有机化学 ›› 2021, Vol. 41 ›› Issue (9): 3593-3607.DOI: 10.6023/cjoc202105032 上一篇 下一篇
研究论文
李英俊a,*( ), 林乐弟a, 刘季红b, 高立信c, 盛丽c, 靳焜d, 刘雪洁a, 杨鸿境a, 李佳c,*(
), 林乐弟a, 刘季红b, 高立信c, 盛丽c, 靳焜d, 刘雪洁a, 杨鸿境a, 李佳c,*( )
)
收稿日期:2021-05-17
修回日期:2021-06-21
发布日期:2021-07-12
通讯作者:
李英俊, 李佳
基金资助:
Yingjun Lia( ), Ledi Lina, Jihong Liub, Lixin Gaoc, Li Shengc, Kun Jind, Xuejie Liua, Hongjing Yanga, Jia Lic(
), Ledi Lina, Jihong Liub, Lixin Gaoc, Li Shengc, Kun Jind, Xuejie Liua, Hongjing Yanga, Jia Lic( )
)
Received:2021-05-17
Revised:2021-06-21
Published:2021-07-12
Contact:
Yingjun Li, Jia Li
Supported by:文章分享

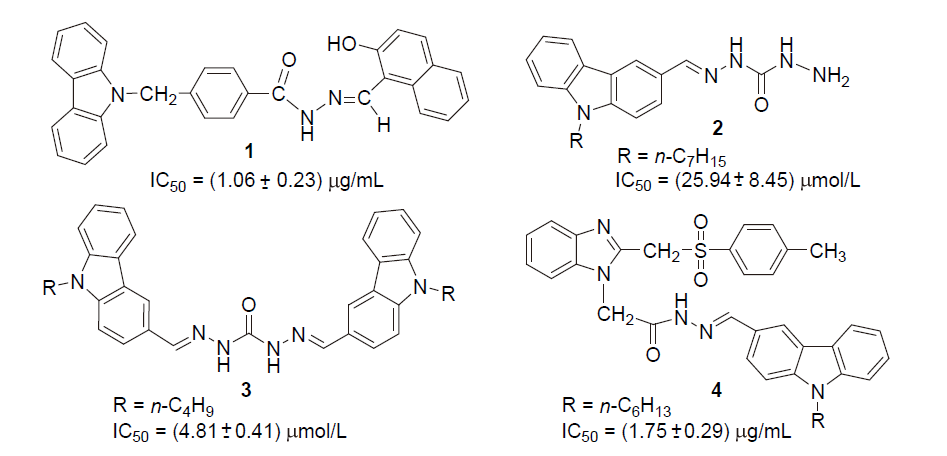
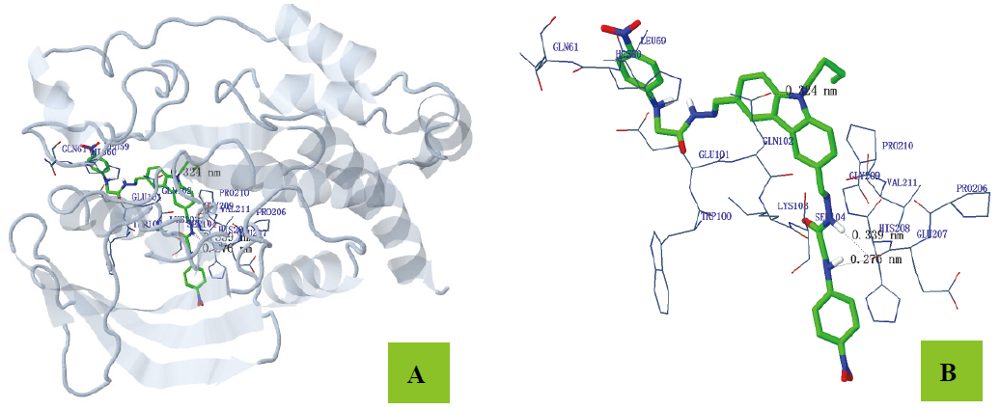
为寻找高效、低毒的新型蛋白酪氨酸磷酸酶1B (PTP1B)抑制剂, 设计并合成出了一系列新型含咔唑环和芳环/芳稠杂环的N-酰腙衍生物6~8和11. 利用IR、1H NMR、13C NMR和2D NMR(包括1H-1H COSY和NOESY)谱及元素分析确定了其结构和构型. 评价了目标化合物对PTP1B的抑制活性. 实验结果表明, 目标化合物对PTP1B均有较强的抑制活性, 除了化合物N'-[9-(2-氯噻唑-5-甲基)咔唑-3-亚甲基]-2-苯氨基乙酰肼(6a)、N'-[9-(2-氯噻唑-5-甲基)咔唑-3-亚甲基]-2-(4-甲基苯氨基)乙酰肼(6b)、N'-[9-(2-氯噻唑-5-甲基)咔唑-3-亚甲基]-2-(3-硝基苯氨基)乙酰肼(6g)和N'-[9-(2-氯噻唑-5-甲基)咔唑-3-亚甲基]-2-(4-硝基苯氨基)乙酰肼(6h)外, 其它化合物的活性均高于阳性对照药物齐墩果酸, 其中N,N'-[(9-丁基咔唑基)-3,6-二亚甲基]-2,2'-[二(4-硝基苯氨基)]双乙酰肼(11b)的活性最高, IC50=(0.89±0.06) μmol/L. 利用分子对接分别研究了代表目标化合物N'-[9-(2-氯噻唑-5-甲基)咔唑-3-亚甲基]-2-(4-溴苯氨基)乙酰肼(6d)、N'-[9-(2-氯噻唑-5-甲基)咔唑-3-亚甲基]-2-((2-(1-萘氧基)甲基)苯并咪唑-1-基)乙酰肼(7f)和11b与PTP1B酶的结合模式.
