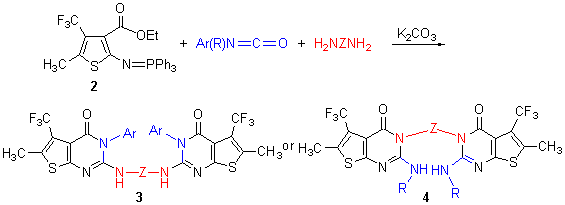
| [1] Chambhare, R. V.; Khadse, B. G.; Bobde, A. S.; Bahekar, R. H. Eur. J. Med. Chem. 2003, 38, 89.[2] Ren Q. Y.; Liang, Y. J.; He, H. W.; Fu, L. W.; Gu, Y. C. Bioorg. Med. Chem. Lett. 2009, 19, 6713.[3] Ren, Q. Y.; Cui, Z. P.; He, H. W.; Gu, Y. C. J. Fluorine Chem. 2007, 128, 1369.[4] Jennings, L. D.; Kincaid, S. L.; Wang, Y. D.; Krishnamurthy, G.; Beyer, C. F.; McGinnis, J. P.; Miranda, M.; Discafani, C. M.; Rabindran, S. K. Bioorg. Med. Chem. Lett. 2005, 15, 4731.[5] Blanco, G.; Quintela, J. M.; Peinador, C. Tetrahedron 2008, 64, 1333.[6] Bonne, D.; Coquerel, Y.; Constantieux, T.; Rodriguez, J. Tetrahedron: Asymmetry 2010, 21,1085.[7] Palacios, F.; Alonso, C.; Aparicio, D. T.; Rubiales, G.; Santos, J. Tetrahedron 2007, 63, 523.[8] Parmar, K.; Suthar, B.; Suthar, A.; Maheta, A. J. Heterocycl. Chem. 2009, 46, 975.[9] Hafez, H. N.; EI-Gazzar, A. B. A. Bioorg. Med. Chem. Lett. 2008, 18, 5222.[10] Snegaroff, K.; Lasssagne, F.; Ababsa, G. B.; Nassar, E.; Ely, S. C. S.; Hesse, S.; Perspicace, E.; Derdour, A.; Mongin, F. Org. Biomol. Chem. 2009, 7, 4782.[11] Pedeboscq, S.; Gravier, D.; Casadebaig, F.; Hou, G.; Gissot, A.; Giorgi, F. D.; Ichas, F.; Cambar, J.; Pometan, J. P. Eur. J. Med. Chem. 2010, 45, 2473.[12] Yang, X. H.; Wu, M. H.; Sun, S. F.; Xie, J. L.; Ding, M. W.; Xia, Q. H. J. Heterocycl. Chem. 2008, 45, 1365.[13] Fang, Z.-D.; Lü, J.-Q. Chin. J. Org. Chem. 2011, 31, 1888 (in Chinese). (方正东, 吕鉴泉, 有机化学, 2011, 31, 1888.)[14] Liang, Y.; He, H. W.; Yang, Z. W. J. Heterocycl. Chem. 2011,48, 88.[15] Song, A.; Lam, K. S. Tetrahedron 2004, 60, 8605.[16] Yang, X. H.; Wu, M. H.; Ding, M. W. Chin. J. Org. Chem. 2010, 30, 1032 (in Chinese). (杨绪红, 吴鸣虎, 丁明武, 有机化学, 2010, 30, 1032.) |