有机化学 ›› 2026, Vol. 46 ›› Issue (5): 2017-2030.DOI: 10.6023/cjoc202602011 上一篇 下一篇
研究论文
胡泽宇a,†, 邓家诚a,†, 李延a,†, 沈磊b, 毛辉b,*( ), 周列锦a,*(
), 周列锦a,*( )
)
收稿日期:2026-02-10
修回日期:2026-02-22
发布日期:2026-03-06
作者简介:†共同第一作者
基金资助:
Zeyu Hua, Jiacheng Denga, Yan Lia, Lei Shenb, Hui Maob,*( ), Liejin Zhoua,*(
), Liejin Zhoua,*( )
)
Received:2026-02-10
Revised:2026-02-22
Published:2026-03-06
Contact:
* E-mail: ljzhou@zjnu.cn;
maohui1988@126.com
About author:These authors contributed equally to this work
Supported by:文章分享
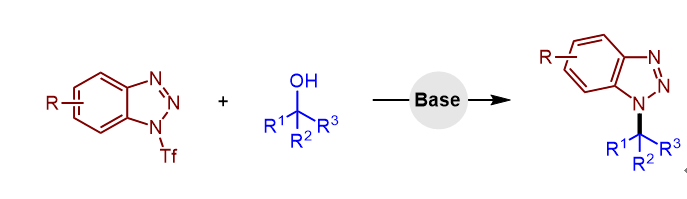
报道了一种实用且高效的sp3-C—N键构建方法, 该方法通过N1-三氟甲磺酰苯并三唑与易于获得的醇类底物实现C—N偶联反应. 该转化在温和条件下即可进行, 并表现出良好的底物普适性. 此外, 该反应体系对吡唑、吲唑等其他杂环底物同样具有良好的兼容性.
胡泽宇, 邓家诚, 李延, 沈磊, 毛辉, 周列锦. N1-三氟甲磺酰苯并三唑与醇的脱氧胺化反应: sp3-C—N键的构建[J]. 有机化学, 2026, 46(5): 2017-2030.
Zeyu Hu, Jiacheng Deng, Yan Li, Lei Shen, Hui Mao, Liejin Zhou. Deoxyamination of N1-Triflylbenzotriazoles with Alcohols for sp3-C—N Bond Formation[J]. Chinese Journal of Organic Chemistry, 2026, 46(5): 2017-2030.
| Entry a | Base | Solvent | Yieldb/% |
|---|---|---|---|
| 1 | Cs2CO3 | DCE | 68 |
| 2 | Cs2CO3 | THF | 67 |
| 3 | Cs2CO3 | PhCl | 62 |
| 4 | Cs2CO3 | Acetone | 17 |
| 5 | Cs2CO3 | MeCN | 44 |
| 6 | Cs2CO3 | 1,4-Dioxane | 77 |
| 7 | NaOMe | 1,4-Dioxane | ndc |
| 8 | K2CO3 | 1,4-Dioxane | 40 |
| 9 | NaOH | 1,4-Dioxane | 13 |
| 10 | t-BuOK | 1,4-Dioxane | 34 |
| 11 | DBU | 1,4-Dioxane | 19 |
| 12d | Cs2CO3 | 1,4-Dioxane | 70 |
| 13e | Cs2CO3 | 1,4-Dioxane | 92 |
| 14f | Cs2CO3 | 1,4-Dioxane | 99g |
| Entry a | Base | Solvent | Yieldb/% |
|---|---|---|---|
| 1 | Cs2CO3 | DCE | 68 |
| 2 | Cs2CO3 | THF | 67 |
| 3 | Cs2CO3 | PhCl | 62 |
| 4 | Cs2CO3 | Acetone | 17 |
| 5 | Cs2CO3 | MeCN | 44 |
| 6 | Cs2CO3 | 1,4-Dioxane | 77 |
| 7 | NaOMe | 1,4-Dioxane | ndc |
| 8 | K2CO3 | 1,4-Dioxane | 40 |
| 9 | NaOH | 1,4-Dioxane | 13 |
| 10 | t-BuOK | 1,4-Dioxane | 34 |
| 11 | DBU | 1,4-Dioxane | 19 |
| 12d | Cs2CO3 | 1,4-Dioxane | 70 |
| 13e | Cs2CO3 | 1,4-Dioxane | 92 |
