化学学报 ›› 2026, Vol. 84 ›› Issue (3): 341-352.DOI: 10.6023/A25110367 上一篇 下一篇
研究论文
赵馨雨a,b, 韩燕楠c, 徐吉磊b,*( ), 安庆大a,*(
), 安庆大a,*( ), 肖作毅a, 苏鑫b, 黄家辉b,*(
), 肖作毅a, 苏鑫b, 黄家辉b,*( )
)
投稿日期:2025-11-12
发布日期:2026-01-20
基金资助:
Zhao Xinyua,b, Han Yannanc, Xu Jileib,*( ), An Qingdaa,*(
), An Qingdaa,*( ), Xiao Zuoyia, Su Xinb, Huang Jiahuib,*(
), Xiao Zuoyia, Su Xinb, Huang Jiahuib,*( )
)
Received:2025-11-12
Published:2026-01-20
Contact:
*E-mail: xujilei@dicp.ac.cn;
anqingda@dlpu.edu.cn;
jiahuihuang@dicp.ac.cn
Supported by:文章分享
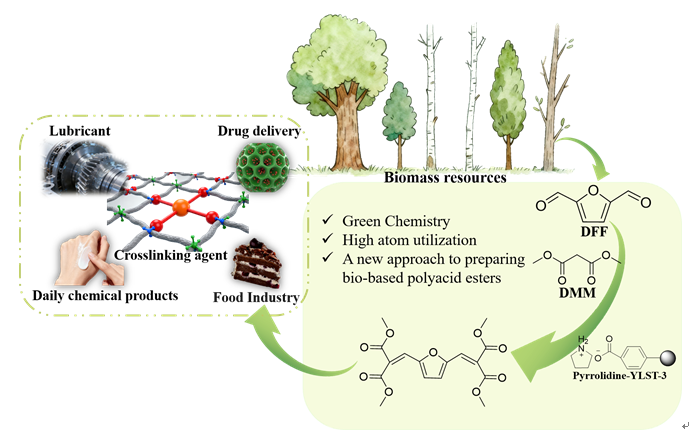
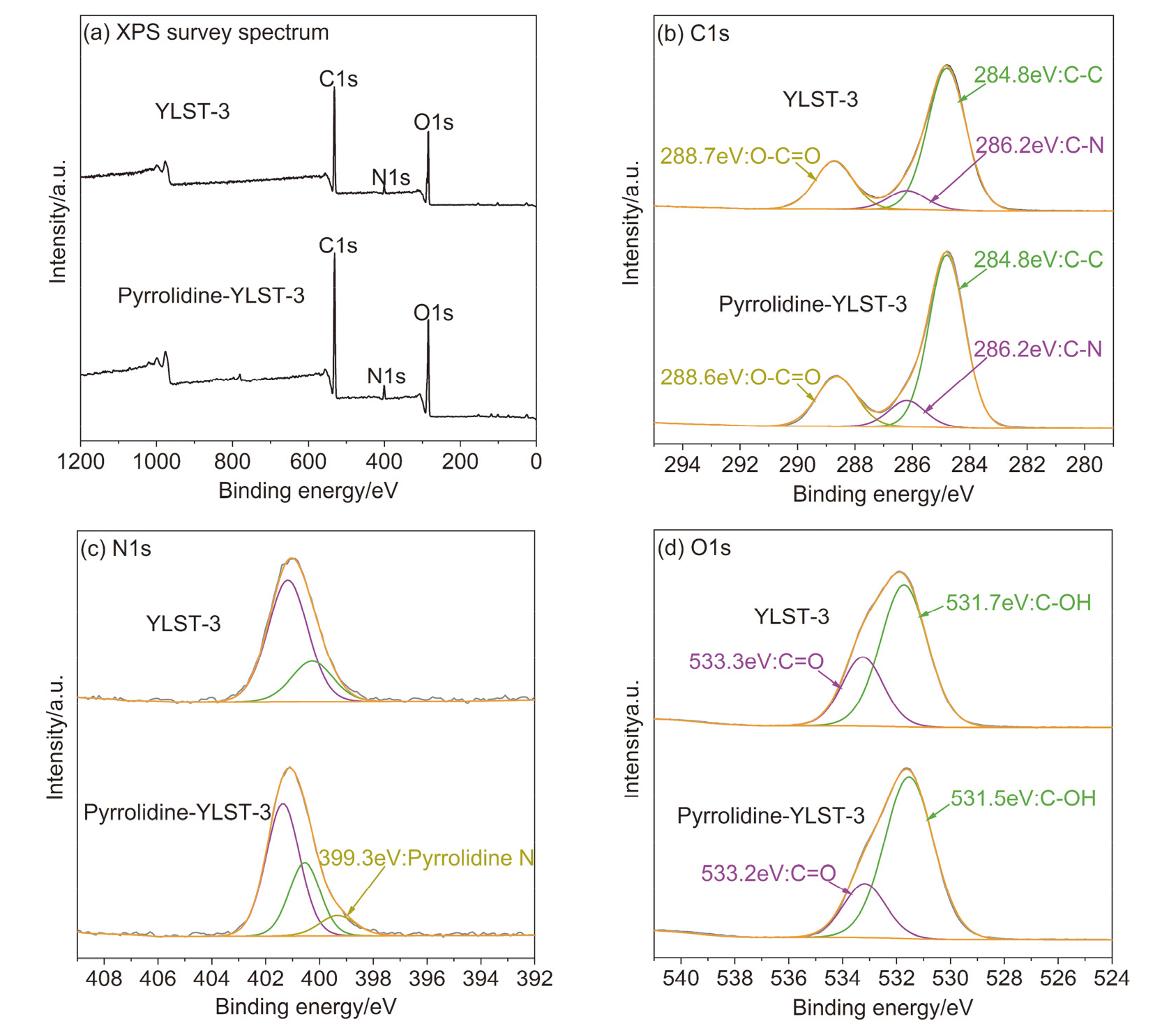
