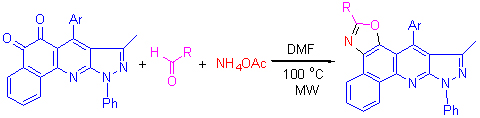
| [1] Alagarsamy, V.; Pathak, U. S. Bioorg. Med. Chem. 2007, 15, 3457.[2] Murugan, V.; Kulkarni, M.; Anand, R. M.; Kumar, E. P.; Suresh, B.; Reddy, V. M. Asian J. Chem. 2006, 18, 900.[3] Alagarsamy, V.; Raja S., V.; Dhanabal, K. Bioorg. Med. Chem. 2007, 15, 235.[4] Selvam, P.; Girija, K.; Nagarajan, G.; De Clerco, E. Indian J. Pharm. Sci. 2005, 67, 484.[5] Godfrey, A. A. A. WO 2005012260, 2005 [Chem. Abstr. 2005, 142, 198095].[6] Chaturvedula, P. V.; Chen, L.; Civiello, R.; Degnan, A. P.; Dubowchik, G. M.; Han, X.; Jiang, X. J.; Macor, J. E.; Poindexter, G. S.; Tora, G. O.; Luo, G. US 2007149503, 2007 [Chem. Abstr. 2007, 147, 118256].[7] Alanine, A.; Gobbi, L. C.; Kolczewski, S.; Luebbers, T.; Peters, J. U.; Steward, L. US 2006293350, 2006 [Chem. Abstr. 2006, 146, 100721].[8] Letourneau, J.; Riviello, C.; Ho, K. K.; Chan, J. H.; Ohlmeyer, M.; Jokiel, P.; Neagu, I.; Morphy, J. R.; Napier, S. E. WO 2006095014, 2006 [Chem. Abstr. 2006, 145, 315012].[9] (a) Dalisay, D. S.; Rogers, E. W.; Edison, A. S.; Molinski, T. F. J. Nat. Prod. 2009, 72, 732.(b) Hagelueken, G.; Albrecht, S. C.; Steinmetz, H.; Jansen, R.; Heinz, D. W.; Kalesse, M.; Schubert, W. D. Angew. Chem., Int. Ed. 2009, 48, 595.(c) Jin, Z. Nat. Prod. Rep. 2006, 23, 464.[10] (a) Vedejs, E.; Barda, D. A. Org. Lett. 2000, 2, 1033.(b) Wan, C.; Zhang, J.; Wang, S.; Fan, J.; Wang, Z. Org. Lett. 2010, 12, 2338.(c) Jiang, H.; Huang, H.; Cao, H.; Qi, C. Org. Lett. 2010, 12, 5561.[11] (a) Ferrini, P. G.; Marxer, A. Angew. Chem., Int. Ed. 1963, 2, 99.(b) Pan, Y.-M.; Zheng, F.-J.; Lin, H.-X.; Zhan, Z.-P. J. Org. Chem. 2009, 74, 3148.(c) Young, G. L.; Smith, S. A.; Taylor, R. J. K. Tetrahedron Lett. 2004, 45, 3797.[12] (a) Williams, D. R.; Lowder, D. P.; Gu, G.-Y.; Brooks, D. A. Tetrahedron Lett. 1997, 38, 331.(b) Meyers, A. I.; Tavares, F. X. J. Org. Chem. 1996, 61, 8207.(c) Uto, A. J. Y.; Wipf, P.; Reno, M. J.; Williams, D. R. Org. Lett. 2000, 2, 1165.[13] (a) Derridj, F.; Djebbar, S.; Benali-Baitich, O.; Doucet, H. J. Organomet. Chem. 2008, 693, 135.(b) Flegeau, E. F.; Popkin, M. E.; Greaney, M. F. Org. Lett. 2006, 8, 2495.(c) Lee, K.; Counceller, C. M.; Stambuli, J. P. Org. Lett. 2009, 11, 1457.[14] (a) Tietze, L. F.; Brasche, G.; Gerike, K. Domino Reactions in Organic Chemistry, Wiley-VCH, Weinheim, 2006.(b) Aaron M. D.; Eric, F. Acc. Chem. Res. 2010, 43, 440.(c) Li, J.; Wang, N.; Li, C. J.; Jia, X. S. Chem. Eur. J. 2012, 18, 9645.(d) Li, J.; Wang, N.; Li, C. J.; Jia, X. S. Org. Lett. 2012, 14, 4994.[15] (a) Fan, W.; Ye, Q.; Xu, H.-W.; Jiang, B.; Wang, S.-L.; Tu, S.-J. Org. Lett. 2013, 15, 2258.(b) Jiang, B.; Li, Q.-Y.; Zhang, H.; Tu, S.-J.; Pindi, S.; Li, G. Org. Lett. 2012, 14, 700.(c)Jiang, B.; Li, Y.; Tu, M.-S.; Wang, S.-L.; Tu, S.-J.; Li, G. J. Org. Chem. 2012, 77, 7497.[16] (a) Jiang, B.; Zhang, G.; Ma, N.; Shi, F.; Tu, S.-J.; Kaur, P.; Li, G. Org. Biomol. Chem. 2011, 9, 3834.(b) Wang, S.-L.; Zhang, G.; Jie, D.; Jiang, B.; Wang, X.-H.; Tu, S.-J. Comb. Chem. High Throughput Screening 2012, 15, 400.[17] (a) Rajesh, S. M.; Bala, B. D.; Perumal, S.; Menendez J. C. Green Chem. 2011, 13, 3248.(b) Wang, S.-L.; Wu, F.-Y.; Cheng, C.; Zhang, G.; Liu, Y.-P.; Jiang, B.; Shi, F.; Tu, S.-J. ACS Comb. Sci. 2011, 13, 135.[18] (a) Mahdavinia, G. H.; Amani, A. M.; Sepehrian, H. Chin. J. Chem. 2012, 30, 703.(b) Wang, L.; Yao, Y.; Wang, Y.; Zou, G. Chin. J. Chem. 2009, 27, 343. |