| 14f | Cs2CO3 | 1,4-Dioxane | 99g |
| [1] |
(a)
doi: 10.1021/acs.chemrev.4c00094 |
|
(b)
doi: 10.1002/anie.v62.4 |
|
|
(c)
doi: 10.1039/D3CS00942D |
|
| [2] |
(a)
doi: 10.1039/C6GC00938G pmid: 22576362 |
|
(b)
doi: 10.31635/ccschem.022.202101682 pmid: 22576362 |
|
|
(c)
doi: 10.1021/jacs.5b05076 pmid: 22576362 |
|
|
(d)
doi: 10.1002/anie.v56.11 pmid: 22576362 |
|
|
(e)
doi: 10.1021/acs.chemrev.0c00736 pmid: 22576362 |
|
|
(f)
doi: 10.1039/C5CS00144G pmid: 22576362 |
|
|
(g)
doi: 10.1039/c2cs35024f pmid: 22576362 |
|
|
(h)
doi: 10.1039/C8CC03856B pmid: 22576362 |
|
|
(i)
doi: 10.1021/acs.orglett.2c03084 pmid: 22576362 |
|
|
(j)
doi: 10.1021/jacs.4c04477 pmid: 22576362 |
|
| [3] |
(a)
doi: 10.1016/j.chempr.2021.10.023 |
|
(b)
doi: 10.1039/c4ob00032c |
|
| [4] |
doi: 10.1016/S0040-4020(01)00722-0 |
| [5] |
(a)
doi: 10.1246/bcsj.40.935 |
|
(b)
doi: 10.1246/bcsj.40.2380 |
|
|
(c)
doi: 10.1021/cr800278z |
|
| [6] |
(a)
doi: 10.1021/op0601013 |
|
(b)
doi: 10.1351/pac200476030507 |
|
|
(c)
|
|
|
(d)
doi: 10.1021/cr300389u |
|
| [7] |
(a)
doi: 10.1021/acs.orglett.9b00757 pmid: 38374064 |
|
(b)
doi: 10.1038/s41467-024-45875-y pmid: 38374064 |
|
| [8] |
(a)
doi: 10.1016/j.ejmech.2014.09.089 pmid: 25293580 |
|
(b)
pmid: 25293580 |
|
| [9] |
(a)
doi: 10.1021/cr900204u pmid: 11848906 |
|
(b)
pmid: 11848906 |
|
| [10] |
|
| [11] |
(a)
doi: 10.1021/cr200031r pmid: 19243194 |
|
(b)
pmid: 19243194 |
|
|
(c)
pmid: 19243194 |
|
|
(d)
doi: 10.1002/chem.v26.52 pmid: 19243194 |
|
|
(e)
doi: 10.1002/tcr.v20.7 pmid: 19243194 |
|
|
(f)
doi: 10.1055/s-0040-1707253 pmid: 19243194 |
|
|
(g)
doi: 10.1002/jlac.v291:1/2 pmid: 19243194 |
|
|
(h)
doi: 10.1021/ja01009a056 pmid: 19243194 |
|
|
(i)
doi: 10.1021/ol900113f pmid: 19243194 |
|
|
(j)
doi: 10.1016/j.tetlet.2009.12.030 pmid: 19243194 |
|
|
(k)
doi: 10.1002/anie.v56.3 pmid: 19243194 |
|
|
(l)
doi: 10.1021/acscatal.7b01025 pmid: 19243194 |
|
|
(m)