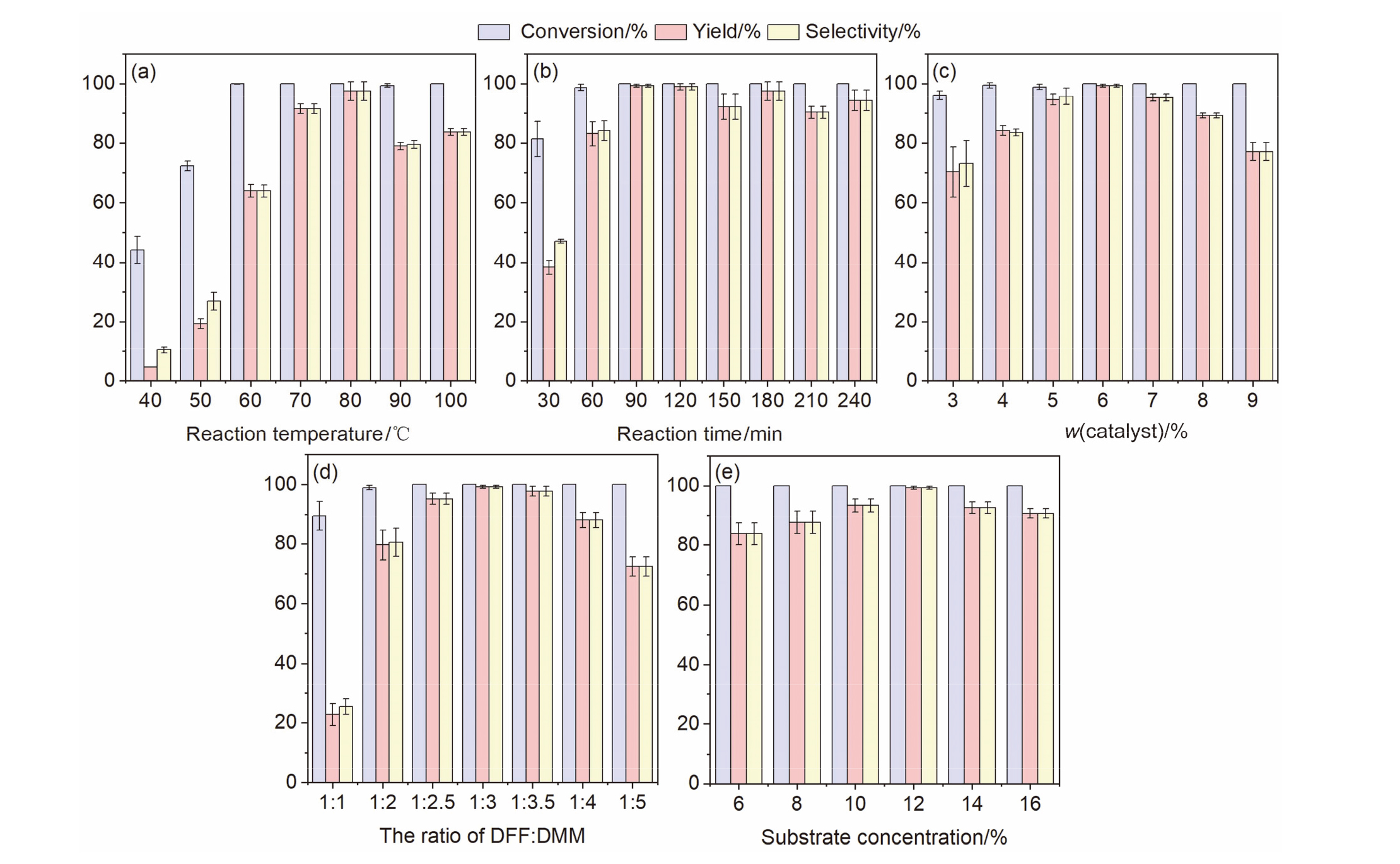
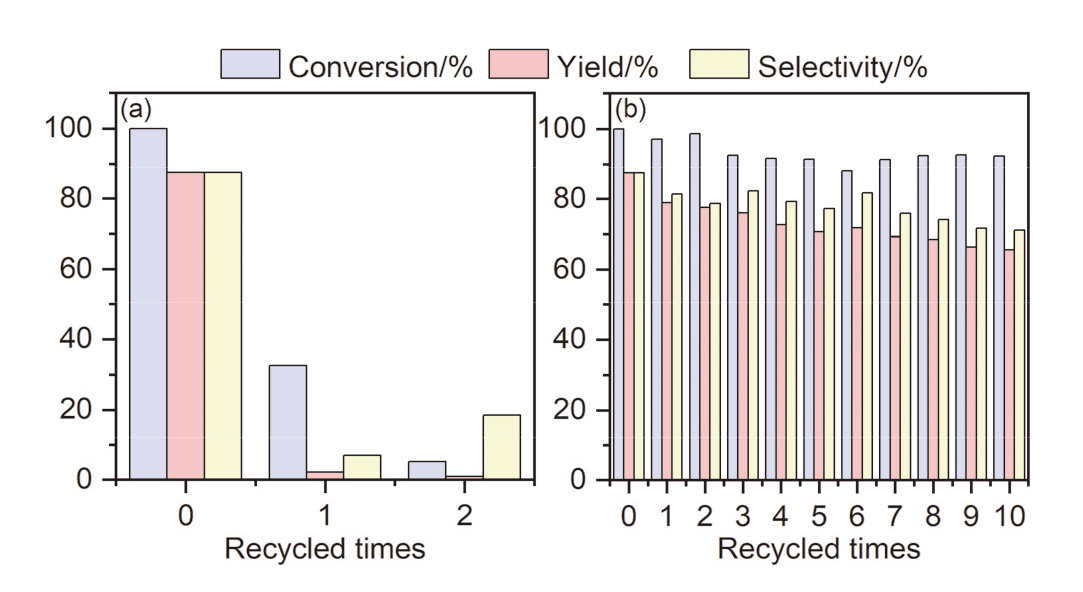
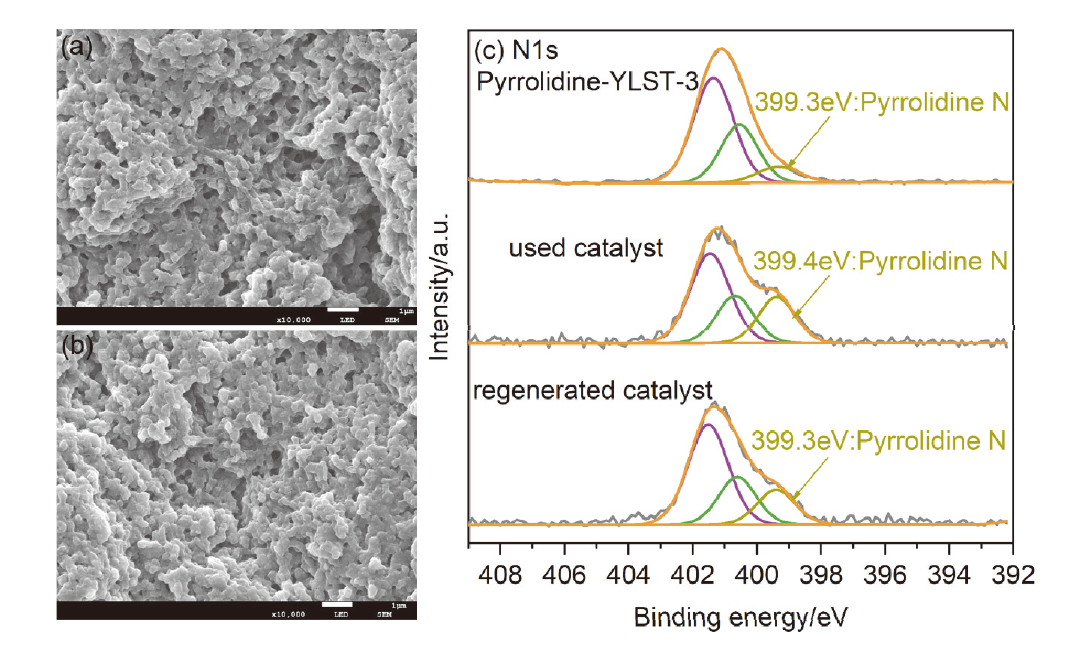
本工作以弱酸性离子交换树脂为载体, 以有机胺为修饰剂, 通过简单的酸碱中和反应制备得到了一系列负载型离子液体催化剂, 并将其应用到生物基多元酸酯的制备当中. 以2,5-呋喃二甲醛(DFF)和丙二酸二甲酯(DMM)为底物, 以活性最佳的Pyrrolidine-YLST-3为催化剂时, 生物基多元酸酯的产率可以达到99.2%. 这种制备方法极大地减少了传统氧化过程中氧气等强氧化剂的使用, 安全性更高, 对设备要求较低. 此外, 目标产物选择性较高, 副产物主要为水, 对环境更加友好, 更加符合绿色化学原则. 通过核磁共振(NMR)、红外光谱(FTIR)、X射线衍射(XRD)和X射线光电子能谱(XPS)等方法对催化剂进行了表征, 发现吡咯烷与离子交换树脂之间发生了质子转移, 形成了新的负载型离子液体. 通过对多种底物的Knoevenagel缩合反应进行研究均取得了较高的收率, 说明Pyrrolidine-YLST-3催化剂具有很强的普适性.
赵馨雨, 韩燕楠, 徐吉磊, 安庆大, 肖作毅, 苏鑫, 黄家辉. 负载型离子液体催化生物基多元酸酯的制备[J]. 化学学报, 2026, 84(3): 341-352.