李英俊, 林乐弟, 刘季红, 高立信, 盛丽, 靳焜, 刘雪洁, 杨鸿境, 李佳. 新型含咔唑环和芳环/芳稠杂环的N-酰腙衍生物的合成及蛋白酪氨酸磷酸酶1B (PTP1B)抑制活性评价[J]. 有机化学, 2021, 41(9): 3593-3607.
Yingjun Li, Ledi Lin, Jihong Liu, Lixin Gao, Li Sheng, Kun Jin, Xuejie Liu, Hongjing Yang, Jia Li. Synthesis and Protein Tyrosine Phosphatase 1B (PTP1B) Inhibitory Activity Evaluation of Novel N-Acylhydrazone Derivatives Containing Carbazole and Aromatic Ring/Aromatic Fused Heterocycle[J]. Chinese Journal of Organic Chemistry, 2021, 41(9): 3593-3607.
| Position | δH | 1H-1H COSY A (B) | NOESY A (B) | |
|---|---|---|---|---|
| A | B | |||
| 1 (1') | 7.86 (d, J=8.5 Hz) | —a | H-2 (—) | / |
| 2 (2') | 7.93 (dd, J=8.8, 1.3 Hz) | 7.86 | H-1, 4 | H-9/H-9' |
| 3 (3') | /b | / | / | / |
| 4 (4') | 8.50 (d, J=1.0 Hz) | 8.47 | H-2 (H-2') | / |
| 5 (5') | 8.24 (d, J=8.0 Hz) | 8.23 (d, J=8.0 Hz) | H-6 (—) | / |
| 6 (6') | 7.29 (t, J=7.5 Hz) | — | H-5, 7 (—) | / |
| 7 (7') | 7.53 (td, J=7.8, 1.0 Hz) | — | H-6, 8 (—) | / |
| 8 (8') | 7.83 (d, J=8.5 Hz) | — | H-7 (—) | H-13 (—) |
| 9 (9') | 8.20 | 8.37 | / | H-2/H-2'/H-10/H-10' |
| 10 (10') | 11.74 | 11.80 | / | H-9/H-9'/H-11/H-11' |
| 11 (11') | 5.68 | 5.17 | / | H-10/H-10'/H-15 (—) |
| 12 (12') | 5.44 | — | / | H-19 (—) |
| 13 (13') | 5.95 | 5.94 | / | H-8 (—)/H-14 (—) |
| 14 (14') | 7.88 | — | / | H-13 (—) |
| 15 (15') | 7.614 (d, J=7.5 Hz) | 7.606 (d, J=7.5 Hz) | H-16 (—) | H-11 (—) |
| 16 (16') | 7.27 (t, J=7.0 Hz) | — | H-15, 17 (—) | / |
| 17 (17') | 7.24 (td, J=7.5, 1.0 Hz) | — | H-16, 18 (—) | / |
| 18 (18') | 7.69 (d, J=7.5 Hz) | — | H-17 (—) | / |
| 19 (19') | 7.10 (d, J=9.5 Hz) | — | H-20 (—) | H-12 (—) |
| 20 (20') | 7.26 (d, J=9.0 Hz) | — | H-19 (—) | / |
| Position | δH | 1H-1H COSY A (B) | NOESY A (B) | |
|---|---|---|---|---|
| A | B | |||
| 1 (1') | 7.86 (d, J=8.5 Hz) | —a | H-2 (—) | / |
| 2 (2') | 7.93 (dd, J=8.8, 1.3 Hz) | 7.86 | H-1, 4 | H-9/H-9' |
| 3 (3') | /b | / | / | / |
| 4 (4') | 8.50 (d, J=1.0 Hz) | 8.47 | H-2 (H-2') | / |
| 5 (5') | 8.24 (d, J=8.0 Hz) | 8.23 (d, J=8.0 Hz) | H-6 (—) | / |
| 6 (6') | 7.29 (t, J=7.5 Hz) | — | H-5, 7 (—) | / |
| 7 (7') | 7.53 (td, J=7.8, 1.0 Hz) | — | H-6, 8 (—) | / |
| 8 (8') | 7.83 (d, J=8.5 Hz) | — | H-7 (—) | H-13 (—) |
| 9 (9') | 8.20 | 8.37 | / | H-2/H-2'/H-10/H-10' |
| 10 (10') | 11.74 | 11.80 | / | H-9/H-9'/H-11/H-11' |
| 11 (11') | 5.68 | 5.17 | / | H-10/H-10'/H-15 (—) |
| 12 (12') | 5.44 | — | / | H-19 (—) |
| 13 (13') | 5.95 | 5.94 | / | H-8 (—)/H-14 (—) |
| 14 (14') | 7.88 | — | / | H-13 (—) |
| 15 (15') | 7.614 (d, J=7.5 Hz) | 7.606 (d, J=7.5 Hz) | H-16 (—) | H-11 (—) |