doi: 10.1039/C7CC07543J pmid: 19243194 |
|
|
(n)
doi: 10.1039/C8QO00778K pmid: 19243194 |
|
|
(o)
doi: 10.1021/acs.orglett.7b03184 pmid: 19243194 |
|
|
(p)
doi: 10.1002/anie.v58.33 pmid: 19243194 |
|
|
(q)
doi: 10.1021/jacs.1c07212 pmid: 19243194 |
|
| [12] |
(a)
doi: 10.1021/acs.orglett.2c02064 |
|
(b)
doi: 10.1039/D3QO00916E |
|
| [13] |
doi: 10.1021/ja01150a029 |
| [14] |
|
| [15] |
doi: 10.1021/acs.orglett.3c00162 |
| [16] |
doi: 10.1039/C7SC00367F |
| [17] |
doi: 10.1002/anie.v56.36 |
| [1] | 张文杰, 段正超, 胡向平. 手性P-N-O配体促进的铱催化芳香酮不对称氢化反应[J]. 有机化学, 2026, 46(5): 2129-2137. |
| [2] | 王文博, 陈贵川, 王锦博, 赵阳, 王琪琳. 吡啶鎓1,4-两性离子硫醇盐在有机合成中的应用[J]. 有机化学, 2026, 46(5): 1845-1870. |
| [3] | 马耀鹏, 朱辰龙, 孙炳峰. 菲诺洛芬的高效合成[J]. 有机化学, 2026, 46(5): 2036-2043. |
| [4] | 郭子心, 许盼, 徐晓锋, 刘振兴. Turbo格氏试剂介导的酰基硅羰基还原反应[J]. 有机化学, 2026, 46(5): 2031-2035. |
| [5] | 夏春年, 陈凯林, 金诺琪, 南欣卉, 汪兵洋, 吴馨怡, 孙强盛, 孙伟. 仿生四齿胺基吡啶锰催化剂/H2O2的醇类化合物高效氧化: 间歇和连续流研究[J]. 有机化学, 2026, 46(3): 986-992. |
| [6] | 顾运琼, 姚嘉伟, 钟智薇, 文舒婷, 杨坤, 刘亚璐, 甘春芳. 氨基酸修饰的孕甾衍生物设计合成及抗肿瘤活性研究[J]. 有机化学, 2026, 46(2): 545-553. |
| [7] | 刘辉杨, 陈都, 苏毅进, 张鹏, 刘超. 频哪醇硼烷快速可控还原酰氯合成醛[J]. 有机化学, 2026, 46(2): 564-569. |
| [8] | 陈奇姝, 杨泊, 稂琪伟, 丁小兵, 李秀秀, 张绪穆. 铱/f-Amphox催化吡啶酮的不对称氢化反应制备手性吡啶醇★[J]. 有机化学, 2025, 45(9): 3326-3334. |
| [9] | 赵友学, 李兮若, 孟洛冰, 李春秀, 范贵生, 许建和. 醇脱氢酶/羰基还原酶与多底物分子适配性研究的进展★[J]. 有机化学, 2025, 45(9): 3175-3185. |
| [10] | 刘丽强, 南军, 段兴宇, 王昊龙, 蔡巷, 王梓诚, 张恩泽, 孙彦民. 三齿P配体钌络合物催化脂肪醇胺N-烷基化反应研究[J]. 有机化学, 2025, 45(8): 2983-2988. |
| [11] | 谢昊池, 秦永康, 杨婷, 李湖进, 孙佳嘉, 钱明成, 赵帅, 侯亚男, 陈新. 可见光介导的烯醇和重氮化合物之间的O—H键官能团化反应[J]. 有机化学, 2025, 45(8): 3004-3016. |
| [12] | 王小燕, 徐长明. Brønsted酸催化烯丙醇的伯胺化反应[J]. 有机化学, 2025, 45(8): 2932-2937. |
| [13] | 杜顺利, 王雅雅, 郭家铭, 徐雪薇, 彭新华. 铜有机磷配合物催化芳烯烃β-C位的硼化反应研究[J]. 有机化学, 2025, 45(7): 2435-2443. |
| [14] | 李旻昊, 王泽溟, 黄庆, 左伟伟. 钴(II)催化的酮不对称转移氢化反应[J]. 有机化学, 2025, 45(7): 2451-2460. |
| [15] | 张艮红, 余若曦, 陈跃刚. 光/电促进醇及其衍生物C—O键活化构筑C(sp2)—C(sp3)键研究进展[J]. 有机化学, 2025, 45(5): 1548-1568. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||