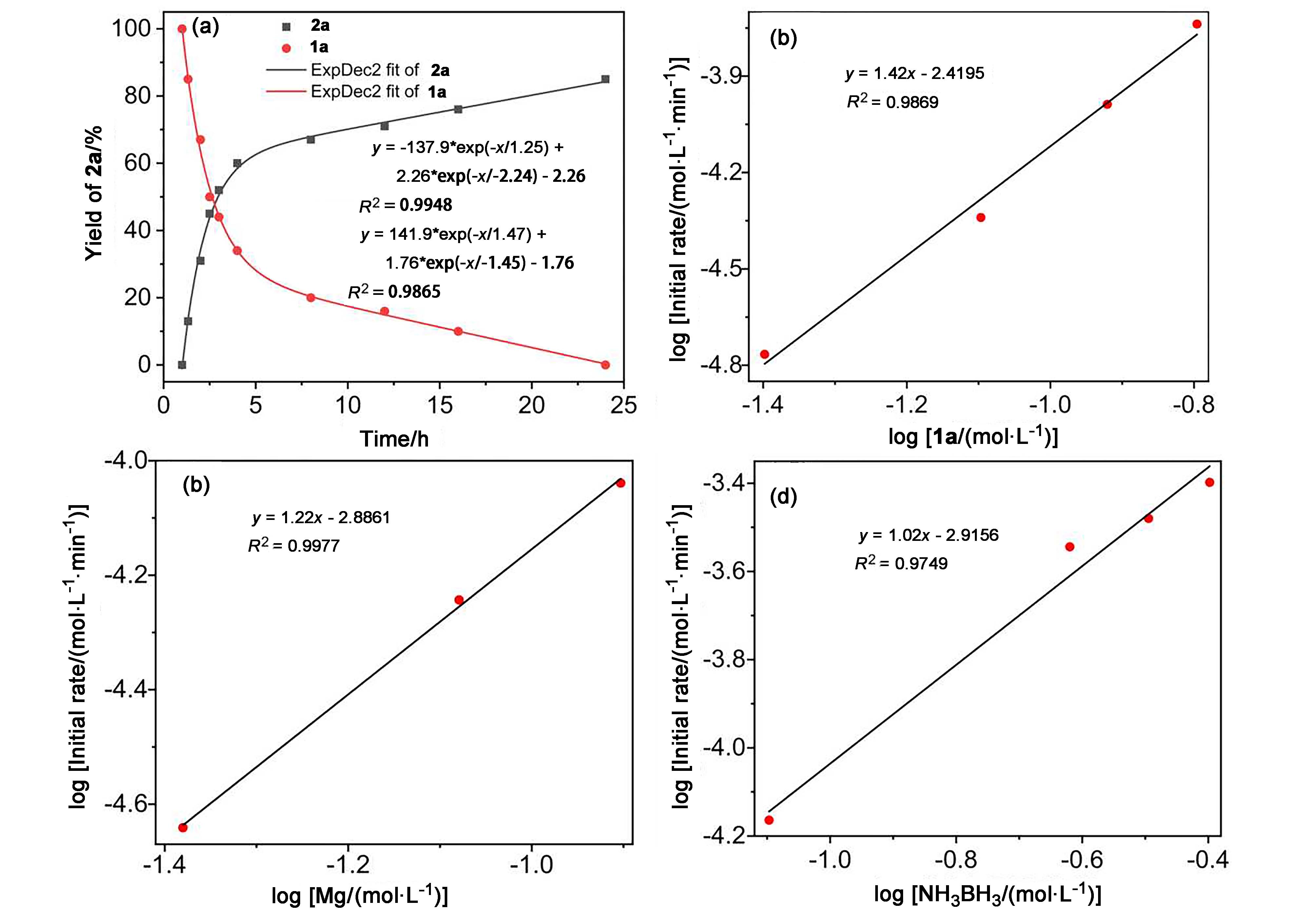
| [1] |
(a)
doi: 10.1021/cr200328h pmid: 26577114 |
|
(b)
doi: 10.1039/d3cs00329a pmid: 26577114 |
|
|
(c)
doi: 10.1002/anie.v60.24 pmid: 26577114 |
|
|
(d)
doi: 10.1002/anie.201507945 pmid: 26577114 |
|
| [2] |
(a)
doi: 10.1021/cr100307m |
|
(b)
doi: 10.1021/acs.joc.3c01767 |
|
|
(c)
doi: 10.1016/S1872-2067(19)63336-X |
|
| [3] |
doi: 10.3390/catal12101260 |
| [4] |
(a)
doi: 10.1039/D0CY00582G pmid: 21902212 |
|
(b)
doi: 10.1002/cctc.v12.18 pmid: 21902212 |
|
|
(c)
doi: 10.1039/D1NJ03760A pmid: 21902212 |
|
|
(d)
doi: 10.1021/acs.orglett.0c01905 pmid: 21902212 |
|
|
(e)
doi: 10.1021/acs.orglett.8b00312 pmid: 21902212 |
|
|
(f)
doi: 10.1021/ol2019899 pmid: 21902212 |
|
|
(g)
doi: 10.1002/anie.v55.44 pmid: 21902212 |
|
| [5] |
(a)
doi: 10.1039/C4CC09471A |
|
(b)
doi: 10.1002/adsc.v358.22 |
|
| [6] |
(a)
doi: 10.1021/acs.chemrev.7b00182 |
|
(b)
doi: 10.1021/acs.accounts.4c00009 |
|
| [7] |
doi: 10.1016/j.tetlet.2004.02.123 |
| [8] |
(a)
doi: 10.1002/chem.v21.14 |
|
(b)
doi: 10.1002/adsc.v357.16/17 |
|
| [9] |
(a)
doi: 10.1002/anie.v46:24 |
|
(b)
doi: 10.1002/anie.v51.45 |
|
| [10] |
(a)
doi: 10.1016/j.jorganchem.2008.02.004 pmid: 31062984 |
|
(b)
doi: 10.1021/acs.orglett.9b01055 pmid: 31062984 |
|
|
(c)
doi: 10.6023/cjoc202110041 pmid: 31062984 |
|
|
(张苗苗, 韩波, 马豪杰, 赵亮, 王记江, 张玉琦, 有机化学, 2022, 42, 1170.)
doi: 10.6023/cjoc202110041 pmid: 31062984 |
|
| [11] |
doi: 10.1021/acscatal.1c02876 |
| [12] |
(a)
doi: 10.1002/anie.200703150 pmid: 20672860 |
|
(b)
doi: 10.1039/b800312m pmid: 20672860 |