| 16 (16') | 7.27 (t, J=7.0 Hz) | — | H-15, 17 (—) | / |
| 17 (17') | 7.24 (td, J=7.5, 1.0 Hz) | — | H-16, 18 (—) | / |
| 18 (18') | 7.69 (d, J=7.5 Hz) | — | H-17 (—) | / |
| 19 (19') | 7.10 (d, J=9.5 Hz) | — | H-20 (—) | H-12 (—) |
| 20 (20') | 7.26 (d, J=9.0 Hz) | — | H-19 (—) | / |
| Compd. | Ar/Ar' | IC50/(μmol•L–1) | Compd. | Ar/Ar' | IC50/(μmol•L–1) |
|---|---|---|---|---|---|
| 6a | C6H5 | 6.14±0.00 | 7a | 4-CH3C6H4 | 1.91±0.32 |
| 6b | 4-CH3C6H4 | 3.05±0.96 | 7b | 2,4-(CH3)2C6H3 | 2.40±0.17 |
| 6c | 4-CH3OC6H4 | 2.26±0.32 | 7c | 4-CH3OC6H4 | 2.46±0.27 |
| 6d | 4-BrC6H4 | 2.01±0.16 | 7d | 4-ClC6H4 | 1.92±0.47 |
| 6e | 3-ClC6H4 | 2.44±0.35 | 7e | 3-O2NC6H4 | 2.35±0.25 |
| 6f | 4-ClC6H4 | 2.91±0.67 | 7f | 1-C10H7 | 1.83±0.11 |
| 6g | 3-O2NC6H4 | 3.53±0.21 | 8 | | 2.71±0.60 |
| 6h | 4-O2NC6H4 | 3.29±0.39 | 11a | 3-O2NC6H4 | 0.98±0.00 |
| OA | 2.93±0.92 | 11b | 4-O2NC6H4 | 0.89±0.06 |
| Compd. | Ar/Ar' | IC50/(μmol•L–1) | Compd. | Ar/Ar' | IC50/(μmol•L–1) |
|---|---|---|---|---|---|
| 6a | C6H5 | 6.14±0.00 | 7a | 4-CH3C6H4 | 1.91±0.32 |
| 6b | 4-CH3C6H4 | 3.05±0.96 | 7b | 2,4-(CH3)2C6H3 | 2.40±0.17 |
| 6c | 4-CH3OC6H4 | 2.26±0.32 | 7c | 4-CH3OC6H4 | 2.46±0.27 |
| 6d | 4-BrC6H4 | 2.01±0.16 | 7d | 4-ClC6H4 | 1.92±0.47 |
| 6e | 3-ClC6H4 | 2.44±0.35 | 7e | 3-O2NC6H4 | 2.35±0.25 |
| 6f | 4-ClC6H4 | 2.91±0.67 | 7f | 1-C10H7 | 1.83±0.11 |
| 6g | 3-O2NC6H4 | 3.53±0.21 | 8 | | 2.71±0.60 |
| 6h | 4-O2NC6H4 | 3.29±0.39 | 11a | 3-O2NC6H4 | 0.98±0.00 |
| OA | 2.93±0.92 | 11b | 4-O2NC6H4 | 0.89±0.06 |
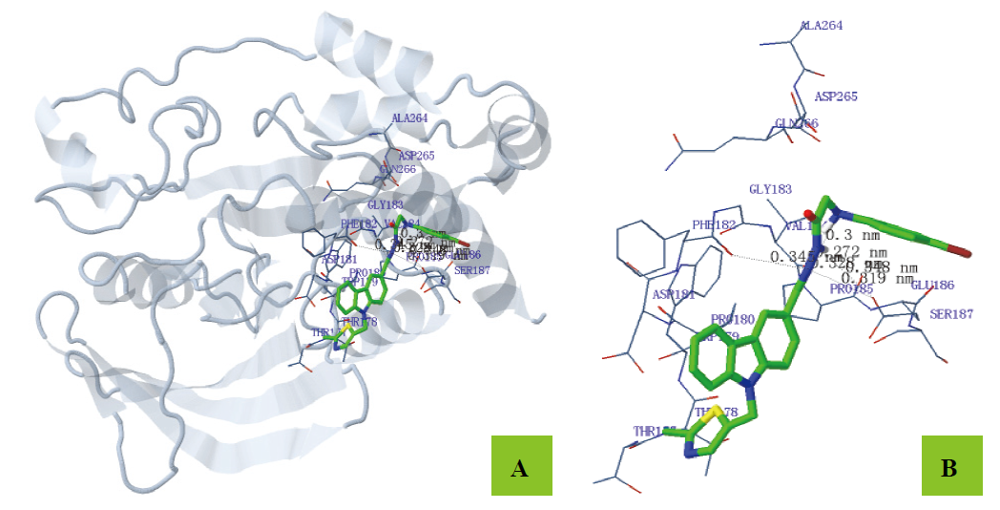
| Est. free energy of binding/(kJ•mol–1) | No. of H-bond | H-bonds (length/nm) | Polar (length/nm) | Hydrophobic (length/nm) | Pi-Pi (length/nm) | Halogen bond (length/nm) |
|---|---|---|---|---|---|---|
| –31.25 | 6 | N(3)-PHE182 (0.345) N(3)-VAL184 (0.328) N(4)-VAL184 (0.272) N(5)-VAL184 (0.300) N(3)-GLU186 (0.319) N(4)-GLU186 (0.348) | H12-GLU186 (0.343) | C(8)-PRO180 (0.372) C(9)-PRO180 (0.375) C(10)-PRO180 (0.331) C(11)-PRO180 (0.305) C(12)-PRO180 (0.326) C(17)-PRO180 (0.350) C(20)-VAL184 (0.370) C(21)-VAL184 (0.354) | C(1)-PHE182 (0.357) C(2)-PHE182 (0.377) | Cl(1)-TRP179 (0.389) |
| Est. free energy of binding/(kJ•mol–1) | No. of H-bond | H-bonds (length/nm) | Polar (length/nm) | Hydrophobic (length/nm) | Pi-Pi (length/nm) | Halogen bond (length/nm) |
|---|---|---|---|---|---|---|
| –31.25 | 6 | N(3)-PHE182 (0.345) N(3)-VAL184 (0.328) N(4)-VAL184 (0.272) N(5)-VAL184 (0.300) N(3)-GLU186 (0.319) N(4)-GLU186 (0.348) | H12-GLU186 (0.343) | C(8)-PRO180 (0.372) C(9)-PRO180 (0.375) C(10)-PRO180 (0.331) C(11)-PRO180 (0.305) C(12)-PRO180 (0.326) C(17)-PRO180 (0.350) C(20)-VAL184 (0.370) C(21)-VAL184 (0.354) | C(1)-PHE182 (0.357) C(2)-PHE182 (0.377) | Cl(1)-TRP179 (0.389) |
