Chinese Journal of Organic Chemistry ›› 2026, Vol. 46 ›› Issue (4): 1730-1738.DOI: 10.6023/cjoc202512050 Previous Articles Next Articles
ARTICLES
收稿日期:2025-12-21
修回日期:2026-02-05
发布日期:2026-03-06
通讯作者:
庞利娟, 黄华文
基金资助:
Yixi Zhanga, Lijuan Pangb,*( ), Huawen Huanga,c,*(
), Huawen Huanga,c,*( )
)
Received:2025-12-21
Revised:2026-02-05
Published:2026-03-06
Contact:
Lijuan Pang, Huawen Huang
Supported by:Share
Yixi Zhang, Lijuan Pang, Huawen Huang. Visible-Light-Induced Aerobic Dehydrogenative Couplings of Quinoxalin-2(1H)-ones with Aldehydes[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1730-1738.
| Entry | Additive (mol%) | H2O/mol% | Solvent | Yielda/% |
|---|---|---|---|---|
| 1 | HBr (25) | 2.0 | DCE | 92 |
| 2c | — | — | DCE | 0 |
| 3d | HBr (25) | — | DCE | 65 |
| 4 | HBr (25) | — | DCE | 84 |
| 5 | HBr (25) | 1.0 | DCE | 89 |
| 6 | HBr (25) | 3.0 | DCE | 88 |
| 7 | HBr (25) | 5.0 | DCE | 62 |
| 8 | HBr (25) | 10.0 | DCE | 51 |
| 9 | HBr (25) | — | DCM | 20 |
| 10 | HBr (25) | — | Acetone | 0 |
| 11 | HBr (25) | — | H2O | 45 |
| 12 | HBr (25) | — | CHCl3 | 53 |
| 13 | HBr (25) | — | MeCN | 35 |
| 14 | TMSCl (25) | — | DCE | 51 |
| 15 | MsOH (25) | — | DCE | 30 |
| 16 | TMSBr (25) | — | DCE | 23 |
| 17 | CF3SO3H (25) | — | DCE | 33 |
| 18 | NaBr (25) | — | DCE | 23 |
| 19 | NaCl (25) | — | DCE | 15 |
| 20 | LiCl (25) | — | DCE | 10 |
| 21 | HBr (20) | — | DCE | 75 |
| 22 | HBr (30) | — | DCE | 91 |
| 23e | HBr (25) | — | DCE | 0 |
| 24f | HBr (25) | — | DCE | Trace |
| 25g | HBr (25) | ___ | DCE | Trace |
