有机化学 ›› 2022, Vol. 42 ›› Issue (12): 4282-4291.DOI: 10.6023/cjoc202211011 上一篇 下一篇
所属专题: 自由基化学专辑
研究论文
收稿日期:2022-11-08
修回日期:2022-11-22
发布日期:2022-11-28
通讯作者:
王树良, 姜波
基金资助:
Xiaoshuang Ji, Rong Fu, Shuliang Wang( ), Wenjuan Hao, Bo Jiang(
), Wenjuan Hao, Bo Jiang( )
)
Received:2022-11-08
Revised:2022-11-22
Published:2022-11-28
Contact:
Shuliang Wang, Bo Jiang
Supported by:文章分享
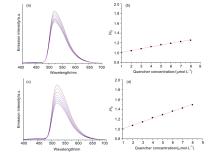
1,n-烯炔合成子参与的涉及原子转移过程的自由基环化碳卤官能化是一类有效且实用的方法, 它通过利用原子转移试剂中碳卤σ键的自由基龟裂及其在烯炔母核π键上的移位来实现卤代环系结构的有序构建, 展现了完全原子经济性. 近年来, 该类反应成为研究热点之一. 报道了一类通用可见光催化的酚/芳胺联-1,6-烯炔和全卤代甲烷(如三氯溴甲烷或四溴化碳)参与的环化碳卤官能团化反应, 在温和且无氧化剂条件下原子经济且高效地合成了一系列作为主要立体异构体的(Z)-2,3-二氢苯并呋喃和(Z)-吲哚啉衍生物, 收率优良, 其中大部分产物仅以(Z)-构型被合成. 该反应具有底物范围广、官能团耐受性高、高原子经济以及高立体选择性等优点, 为2,3-二氢苯并呋喃和吲哚啉杂环的合成提供一类绿色、原子经济、高效的合成策略.
季晓霜, 付荣, 王树良, 郝文娟, 姜波. 可见光驱动酚/芳胺联-1,6-烯炔与全卤代甲烷的Kharasch反应[J]. 有机化学, 2022, 42(12): 4282-4291.
Xiaoshuang Ji, Rong Fu, Shuliang Wang, Wenjuan Hao, Bo Jiang. Visible-Light-Driven Photocatalytic Kharasch Reaction of Phenol/ Arylamine-Linked 1,6-Enynes with Perhalogenated Methane[J]. Chinese Journal of Organic Chemistry, 2022, 42(12): 4282-4291.
| Entry | Variation from the established conditions | Yieldb/% |
|---|---|---|
| 1 | None | 92 |
| 2 | NaOAc instead of K2HPO4 | 83 |
| 3 | K3PO4 instead of K2HPO4 | 87 |
| 4 | DMAP instead of K2HPO4 | 82 |
| 5 | TEA instead of K2HPO4 | 75 |
| 6 | DMF instead of EtOH | 86 |
| 7 | DCE instead of EtOH | 61 |
| 8 | Acetone instead of EtOH | 37 |
| 9 | 1,4-Dioxane instead of EtOH | 71 |
| 10 | Ru(ppy)2Cl instead of fac-Ir(ppy)3 | 78 |
| 11 | Eosin Y instead of fac-Ir(ppy)3 | 73 |
| 12 | 4CzIPN instead of fac-Ir(ppy)3 | 66 |
| 13 | 0.10 mol% of fac-Ir(ppy)3 | 92 |
| Entry | Variation from the established conditions | Yieldb/% |
|---|---|---|
| 1 | None | 92 |
| 2 | NaOAc instead of K2HPO4 | 83 |
| 3 | K3PO4 instead of K2HPO4 | 87 |
| 4 | DMAP instead of K2HPO4 | 82 |
| 5 | TEA instead of K2HPO4 | 75 |
| 6 | DMF instead of EtOH | 86 |
| 7 | DCE instead of EtOH | 61 |
| 8 | Acetone instead of EtOH | 37 |
| 9 | 1,4-Dioxane instead of EtOH | 71 |
| 10 | Ru(ppy)2Cl instead of fac-Ir(ppy)3 | 78 |
| 11 | Eosin Y instead of fac-Ir(ppy)3 | 73 |
| 12 | 4CzIPN instead of fac-Ir(ppy)3 | 66 |
| 13 | 0.10 mol% of fac-Ir(ppy)3 | 92 |

| [1] |
(a) Chen, C.; Weisel, M. Synlett 2013, 24, 189.
doi: 10.1055/s-0032-1317704 |
|
(b) Zheng, S.-L.; Yu, W.-Y.; Xu, M.-X.; Che, C.-M. Tetrahedron Lett. 2003, 44, 1445.
doi: 10.1016/S0040-4039(02)02860-5 |
|
| [2] |
(a) Macías, F. A.; Varela, R. M.; Torres, A.; Molinillo, J. M. G. J. Nat. Prod. 1999, 62, 1636.
doi: 10.1021/np990249y |
|
(b) Sarkar, D.; Ghosh, M. K. Tetrahedron Lett. 2017, 58, 4336.