|
|
(c)
doi: 10.1021/cr100088b pmid: 20672860 |
|
| [13] |
(a)
pmid: 25343603 |
|
(b)
doi: 10.1038/nchem.2063 pmid: 25343603 |
|
| [14] |
doi: 10.1039/c0cc03163a |
| [15] |
doi: 10.1021/jacs.6b04271 |
| [16] |
doi: 10.1002/anie.v55.47 |
| [17] |
doi: 10.1021/acs.joc.2c01744 |
| [18] |
(a)
doi: 10.1021/acs.orglett.2c03326 |
|
(b)
doi: 10.1002/cjoc.v43.1 |
|
| [19] |
doi: 10.1021/jacs.6b07245 |
| [20] |
doi: 10.1021/acs.orglett.6b02610 |
| [21] |
doi: 10.1021/jacs.9b00594 |
| [22] |
doi: 10.1002/cjoc.v42.15 |
| [23] |
doi: 10.1021/acs.orglett.4c02090 pmid: 38976355 |
| [24] |
doi: 10.1021/acs.organomet.0c00091 |
| [25] |
doi: 10.1039/D1QO00672J |
| [26] |
doi: 10.1021/acs.orglett.3c04051 |
| [27] |
(a)
doi: 10.1021/acs.chemrev.1c00641 pmid: 35254061 |
|
(b)
doi: 10.1016/j.chempr.2019.02.002 pmid: 35254061 |
|
| [28] |
(a)
doi: 10.1002/chem.v15:2 |
|
(b)
doi: 10.1021/jo100763f |
|
|
(c)
doi: 10.1002/anie.v50.42 |
|
| [29] |
(a)
doi: 10.1021/acs.chemrev.6b00043 |
|
(b)
doi: 10.1039/C5CS00880H |
|
| [30] |
(a)
doi: 10.1039/c2cc30565h pmid: 27271460 |
|
(b)
doi: 10.1002/chem.201203190 pmid: 27271460 |
|
|
(c)
doi: 10.1039/C3SC52793J pmid: 27271460 |
|
|
(d)
doi: 10.1002/chem.201601623 pmid: 27271460 |
|
|
(e)
doi: 10.1021/jacs.7b06719 pmid: 27271460 |
|
| [31] |
doi: 10.1039/C6CC06805G |
| [32] |
doi: 10.1021/jacs.6b03689 |
| [33] |
doi: 10.1002/anie.201902188 pmid: 30977970 |
| [34] |
doi: 10.1021/acs.orglett.9b03131 |
| [35] |
doi: 10.6023/cjoc202208042 |
|
(张苗苗, 陈荣, 焦红梅, 马豪杰, 韩波, 张玉琦, 王记江, 有机化学, 2023, 43, 1462.)