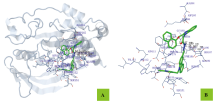
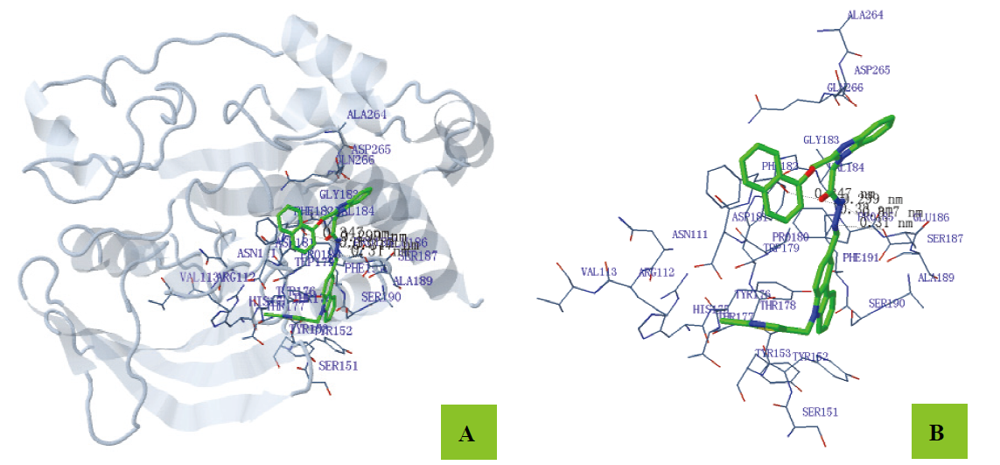
| Est. free energy of binding/(kJ•mol–1) | No. of H-bond | H-bonds (length/nm) | Polar (length/nm) | Hydrophobic (length/nm) | Pi-Pi (length/nm) |
|---|---|---|---|---|---|
| –26.11 | 5 | N(4)-PHE182 (0.347) N(4)-VAL184 (0.299) N(3)-VAL184 (0.330) N(4)-GLU186 (0.317) N(3)-GLU186 (0.310) | H(12)-GLU186 0.328 | C(17)-PRO180 (0.342) C(11)-PRO180 (0.299) C(10)-PRO180 (0.313) C(9)-PRO180 (0.370) C(12)-PRO180 (0.345) C(27)-PHE182 (0.371) C(1)-PRO185 (0.349) C(12)-PRO185 (0.384) | C(3)-TYR152 (0.387) C(31)-PHE182 (0.356) C(32)-PHE182 (0.359) C(34)-PHE182 (0.357) C(35)-PHE182 (0.385) |
| Est. free energy of binding/(kJ•mol–1) | No. of H-bond | H-bonds (length/nm) | Polar (length/nm) | Hydrophobic (length/nm) | Pi-Pi (length/nm) |
|---|---|---|---|---|---|
| –26.11 | 5 | N(4)-PHE182 (0.347) N(4)-VAL184 (0.299) N(3)-VAL184 (0.330) N(4)-GLU186 (0.317) N(3)-GLU186 (0.310) | H(12)-GLU186 0.328 | C(17)-PRO180 (0.342) C(11)-PRO180 (0.299) C(10)-PRO180 (0.313) C(9)-PRO180 (0.370) C(12)-PRO180 (0.345) C(27)-PHE182 (0.371) C(1)-PRO185 (0.349) C(12)-PRO185 (0.384) | C(3)-TYR152 (0.387) C(31)-PHE182 (0.356) C(32)-PHE182 (0.359) C(34)-PHE182 (0.357) C(35)-PHE182 (0.385) |
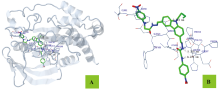
| Est. free energy of binding/(kJ•mol–1) | No. of H-bond | H-bonds (length/nm) | Polar a (length/nm) | Hydrophobic (length/nm) | Pi-Pi (length/nm) |
|---|---|---|---|---|---|
| –20.25 | 3 | N(1)-GLN102 (0.324) N(3)-GLU207 (0.339) N(6)-GLU207 (0.276) | O(2)-LYS103 (0.384) H(24)-HIS208 (0.375) | C(20)-HIS60 (0.325) C(3)-PRO210 (0.374) C(4)-PRO210 (0.332) | C(32)-HIS60 (0.384) C(33)-HIS60 (0.335) C(11)-HIS60 (0.389) C(34)-HIS60 (0.349) C(27)-HIS208 (0.377) C(28)-HIS208 (0.358) C(29)-HIS208 (0.383) |
| Est. free energy of binding/(kJ•mol–1) | No. of H-bond | H-bonds (length/nm) | Polar a (length/nm) | Hydrophobic (length/nm) | Pi-Pi (length/nm) |
|---|---|---|---|---|---|
| –20.25 | 3 | N(1)-GLN102 (0.324) N(3)-GLU207 (0.339) N(6)-GLU207 (0.276) | O(2)-LYS103 (0.384) H(24)-HIS208 (0.375) | C(20)-HIS60 (0.325) C(3)-PRO210 (0.374) C(4)-PRO210 (0.332) | C(32)-HIS60 (0.384) C(33)-HIS60 (0.335) C(11)-HIS60 (0.389) C(34)-HIS60 (0.349) C(27)-HIS208 (0.377) C(28)-HIS208 (0.358) C(29)-HIS208 (0.383) |
| [1] |
Ji, W. J.; Chen, X. L.; Lv, J.; Wang, M.; Ren, S. T.; Yuan, B. X.; Wang, B.; Chen, L. N. Int. J. Endocrinol. 2014, 2014, 312452.