Zhao Xinyu, Han Yannan, Xu Jilei, An Qingda, Xiao Zuoyi, Su Xin, Huang Jiahui. Preparation of Bio-based Polyacid Esters Catalyzed by Supported Ionic Liquids[J]. Acta Chimica Sinica, 2026, 84(3): 341-352.
| Entry | Catalyst | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | Pyrrolidine-YLST-3 | 100 | 79.1 | 79.1 |
| 2 | Pyrrolidine-D113 | 100 | 63.8 | 63.8 |
| 3 | Ethanolamine-YLST-3 | 97.0 | 60.0 | 61.5 |
| 4 | Ethanolamine-D113 | 99.2 | 73.4 | 74.0 |
| 5 | Diethanolamine-YLST-3 | 11.6 | 0 | 0 |
| 6 | Diethanolamine-D113 | 9.9 | 0 | 0 |
| 7b | Pyrrolidine-YLST-3 | 92.7 | 68.9 | 74.4 |
| 8b | Ethanolamine-D113 | 98.7 | 58.8 | 59.5 |
| 9 | YLST-3 | 18.2 | 0 | 0 |
| 10 | Pyrrolidine | 100 | 19.4 | 19.4 |
| Entry | Catalyst | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | Pyrrolidine-YLST-3 | 100 | 79.1 | 79.1 |
| 2 | Pyrrolidine-D113 | 100 | 63.8 | 63.8 |
| 3 | Ethanolamine-YLST-3 | 97.0 | 60.0 | 61.5 |
| 4 | Ethanolamine-D113 | 99.2 | 73.4 | 74.0 |
| 5 | Diethanolamine-YLST-3 | 11.6 | 0 | 0 |
| 6 | Diethanolamine-D113 | 9.9 | 0 | 0 |
| 7b | Pyrrolidine-YLST-3 | 92.7 | 68.9 | 74.4 |
| 8b | Ethanolamine-D113 | 98.7 | 58.8 | 59.5 |
| 9 | YLST-3 | 18.2 | 0 | 0 |
| 10 | Pyrrolidine | 100 | 19.4 | 19.4 |
| Entry | Solvent | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | ethyl acetate | 100 | 89.9 | 89.9 |
| 2 | tetrahydrofuran | 99.3 | 81.1 | 81.7 |
| 3 | dichloromethane | 100 | 82.3 | 82.3 |
| 4 | chloroform | 100 | 61.9 | 61.9 |
| 5 | acetonitrile | 100 | 79.1 | 79.1 |
| 6 | methanol | 100 | 44.8 | 44.8 |
| 7 | ethanol | 100 | 54.5 | 54.5 |
| 8 | isopropanol | 100 | 54.8 | 54.8 |
| 9 | DMSO | 100 | 6.8 | 6.80 |
| 10 | DMF | 100 | 17.5 | 17.5 |
| Entry | Solvent | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|
| 1 | ethyl acetate | 100 | 89.9 | 89.9 |
| 2 | tetrahydrofuran | 99.3 | 81.1 | 81.7 |
| 3 | dichloromethane | 100 | 82.3 | 82.3 |
| 4 | chloroform | 100 | 61.9 | 61.9 |
| 5 | acetonitrile | 100 | 79.1 | 79.1 |
| 6 | methanol | 100 | 44.8 | 44.8 |
| 7 | ethanol | 100 | 54.5 | 54.5 |
| 8 | isopropanol | 100 | 54.8 | 54.8 |
| 9 | DMSO | 100 | 6.8 | 6.80 |
| 10 | DMF | 100 | 17.5 | 17.5 |
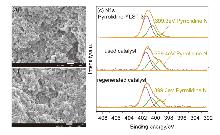
| Entry | Catalyst | Elemental content/% | ||||
|---|---|---|---|---|---|---|
| N | C | H | O | |||
| 1 | Pyrrolidine-YLST-3 | 7.0 | 54.6 | 8.4 | 30.0 | |