| Entry | Additive (mol%) | H2O/mol% | Solvent | Yielda/% |
|---|---|---|---|---|
| 1 | HBr (25) | 2.0 | DCE | 92 |
| 2c | — | — | DCE | 0 |
| 3d | HBr (25) | — | DCE | 65 |
| 4 | HBr (25) | — | DCE | 84 |
| 5 | HBr (25) | 1.0 | DCE | 89 |
| 6 | HBr (25) | 3.0 | DCE | 88 |
| 7 | HBr (25) | 5.0 | DCE | 62 |
| 8 | HBr (25) | 10.0 | DCE | 51 |
| 9 | HBr (25) | — | DCM | 20 |
| 10 | HBr (25) | — | Acetone | 0 |
| 11 | HBr (25) | — | H2O | 45 |
| 12 | HBr (25) | — | CHCl3 | 53 |
| 13 | HBr (25) | — | MeCN | 35 |
| 14 | TMSCl (25) | — | DCE | 51 |
| 15 | MsOH (25) | — | DCE | 30 |
| 16 | TMSBr (25) | — | DCE | 23 |
| 17 | CF3SO3H (25) | — | DCE | 33 |
| 18 | NaBr (25) | — | DCE | 23 |
| 19 | NaCl (25) | — | DCE | 15 |
| 20 | LiCl (25) | — | DCE | 10 |
| 21 | HBr (20) | — | DCE | 75 |
| 22 | HBr (30) | — | DCE | 91 |
| 23e | HBr (25) | — | DCE | 0 |
| 24f | HBr (25) | — | DCE | Trace |
| 25g | HBr (25) | ___ | DCE | Trace |

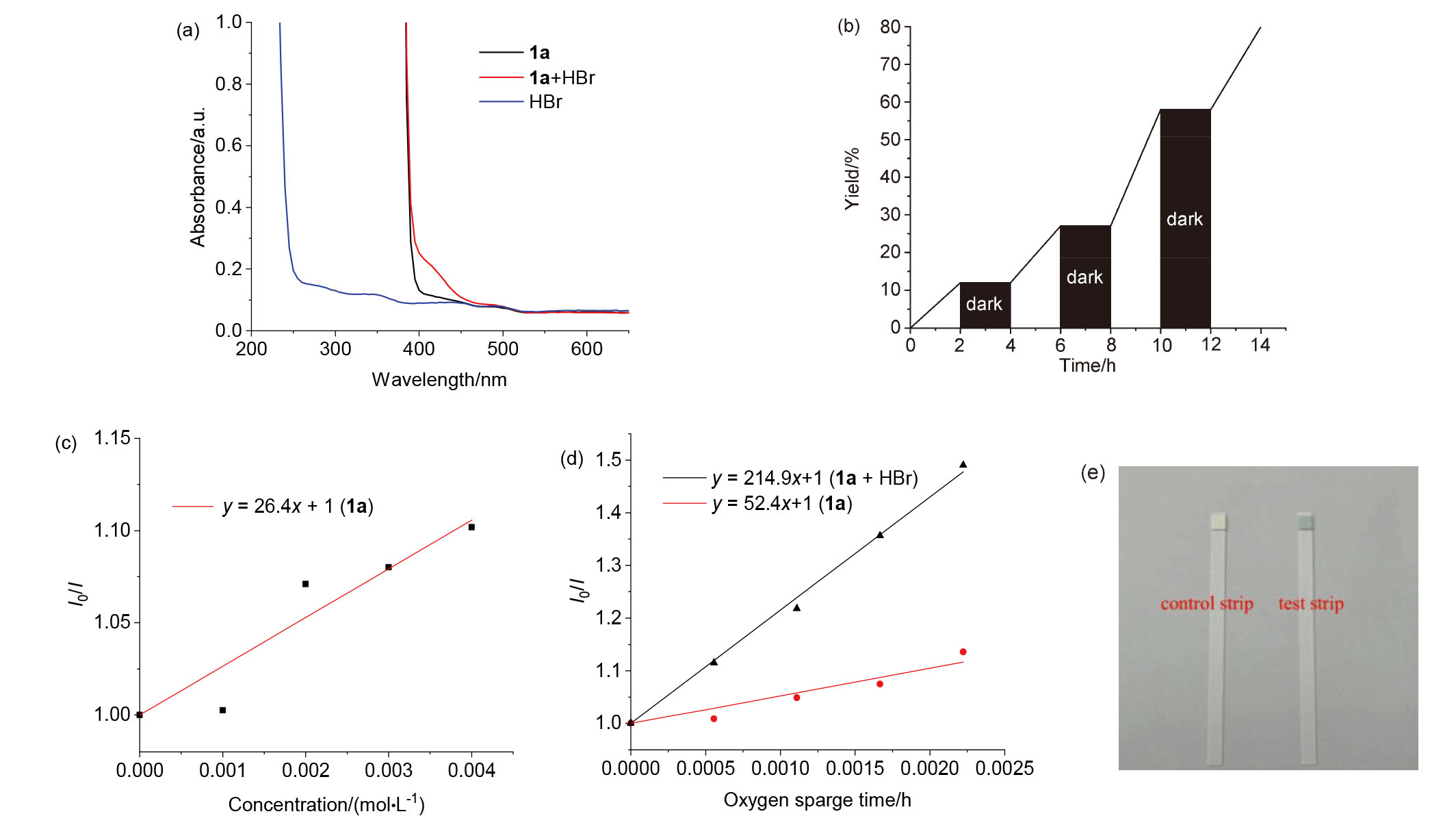
| [1] |
|
| [2] |
|
| [3] |
|
|
(董江湖, 宣良明, 王池, 赵晨熙, 王海峰, 严琼姣, 汪伟, 陈芬儿, 有机化学, 2024, 44, 111.)
|
|
| [4] |
|
|
(李文雅, 王煜, 陈江琦, 史丹, 张良, 余小春, 王正军, 有机化学, 2024, 44, 2110 )
|
|
| [5] |
|
| [6] |
|
| [7] |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
|
| [12] |
|
| [13] |
|
| [14] |
|
| [15] |
|
| [16] |
|
| [17] |
|
| [18] |
|
| [19] |
|
| [20] |
|
| [21] |
|
| [22] |
|
| [23] |
|
| [24] |
|
| [25] |
|
| [26] |
|
| [27] |
|
| [28] |
|
| [29] |
|
| [30] |
|
| [31] |
|
| [32] |
Kiran; Rani, P.; Chahal, S.; Sindhu, J.; Kumar, S.; Varma, R. S.; Singh, R. New J. Chem. 2021, 45, 18722.