|
| [2] |
Yang, L. L.; Chen, F.; Gao, C.; Chen, J. B.; Li, J. Y.; Liu, S. Y.; Zhang, Y. Y.; Wang, Z. Y.; Qian, S. J. Enzyme Inhib. Med. Chem. 2020, 35, 152.
|
| [3] |
Salinas-Arellano, E.; Pérez-Vásquez, A.; Rivero-Cruz, I.; Torres- Colin, R.; González-Andrade, M.; Rangel-Grimaldo, M.; Mata, R. Molecules 2020, 25, 3530.
doi: 10.3390/molecules25153530 |
| [4] |
Krishnan, N.; Konidaris, K. F.; Gasser, G.; Tonks, N. K. J. Biol. Chem. 2018, 293, 1517.
|
| [5] |
Guo, S. J.; Wang, L. J.; Chen, D.; Jiang, B. RSC Adv. 2020, 10, 3429.
doi: 10.1039/C9RA10660J |
| [6] |
Costa, G. C.; Montagnoli, T. L.; Silva, M. M. C.; Gamba, L. E. R.; Alves, B. E. O.; Silva, M. M. C.; Trachez, M. M.; Nascimento, J. H. M.; Pimentel-Coelho, P. M.; Mendez-Otero, R.; Lima, L. M.; Barreiro, E. J.; Sudo, R. T.; Zapata-Sudo, G. Drug Des. Dev. Ther. 2020, 14, 3337.
doi: 10.2147/DDDT.S258459 |
| [7] |
Lu, Y. J.; Zhao, Z. D.; Chen, Y. X.; Wang, J.; Xu, S. C.; Gu, Y. J. Asian Nat. Prod. Res. 2021, 23, 545.
|
| [8] |
Liu, K.; Ding, Y. Y.; Kang, C. M. Pharm. Chem. J. 2020, 54, 345.
doi: 10.1007/s11094-020-02215-w |
| [9] |
Mota, F. V. B.; Neta, M. S. A.; Franco, E. S.; Bastos, I. V. G. A.; Correia da Araújo, L. C; Cabral da Silva, S.; Bezerra de Oliveira, T.; Souza, E. K.; Moraes de Almeida, V.; Ximenes, R. M.; Maia, M. B. S.; Junior, F. J. B. M.; Marchand, P.; Rodolfo de Faria, A.; Gonçalves da Silva, T. Med. Chem. Commun. 2019, 10, 1916.
doi: 10.1039/C9MD00276F |
| [10] |
Sulaiman, A. T.; Sarsam, S. W. Iraqi J. Pharm. Sci. 2020, 29, 207.
|
| [11] |
Aarjane, M.; Aouidate, A.; Slassi, S.; Amine, A. Arab. J. Chem. 2020, 13, 6236.
|
| [12] |
Senthilkumar, S.; Seralathan, J.; Muthukumaran, G. J. Mol. Struct. 2021, 1226, 129354.
doi: 10.1016/j.molstruc.2020.129354 |
| [13] |
Yang, Z.-B.; Li, P.; He, Y. J. Molecules 2019, 24, 3766.
doi: 10.3390/molecules24203766 |
| [14] |
Silva, M. F.; Lima, E. T.; Pruccoli, L.; Castro, N. G.; Guimarães, M. J. R.; Silva, F. M. R.; Nadur, N. F.; Luiz de Azevedo, L.; Kümmerle, A. E.; Guedes, I. A.; Dardenne, L. E.; Gontijo, V. S.; Tarozzi, A.; Viegas Jr, C. Molecules 2020, 25, 3165.
doi: 10.3390/molecules25143165 |
| [15] |
Palwankar, S. M.; Kale, P. P.; Kadu, P. K.; Prabhavalkar, K. J. Rep. Pharm. Sci. 2020, 9, 79.
|
| [16] |
Aminah, N. S.; Thant, T. M.; Kristanti, A. N.; Ramadhan, R.; Aung, H. T.; Takaya, Y. Nat. Prod. Commun. 2020, 14, 1.