| 2 | used catalyst | 4.8 | 54.5 | 7.2 | 33.5 | |
| 3 | regenerated catalyst | 5.9 | 52.1 | 7.6 | 34.4 | |
| Entry | Catalyst | Elemental content/% | ||||
|---|---|---|---|---|---|---|
| N | C | H | O | |||
| 1 | Pyrrolidine-YLST-3 | 7.0 | 54.6 | 8.4 | 30.0 | |
| 2 | used catalyst | 4.8 | 54.5 | 7.2 | 33.5 | |
| 3 | regenerated catalyst | 5.9 | 52.1 | 7.6 | 34.4 | |
| Entry | Substrate 1 | Substrate 2 | Product | Temp./℃ | Time/min | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|---|---|---|---|
| 1a |  | 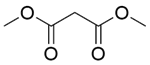 | 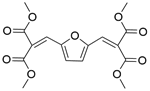 | 80 | 90 | 100 | 99.2 | 99.2 |
| 2a |  |  |  | 60 | 120 | 100 | 65.3 | 65.3 |
| 3a |  | 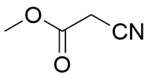 | 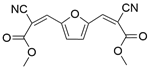 | 80 | 30 | 100 | 61.9 | 61.9 |
| 4a | 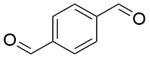 | 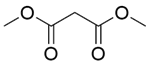 | 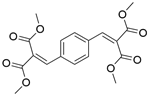 | 100 | 180 | 98.2 | 77.3 | 78.8 |
| 5b | 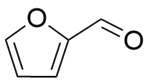 | 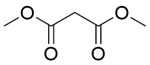 | 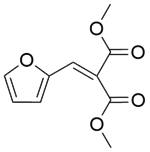 | 50 | 120 | 100 | 96.7 | 96.7 |
| 6b |  | 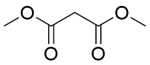 | 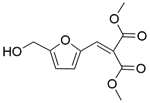 | 70 | 120 | 100 | 82.6 | 82.6 |
| 7b | 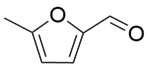 | 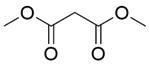 | 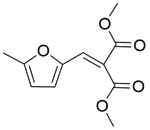 | 70 | 120 | 99.8 | 94.3 | 94.5 |
| 8b | 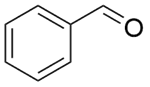 | 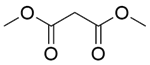 | 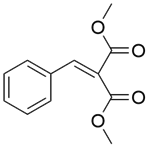 | 70 | 120 | 94.5 | 92.1 | 97.5 |
| 9c | 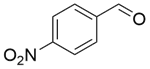 | 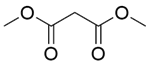 | 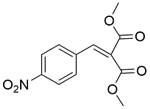 | 80 | 300 | 91.9 | 83.4 | 90.7 |
| 10b | 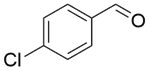 | 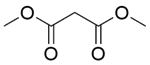 | 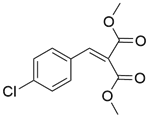 | 80 | 300 | 95.5 | 76.3 | 80.3 |
| 11b | 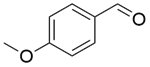 | 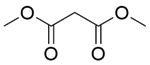 | 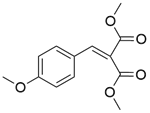 | 80 | 300 | 90.2 | 86.6 | 96.1 |
| 12c | 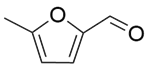 |  | 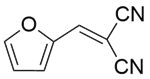 | 40 | 120 | 97.9 | 76.9 | 78.5 |
| 13b | 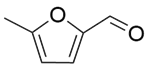 | 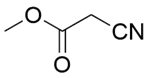 | 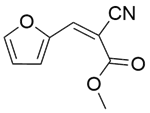 | 40 | 120 | 100 | 97.0 | 97.0 |