doi: 10.6023/cjoc202208042 |
|
| [36] |
(a)
doi: 10.1021/acs.joc.3c02954 |
|
(b)
doi: 10.1021/ja200723n |
|
|
(c)
doi: 10.1021/acs.orglett.5c00997 |
|
| [37] |
(a)
doi: 10.1002/adsc.v364.21 pmid: 16330129 |
|
(b)
doi: 10.1021/acs.orglett.0c01273 pmid: 16330129 |
|
|
(c)
doi: 10.1021/acs.orglett.0c02128 pmid: 16330129 |
|
|
(d)
pmid: 16330129 |
| [1] | Jialong Xu, Quan Kong, Jianhui Chen, Mingyue Zhang, Xiaoming Ji, Biao Cheng. Recent Advances in Low-Valent Tungsten-Catalyzed Organic Reactions [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 759-772. |
| [2] | Xiaoxiang Xi, Ming Gao, Libiao Han. Rhodium-Catalyzed Reduction of RS—SR to Thiophenols/Thiols with H2 [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 859-865. |
| [3] | Huiyang Liu, Du Chen, Yijin Su, Peng Zhang, Chao Liu. Rapid and Controlled Reduction of Acyl Chlorides to Aldehydes Using Pinacolborane [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 564-569. |
| [4] | Shunli Du, Yaya Wang, Jiaming Guo, Xuewei Xu, Xinhua Peng. Study on the Borylation of β-C Positions of Aryl Olefins Catalyzed by Copper Organophosphorus Complexes [J]. Chinese Journal of Organic Chemistry, 2025, 45(7): 2435-2443. |
| [5] | Zhen Liang, Weiyan Xu, Yi Chen, Huayu Qiu, Yezhe Zhao, Jiabin Shen, Min Wang. Iridium-Catalyzed Synthesis of Indole Derivatives from N-Aryl-2-aminopyridines and Vinylene Carbonate [J]. Chinese Journal of Organic Chemistry, 2025, 45(6): 2149-2156. |
| [6] | Jie Jiang, Jiali Li, Ruohan Pan, Yu Chen, Jiale Liu, Yucai Tang. Photoinduced Difluoromethylation/Cyclization of 2-Aryl Indoles with HCF2SO2Na [J]. Chinese Journal of Organic Chemistry, 2025, 45(4): 1239-1248. |
| [7] | Yepeng Xu, Yi Ruan, Jianfeng Zheng, Peiqiang Huang. Synthesis of (–)-Sedacryptine and (–)-Geissman-Waiss Lactone by Applying Methods for the Direct Transformation of Amides [J]. Chinese Journal of Organic Chemistry, 2025, 45(3): 988-995. |
| [8] | Xueying Yang, Yuanshuang Xu, Xinying Zhang, Xuesen Fan. Synthesis of C2-Spiroindolines Based on the Cascade Reaction of 2-Aryl-3H-indoles with Cyclopropanols [J]. Chinese Journal of Organic Chemistry, 2025, 45(2): 694-706. |
| [9] | Dengpeng Xia, Qi Wu, Zhihua Cai, Guangfen Du. Selective Synthesis of 3-Selenoindoles via Selenation of Indoles under Catalyst-Free Condition [J]. Chinese Journal of Organic Chemistry, 2025, 45(1): 349-357. |
| [10] | Mengna Yang, Yucai Tang, Jie Jiang, Jiali Li, Ruohan Pan, Yu Chen, Jinglin Duan, Songbai Zhang. Copper(I)-Catalyzed Acylation/Cyclization of 2-Aryl-N-acryloyl Indole toward Indolo[2,1-α]isoquinoline Derivatives [J]. Chinese Journal of Organic Chemistry, 2025, 45(1): 307-318. |
| [11] | Junyong Wang, Na Li, Jie Ke, Chuan He. Recent Advances in Electrochemical Silylation [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 927-939. |
| [12] | Hongbing Chen, Sijia Yang, Zhipeng Ye, Kai Chen, Haoyue Xiang, Hua Yang. Electrocatalytic Reduction of Quinolines and Ketones by Using Lewis Base-Ligated Borane as a Hydrogen Donor [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 966-971. |
| [13] | Jian Huang, Wenzhen Zhang. Advances in Electrochemical Cathodic Reductive Reactions Involving Carbon-Nitrogen Bonds [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 825-839. |
| [14] | Fangying Duan, Menglei Yuan, Jian Zhang. Paired Electrolysis for Inorganic Small Molecules Reduction Coupled with Alternative Oxidation Reactions [J]. Chinese Journal of Organic Chemistry, 2024, 44(3): 809-824. |
| [15] | Jing Xiao, Yuxiang Xie, Libiao Han. Advances in Organic Reactions Using Phosphorus Acid as Reducing Agent [J]. Chinese Journal of Organic Chemistry, 2024, 44(12): 3702-3712. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||