|
| [17] |
Chena, Y. M.; Cao, N. K.; Lv, H. N.; Zeng, K. W.; Yuan, J. Q.; Guo, X. Y.; Zhao, M. B.; Tu, P. F.; Jiang, Y. Phytochemistry 2020, 170, 112186.
doi: 10.1016/j.phytochem.2019.112186 |
| [18] |
Liu, Y. H.; Wu, Y. B.; Sun, L. Q.; Gu, Y. X.; Hu, L. X. Eur. J. Med. Chem. 2020, 191, 112181.
doi: 10.1016/j.ejmech.2020.112181 |
| [19] |
Yamashita, S.; Honda, R.; Fukuoka, M.; Kimurab, T.; Hosokawa-Mutoc, J.; Kuwataa, K. Prion 2020, 14, 42.
doi: 10.1080/19336896.2020.1714372 pmid: 31971853 |
| [20] |
Wang, W. B.; Zhou, Z. G.; Zhou, X, H.; Chen, L. M.; Bie, S.; Jing, Z. J. AMB Expr. 2020, 10, 148.
doi: 10.1186/s13568-020-01074-8 |
| [21] |
Taheri, B.; Taghavi, M.; Zarei, M.; Chamkouri, N.; Mojaddami, A. Bull. Chem. Soc. Ethiop. 2020, 34, 377.
|
| [22] |
Balakrishnan, R.; Vijayraja, D.; Jo, S.-H.; Ganesan, P.; Su-Kim, I.; Choi, D.-K. Antioxidants 2020, 9, 101.
doi: 10.3390/antiox9020101 |
| [23] |
Sadeghian, B.; Sakhteman, A.; Faghih, Z.; Nadri, H.; Edraki, N.. IRaji, A.; Sadeghian, I.; Rezaei, Z. J. Mol. Struct. 2020, 1221, 128793.
|
| [24] |
Telehoiu, A. T.; Nuţă, D. C.; Căproiu, M. T.; Dumitrascu, F.; Zarafu, I.; Ioniţă, P.; Badiceanu, C. D.; Avram, S.; Chifiriuc, M. C.; Bleotu, C.; Limban, C. Molecules 2020, 25, 266.
doi: 10.3390/molecules25020266 |
| [25] |
Sutkuvienė, S.; Sakalauskaitė, S.; Kuliešienė, N.; Ragelienė, L.; Daugelavičius, R. Biologija 2020, 66, 80.
|
| [26] |
Caruso, A.; Ceramella, J.; Iacopetta, D.; Saturnino, C.; Mauro, M. V.; Bruno, R.; Aquaro, S.; Sinicropi, M. S. Molecules 2019, 24, 1912.
doi: 10.3390/molecules24101912 |
| [27] |
Ramalingam, A.; Sarvanan, J. Int. J. Pharm. Investigation 2020, 10, 594.
|
| [28] |
Du, L. F.; Wang, X. Y.; Cui, G. N.; Xu, B. L. Bioorg. Med. Chem. 2021, 29, 115878.
|
| [29] |
Alsantali, R. I.; Hussein, E. M.; Obaid, R. J.; Morad, M.; Altass, H. M.; Alharbi, A.; Hameed, A. M.; Jassas, R. S.; Abourehab, M. A. S.; Asghar, B. H.; Moussa, Z.; Ahmed, S. A. Arab. J. Chem. 2020, 13, 5451.
|
| [30] |
Wu, D. X.; Liu, M. H.; Li, Z.; Dang, M. M.; Liu, X. P.; Li, J. M.; Huang, L.; Ren, Y. G.; Zhang, Z.; Liu, W. D.; Liu, A. P. Heterocycl. Commun. 2019, 25, 8.
doi: 10.1515/hc-2019-0003 |
| [31] |
Abdel-Galil, E.; Girges, M. M.; Said, G. E. ChemistrySelect 2020, 5, 3075.
doi: 10.1002/slct.v5.10 |
| [32] |
Shinde, V.; Mahulikar, P.; Mhaske, P. C.; Chakraborty, S.; Choudhari, A.; Phalle, S.; Choudhari, P.; Sarkar, D. Med. Chem. Res. 2019, 28, 805.
doi: 10.1007/s00044-019-02310-y |
| [33] |
Singh, I. P.; Gupta, S.; Kumar, S. Med. Chem. 2020, 16, 4.
doi: 10.2174/1573406415666190614101253 |
| [34] |
Morcoss, M. M.; Abdelhafez, E. S. M. N.; Abdel-Rahman, H. M.; Abdel-Aziz, M.; El-Ella, D. A. A. J. Adv. Biomed. Pharm. Sci. 2020, 3, 45.
|
| [35] |
Özdemir, A.; Turanlý, S.; Çalýþkan, B.; Arka, M.; Banoglu, E. Pharm. Chem. J. 2020, 53, 1036.
|
| [36] |
Tokuhara, H.; Imaeda, Y.; Fukase, Y.; Iwanaga, K.; Taya, N.; Watanabe, K.; Kanagawa, R.; Matsuda, K.; Kajimoto, Y.; Kusumoto, K.; Kondo, M.; Snell, G.; Behnke, C. A.; Kuroita, T. Bioorg. Med. Chem. 2018, 26, 3261.
|
| [37] |
Adegboye, A. A.; Khan, K. M.; Salar, U.; Aboaba, S. A.; Kanwal, Chigurupati, S.; Fatima, I.; Taha, M.; Wadood, A.; Mohammad, J. I.; Khan, H.; Perveen, S. Eur. J. Med. Chem. 2018, 150, 248.