| Entry | Substrate 1 | Substrate 2 | Product | Temp./℃ | Time/min | Conversion/% | Yield/% | Selectivity/% |
|---|---|---|---|---|---|---|---|---|
| 1a |  |  |  | 80 | 90 | 100 | 99.2 | 99.2 |
| 2a |  |  |  | 60 | 120 | 100 | 65.3 | 65.3 |
| 3a |  |  |  | 80 | 30 | 100 | 61.9 | 61.9 |
| 4a |  |  |  | 100 | 180 | 98.2 | 77.3 | 78.8 |
| 5b |  |  |  | 50 | 120 | 100 | 96.7 | 96.7 |
| 6b |  |  |  | 70 | 120 | 100 | 82.6 | 82.6 |
| 7b |  |  |  | 70 | 120 | 99.8 | 94.3 | 94.5 |
| 8b |  |  |  | 70 | 120 | 94.5 | 92.1 | 97.5 |
| 9c |  |  |  | 80 | 300 | 91.9 | 83.4 | 90.7 |
| 10b |  |  |  | 80 | 300 | 95.5 | 76.3 | 80.3 |
| 11b |  |  |  | 80 | 300 | 90.2 | 86.6 | 96.1 |
| 12c |  |  |  | 40 | 120 | 97.9 | 76.9 | 78.5 |
| 13b |  |  |  | 40 | 120 | 100 | 97.0 | 97.0 |
| [1] |
|
| [2] |
doi: 10.1016/j.gr.2023.09.020 |
| [3] |
|
| [4] |
doi: 10.1002/cptc.v4.8 |
| [5] |
doi: 10.14419/ijac.v3i2 |
| [6] |
doi: 10.1016/j.fuel.2024.133423 |
| [7] |
doi: 10.6023/A24080246 |
|
(鞠嘉浩, 徐吉磊, 王康军, 黄家辉, 化学学报, 2024, 82, 1216.)
doi: 10.6023/A24080246 |
|
| [8] |
|
| [9] |
|
| [10] |
|
| [11] |
doi: 10.3390/polym14183868 |
| [12] |
doi: 10.6023/A24100329 |
|
(辛翠, 蒋俊, 邓紫微, 欧丽娟, 何卫民, 化学学报, 2024, 82, 1109.)
doi: 10.6023/A24100329 |
|
| [13] |
doi: 10.6023/A23120542 |
|
(郭建荣, 张书玉, 贺军辉, 任世学, 化学学报, 2024, 82, 242.)
doi: 10.6023/A23120542 |
|
| [14] |
|
| [15] |
doi: 10.1016/j.enconman.2009.03.013 |
| [16] |
doi: 10.1039/D3GC04663J |
| [17] |
|
| [18] |
doi: 10.3390/ijms26020727 |
| [19] |
doi: 10.1002/vnl.v30.4 |
| [20] |
doi: 10.1177/10915818231158272 |
| [21] |
doi: 10.3390/ijms20184333 |
| [22] |
|
| [23] |
doi: 10.1002/adhm.v4.2 |
| [24] |
doi: 10.1002/ls.161 |
| [25] |
doi: 10.1016/j.jconrel.2023.01.002 |
| [26] |
doi: 10.1038/nbt0892-894 pmid: 1368984 |
| [27] |
doi: 10.3389/fmicb.2017.02184 pmid: 29163455 |
| [28] |
doi: 10.1002/jctb.v50:4 |
| [29] |
doi: 10.1016/j.electacta.2022.140796 |
| [30] |
doi: 10.1016/j.tetlet.2004.09.037 |
| [31] |
doi: 10.1016/j.apcata.2004.10.039 |
| [32] |
doi: 10.2478/pjct-2018-0061 |
| [33] |
doi: 10.3390/catal10121443 |
| [34] |
|
|
(李建, 刘燕, 干丰丰, 杨运信, 分析试验室, 2017, 36, 726.)
|
|
| [35] |
doi: 10.1021/acssuschemeng.8b04310 |
| [36] |
doi: 10.1016/j.gee.2023.02.004 |
| [37] |
doi: 10.6023/A25030099 |
|
(刘珊珊, 董微微, 李珍珍, 张瑶瑶, 李超, 焦林郁, 化学学报, 2025, 83, 479.)