|
| [33] |
|
| [34] |
|
| [35] |
|
| [36] |
|
| [37] |
|
| [38] |
|
| [39] |
|
| [40] |
|
| [41] |
|
| [42] |
|
| [43] |
|
| [44] |
|
| [1] | Fu Ye, Weiming Yuan. Recent Advances in Dicarbofunctionalization of Alkynes via Photoredox Catalysis [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1146-1165. |
| [2] | Lingxu Meng, Lin Shen, Xiangling Luo, Yumei Lin, Lei Gong. Design of Photoactive Cr- and Co-Based Complexes and Their Applications in Organic Synthesis [J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1513-1528. |
| [3] | Xianting Huang, Xiaoqian Shen, Qingxu Wang, Zhongquan Liu. A Catalyst and Solvent-Free Photochemical Acylation of N-Heteroarenes with Keto Acids [J]. Chinese Journal of Organic Chemistry, 2026, 46(3): 951-960. |
| [4] | Liangxin Fan, Mengyao Shi, Xingyuan Liu, Zhenliang Pan, Guoyu Yang, Lulu Wu, Xiaoping Xue, Li Dai, Cuilian Xu. Synthesis of N-Ester Carbazoles Using Diethyl Azodicarboxylate as Carbon Source [J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 612-623. |
| [5] | Yinsheng Miao, Lei Zhang, Jianqi Chen, Lili Wang, Zheng Duan. Synthesis of Pyrazolo(iso)quinoline Derivatives from Aryl Alkynes and N-Imine(iso)quinoline Ylides Catalyzed by Bisphosphonium Salt [J]. Chinese Journal of Organic Chemistry, 2025, 45(8): 2876-2884. |
| [6] | Genwei Gao, Zhen Li, Yan Li, Xi Lu. Recent Progress on the Construction of C(sp2)—C(sp3) Bonds via Photoredox/Nickel Dual Catalysis [J]. Chinese Journal of Organic Chemistry, 2025, 45(6): 1905-1919. |
| [7] | Hui Li, Ablimit Abdukader, Lei Zhou. Radical Denitrogenative Ring Opening of N-Benzyl-benzotriazoles for the Synthesis of 6-Substituted Phenanthridines under Visible Light Conditions [J]. Chinese Journal of Organic Chemistry, 2025, 45(5): 1770-1777. |
| [8] | Feng Lin, Yan Zhang, Ming Wu, Huiyan Liu, Wen-Juan Hao, Bo Jiang. Visible-Light-Induced Annulative Acylative Difunctionalization of 1,6-Enynes for Accessing 1-Indanones [J]. Chinese Journal of Organic Chemistry, 2025, 45(5): 1729-1738. |
| [9] | Yang Hong, Hongping Deng. Research Progress on Photocatalyzed Protic C(sp3)—HFunctionalization [J]. Chinese Journal of Organic Chemistry, 2025, 45(5): 1569-1590. |
| [10] | Chen Li, Yi Jiao, Xiaoran Shi, Yiqiang Yang, Shouyun Yu. Photoredox-Catalyzed C-Glycosylation Reactions [J]. Chinese Journal of Organic Chemistry, 2025, 45(5): 1423-1459. |
| [11] | Mengna Yang, Yucai Tang, Jie Jiang, Jiali Li, Ruohan Pan, Yu Chen, Jinglin Duan, Songbai Zhang. Copper(I)-Catalyzed Acylation/Cyclization of 2-Aryl-N-acryloyl Indole toward Indolo[2,1-α]isoquinoline Derivatives [J]. Chinese Journal of Organic Chemistry, 2025, 45(1): 307-318. |
| [12] | Wenduo Li, Na'na Wei, Nan Feng. Boryl Radical-Promoted Synthesis of Biaryls and Benzylcarboxylic Acids Methyl Ester via C—C Bond Formation Reactions [J]. Chinese Journal of Organic Chemistry, 2024, 44(6): 1853-1861. |
| [13] | Jiyu Liu, Shengyu Li, Kuan Chen, Yin Zhu, Yuan Zhang. Triphenylamine-Based Ordered Mesoporous Polymer as a Metal-Free Photocatalyst for Oxidation of Thiols to Disulfide [J]. Chinese Journal of Organic Chemistry, 2024, 44(2): 605-612. |
| [14] | Yanyan Zhang, Zhuzhu Zhang, Shengqing Zhu, Lingling Chu. Recent Advances in Nickel Catalyzed Asymmetric Acylation Reactions [J]. Chinese Journal of Organic Chemistry, 2023, 43(3): 1023-1035. |
| [15] | Hongyu Hou, Yuanyuan Cheng, Bin Chen, Chenho Tung, Lizhu Wu. α-Acylation of Olefins via Photocatalysis [J]. Chinese Journal of Organic Chemistry, 2023, 43(3): 1012-1022. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||