|
| [38] |
Brown, A. D.; Bagal, S. K.; Blackwell, P.; Blakemore, D. C.; Brown, B.; Bungay, P. J.; Corless, M.; Crawforth, J.; Fengas, D.; Fenwick, D. R.; Gray, V.; Kemp, M.; Klute, W.; Sanz, L. M.; Miller, D.; Murata, Y.; Payne, C. E.; Skerratt, S.; Stevens, E. B.; Warmus, J. S. Bioorg. Med. Chem. 2019, 27, 230.
doi: 10.1016/j.bmc.2018.12.002 |
| [39] |
Antoci, V.; Cucu, D.; Zbancioc, G.; Moldoveanu, C.; Mangalagiu, V.; Amariucai-Mantu, D.; Aricu, A.; Mangalagiu, I. I. Future Med. Chem. 2020, 12, 207.
doi: 10.4155/fmc-2019-0063 |
| [40] |
Zhang, T. R.; Liu, Q. Q.; Ren, Y. J. Tetrahedron 2020, 76, 131027.
doi: 10.1016/j.tet.2020.131027 |
| [41] |
Spasov, A. A.; Zhukovskaya, O. N.; Maltsev, D. V.; Miroshnikov, M. V.; Skripka, M. O.; Sultanova, K. T.; Morkovnik, A. S. Russ. J. Bioorg. Chem. 2020, 46, 107.
|
| [42] |
Rahim, F.; Ullah, K.; Ullah, H.; Wadood, A.; Taha, M.; Rehman, A. U.; Uddin, I.; Ashraf, M.; Shaukat, A.; Rehman, W.; Hussain, S.; Khan, K. M. Bioorg. Chem. 2015, 58, 81.
doi: 10.1016/j.bioorg.2014.12.001 |
| [43] |
Zhan, J. Y.; Ma, K.; Zheng, Q. C.; Yang, G. H.; Zhang, H. X. J. Biomol. Struct. Dyn. 2019, 37, 1724.
|
| [44] |
Rahima, F.; Tariq, S.; Taha, M.; Ullah, H.; Zaman, K.; Uddin, I.; Wadood, A.; Khan, A. A.; Rehman, A. U.; Uddin, N.; Zafar, S.; Shah, S. A. A. Bioorg. Chem. 2019, 92, 103284.
doi: 10.1016/j.bioorg.2019.103284 |
| [45] |
Zhang, Q.; Ding, D. R.; Zeng, S. X.; Ye, Q. Z.; Lu, H. PloS One 2012, 7, e46294.
doi: 10.1371/journal.pone.0046294 |
| [46] |
Ali, R. S.; Saad, H. A. Molecules 2018, 23, 693.
doi: 10.3390/molecules23030693 |
| [47] |
Sivendran, S.; Jones, V.; Sun, D. Q.; Wang, Y.; Grzegorzewicz, A. E.; Scherman, M. S.; Napper, A. D.; McCammon, J. A.; Lee, R. E.; Diamond, S. L.; McNeil, M. Bioorg. Med. Chem. 2010, 18, 896.
doi: 10.1016/j.bmc.2009.11.033 |
| [48] |
Upadhyay, K.; Manvar, A.; Loddo, R.; Colla, P. L.; Virsodiya, V.; Trivedi, J.; Chaniyara, R.; Shah, A. Med. Chem. Res. 2013, 22, 3675.
|
| [49] |
Li, Y. J.; Wang, S. Y.; Jin, K.; Gao, L. X.; Sheng, L.; Zhang, N.; Liu, J.-H.; Li, J. Chin. J. Org. Chem. 2019, 39, 491. (in Chinese).
|
|
( 李英俊, 王思远, 靳焜, 高立信, 盛丽, 张楠, 刘继红, 李佳, 有机化学, 2019, 39, 491.)
|
|
| [50] |
Li, Y. J.; Liu, X. J.; Liu, J. H.; Gao, L. X.; Jin, K.; Sheng, L.; Yang, H. J.; Lin, L. D.; Li, J. Chin. J. Org. Chem. 2020, 40, 478. (in Chinese).
|
|
( 李英俊, 刘雪洁, 刘季红, 高立信, 靳焜, 盛丽, 杨鸿境, 林乐弟, 李佳, 有机化学, 2020, 40, 478.)
|
|
| [51] |
Li, Y. J.; Zhao, Y.; Jin, K.; Gao, L. X.; Sheng, L.; Liu, X. J.; Yang, H. J.; Lin, L. D.; Li, J. Chin. J. Org. Chem. 2019, 39, 2599. (in Chinese).
|
|
( 李英俊, 赵月, 靳焜, 高立信, 盛丽, 刘雪洁, 杨鸿境, 林乐弟, 李佳, 有机化学, 2019, 39, 2599.)
|
|
| [52] |
Hidrazitler, A. K.; Karakterizasyonu, S. Hacettepe J. Biol. Chem. 2018, 46, 169.
|
| [53] |
Ning, Y.-C. Structural Identification of Organic Compounds and Organic Spectroscopy, 2nd ed., Science Press, Beijing, 2000, p. 53. (in Chinese).
|
|
( 宁永成, 有机化合物结构鉴定与有机波谱学, 第2版, 科学出版社, 北京, 2000, p. 53.)
|
|
| [54] |
Li, Y. J.; Zhang, N.; Jin, K.; Xu, Y. T.; Wang, S. Y.; Zhou, X. X. Chin. J. Org. Chem. 2017, 37, 2640. (in Chinese).