doi: 10.6023/A25030099 |
|
| [38] |
|
| [39] |
doi: 10.2174/1385272827666230406082857 |
| [40] |
doi: 10.1007/s10562-017-2255-6 |
| [41] |
doi: 10.1016/j.jscs.2019.01.001 |
| [42] |
doi: 10.1002/slct.v5.46 |
| [43] |
doi: 10.1002/macp.v205:7 |
| [44] |
doi: 10.1016/S0022-2860(01)00719-0 |
| [45] |
doi: 10.1134/S0022476617050080 |
| [46] |
pmid: 17044721 |
| [47] |
doi: 10.1063/1.3633699 |
| [48] |
doi: 10.1002/aic.v42:1 |
| [49] |
doi: 10.1016/j.tsf.2015.07.051 |
| [50] |
pmid: 15253839 |
| [51] |
doi: 10.1021/acs.joc.2c00047 |
| [52] |
doi: 10.1016/j.electacta.2022.141512 |
| [53] |
doi: 10.1021/acs.iecr.9b01375 |
| [54] |
doi: 10.1016/j.molstruc.2018.04.078 |
| [55] |
doi: 10.1016/j.catcom.2020.105954 |
| [56] |
doi: 10.1039/C6RA25595G |
| [57] |
doi: 10.2174/1570179412666150505185134 |
| [1] | 王怡可, 陈丽萍, 舒敬利, 朱雪华, 汪洋. 基于相转移催化的水相烯烃高效反马氏硫氢化反应[J]. 化学学报, 2026, 84(3): 299-304. |
| [2] | 刘雨涵, 高盼. 使用机械化学生成的钙基重格氏试剂(R-CaX)对有机卤化物进行直接硼化[J]. 化学学报, 2024, 82(11): 1114-1119. |
| [3] | 刘坜, 郑刚, 范国强, 杜洪光, 谭嘉靖. 4-酰基/氨基羰基/烷氧羰基取代汉斯酯参与的有机反应研究进展[J]. 化学学报, 2023, 81(6): 657-668. |
| [4] | 李靖鹏, 杨棋, 张周, 曾贵云, 刘腾, 黄超. 多组分连续流动高选择性合成(Z)-N-乙烯基环N,O-缩醛衍生物[J]. 化学学报, 2022, 80(11): 1463-1468. |
| [5] | 李玲玲, 刘宇, 宋术岩, 张洪杰. 配位环境可调的Cu单原子的合成及催化加氢性能研究※[J]. 化学学报, 2022, 80(1): 16-21. |
| [6] | 魏哲宇, 常亚林, 余焓, 韩生, 魏永革. Anderson型杂多酸作为催化剂在有机合成中的应用[J]. 化学学报, 2020, 78(8): 725-732. |
| [7] | 董奎, 刘强, 吴骊珠. 放氢交叉偶联反应[J]. 化学学报, 2020, 78(4): 299-310. |
| [8] | 叶文波, 晏子聪, 万常峰, 侯豪情, 汪志勇. 一种新的肉桂酸类化合物的脱羧/甲基化反应[J]. 化学学报, 2018, 76(2): 99-102. |
| [9] | 张文强, 李秋艳, 杨馨雨, 马征, 王欢欢, 王晓军. 含苯并噻二唑结构单元的金属有机骨架在可见光催化需氧氧化反应中的应用[J]. 化学学报, 2017, 75(1): 80-85. |
| [10] | 裴朋昆, 张凡, 易红, 雷爱文. 可见光促进的苄位Csp3-H键活化官能团化反应[J]. 化学学报, 2017, 75(1): 15-21. |
| [11] | 陆庆全, 易红, 雷爱文. 自然氧化偶联及其在碳氢功能化反应中的应用[J]. 化学学报, 2015, 73(12): 1245-1249. |
| [12] | 杨旭石, 黄建林, 朱凤霞, 李和兴. 乙基桥联有序介孔有机硅负载Pd(II)有机金属催化剂用于水介质Barbier反应[J]. 化学学报, 2010, 68(03): 217-221. |
| [13] | 曹洁明,郑明波,陆鹏,邓少高,陈勇平,文凡,郭静,张防,陶杰. 利用还原性多糖合成银纳米粒子[J]. 化学学报, 2005, 63(16): 1541-1544. |
| [14] | 戴立信,陈耀全. 创造更美好的生活和更清洁的环境: 化学的回顾与展望[J]. 化学学报, 2000, 58(1): 1-5. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||