|
|
( 李英俊, 张楠, 靳焜, 许永廷, 王思远, 周晓霞, 有机化学, 2017, 37, 2640.)
|
|
| [55] |
Li, Y. J.; Liu, L. J.; Jin, K.; Sun, S. Q.; Xu, Y. T. Acta Chim. Sinica 2010, 68, 1577. (in Chinese).
|
|
( 李英俊, 刘丽军, 靳焜, 孙淑琴, 许永廷, 化学学报, 2010, 68, 1577.)
|
|
| [56] |
Li, Y. J.; Yang, H. J.; Jin, K.; Lin, L. D.; Liu, X. J. Chin. J. Magn. Reson. 2020, 37, 496. (in Chinese).
|
|
李英俊, 杨鸿境, 靳焜, 林乐弟, 刘雪洁, 波谱学杂志, 2020, 37, 496. (in Chinese).
|
|
| [57] |
Li, Y. J.; Li, J. Y.; Xu, Y. T.; Jin, K.; Cao, X. Chin. J. Org. Chem. 2017, 37, 896. (in Chinese).
|
|
( 李英俊, 李继阳, 许永廷, 靳焜, 曹欣, 有机化学, 2017, 37, 896.)
|
|
| [58] |
Grigoras, M.; Antonoaia, N.-C. Eur. Polym. J. 2005, 41, 1079.
doi: 10.1016/j.eurpolymj.2004.11.019 |
| [59] |
Sun, L. P.; Shen, Q.; Piao, H. H.; Ma, W. P.; Gao, L. X.; Zhang, W.; Nan, F. J.; Li, J.; Piao, H. R. Eur. J. Med. Chem. 2011, 46, 3630.
|
| [1] | 邹发凯, 王能中, 姚辉, 王慧, 刘明国, 黄年玉. 1β-/3R-芳基硫代糖的区域与立体选择性合成[J]. 有机化学, 2024, 44(2): 593-604. |
| [2] | 吴思敏, 唐嘉欣, 周于佳, 徐学涛, 张昊星, 王少华. 2β-Acetoxyferruginol去醋酸基骨架衍生物抑制α-葡萄糖苷酶活性研究[J]. 有机化学, 2024, 44(2): 613-621. |
| [3] | 李路瑶, 贺忠文, 张振国, 贾振华, 罗德平. 三芳基碳正离子在有机合成中的应用[J]. 有机化学, 2024, 44(2): 421-437. |
| [4] | 梅青刚, 李清寒. 可见光促进C(3)(杂)芳硫基吲哚化合物的合成研究进展[J]. 有机化学, 2024, 44(2): 398-408. |
| [5] | 冯康博, 陈炯, 古双喜, 王海峰, 陈芬儿. 全连续流反应技术在药物合成中的新进展(2019~2022)[J]. 有机化学, 2024, 44(2): 378-397. |
| [6] | 李鹏辉, 谢青洋, 万福贤, 张元红, 姜林. 含环丙基的新型取代嘧啶-5-甲酰胺的合成及杀菌活性研究[J]. 有机化学, 2024, 44(2): 650-656. |
| [7] | 杨维清, 葛宴兵, 陈元元, 刘萍, 付海燕, 马梦林. 1,8-萘酰亚胺衍生物的设计、合成及其对半胱氨酸的识别研究[J]. 有机化学, 2024, 44(1): 180-194. |
| [8] | 赵茜帆, 陈永正, 张世明. 碳基非金属催化剂在有机合成领域的应用及机理研究[J]. 有机化学, 2024, 44(1): 137-147. |
| [9] | 陈珊, 陈志林, 胡琼, 蒙艳双, 黄悦, 陶萍芳, 卢丽如, 黄国保. 含双硫脲基团分子钳在非极性溶剂中识别中性分子[J]. 有机化学, 2024, 44(1): 277-281. |
| [10] | 王化坤, 任晓龙, 宣宜宁. 卤盐催化的α,β-环氧羧酸酯与异氰酸酯[3+2]环加成反应研究[J]. 有机化学, 2024, 44(1): 251-258. |
| [11] | 金玉坤, 任保轶, 梁福顺. 可见光介导的三氟甲基的选择性C-F键断裂及其在偕二氟类化合物合成中的应用[J]. 有机化学, 2024, 44(1): 85-110. |
| [12] | 马翠云, 罗海澜, 张福华, 郭丹, 陈树兴, 王飞. 3-Pyrrolyl BODIPY的绿色生物合成、光物理性质及应用研究[J]. 有机化学, 2024, 44(1): 216-223. |
| [13] | 王博珍, 张婕, 粘春惠, 金茗茗, 孔苗苗, 李物兰, 何文斐, 吴建章. 含有3,4-二氯苯基的酰胺类化合物的合成及抗肿瘤活性研究[J]. 有机化学, 2024, 44(1): 232-241. |
| [14] | 于士航, 刘嘉威, 安碧玉, 边庆花, 王敏, 钟江春. 黑腹尼虎天牛接触性信息素的不对称合成[J]. 有机化学, 2024, 44(1): 301-308. |
| [15] | 李阳, 袁锦鼎, 赵頔. 低共熔溶剂1,3-二甲基脲/L-(+)-酒石酸中(E)-2-苯乙烯基喹啉-3-羧酸类衍生物的绿色合成[J]. 有机化学, 2023, 43(9): 3268-3276. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||