doi: 10.1016/j.tetlet.2017.09.081 |
|
| [3] |
Beldjoudi, N.; Mambu, L.; Labaïed, M.; Grellier, P.; Ramanitrahasimbola, D.; Rasoanaivo, P.; Martin, M. T.; Frappier, F. J. Nat. Prod. 2003, 66, 1447.
pmid: 14640516 |
| [4] |
(a) Iida, T.; Ito, K. Phytochemistry 1983, 22, 763.
doi: 10.1016/S0031-9422(00)86979-4 |
|
(b) Horne, D. A.; Yakushijin, K.; Buchi, G. Tetrahedron Lett. 1999, 40, 5443.
doi: 10.1016/S0040-4039(99)01063-1 |
|
| [5] |
Nevagi, R. J.; Dighe, S. N.; Dighe, S. N. Eur. J. Med. Chem. 2015, 97, 561.
doi: 10.1016/j.ejmech.2014.10.085 |
| [6] |
Diaz, P. S.; Phatak, S.; Xu, J.; Fronczek, F. R.; Astruc-Diaz, F.; Thompson, C. M.; Cavasotto, C. N.; Naguib, M. ChemMedChem 2009, 4, 1615.
doi: 10.1002/cmdc.200900226 |
| [7] |
Laurita, T.; D’Orsi, R.; Chiummiento, L.; Funicello, M.; Lupattell, P. Synthesis 2020, 52, 1451.
doi: 10.1055/s-0039-1690820 |
| [8] |
(a) Dapkekar, A. B.; Sreenivasulu, C.; Kishore, D. R.; Satyanarayana, G. Asian J. Org. Chem. 2022, 11, e202200012.
|
|
(b) Ohmura, T.; Kusaka, S.; Suginome, M. Chem. Commun. 2021, 57, 13542.
doi: 10.1039/D1CC05684K |
|
|
(c) Zhang, X.; Sivaguru, P.; Zanoni, G.; Han, X.; Tong, M.; Bi, X. ACS Catal. 2021, 11, 14293.
doi: 10.1021/acscatal.1c04523 |
|
| [9] |
(a) Wang, J.-Y.; Hao, W.-J.; Tu, S.-J.; Jiang, B. Org. Chem. Front. 2020, 7, 1743.
doi: 10.1039/D0QO00387E |
|
(b) Zhou, S.; Cai, B.; Hua, C.; Cheng, X.; Lia, L.; Xuan, J. Chin. Chem. Lett. 2021, 32, 2577.
doi: 10.1016/j.cclet.2021.03.010 |
|
| [10] |
Zhou, F.; Cheng, Y.; Liu, X.-P.; Chen, J.-R.; Xiao, W.-J. Chem. Commun. 2019, 55, 3117.
doi: 10.1039/C9CC00727J |
| [11] |
(a) Wu, C.; Cheng, H.-G.; Chen, R.; Chen, H.; Liu, Z.-S.; Zhang, J.; Zhang, Y.; Zhu, Y.; Geng, Z.; Zhou, Q. Org. Chem. Front. 2018, 5, 2533.
doi: 10.1039/C8QO00348C |
|
(b) Li, Y.; Shi, D.; Tang, Y.; He, X.; Xu, S. J. Org. Chem. 2018, 83, 9464.
doi: 10.1021/acs.joc.8b01166 |
|
|
(c) Liu, S.; Zang, Y.; Huang, H.; Sun, J. Org. Lett. 2020, 22, 8219.
doi: 10.1021/acs.orglett.0c02729 |
|
| [12] |
(a) Hu, M.; Song, R.-J.; Li, J.-H. Angew. Chem., Int. Ed. 2015, 54, 608.
pmid: 31465237 |
|
(b) Hu, M.; Liu, B.; Ouyang, X.-H.; Song, R.-J.; Li, J.-H. Adv. Synth. Catal. 2015, 357, 3332.
doi: 10.1002/adsc.201500336 pmid: 31465237 |
|
|
(c) Zhang, J.; Cheng, S.; Cai, Z.; Liu, P.; Sun, P. J. Org. Chem. 2018, 83, 9344.
doi: 10.1021/acs.joc.8b01265 pmid: 31465237 |
|
|
(d) Wu, W.; Yi, S.; Huang, W.; Luo, D.; Jiang, H. Org. Lett. 2017, 19, 2825.
doi: 10.1021/acs.orglett.7b00980 pmid: 31465237 |
|
|
(e) Wang, L.; Zhang, M.; Zhang, Y.; Liu, Q.; Zhao, X.; Li, J.-S.; Luo, Z.; Wei, W. Chin. Chem. Lett. 2020, 31, 67.
doi: 10.1016/j.cclet.2019.05.041 pmid: 31465237 |
|
|
(f) Wang, L.; Zhang, Y.; Zhang, M.; Bao, P.; Lv, X.; Liu, H.-G.; Zhao, X.; Li, J.-S.; Luo, Z.; Wei, W. Tetrahedron Lett. 2019, 60, 1845.
doi: 10.1016/j.tetlet.2019.06.017 pmid: 31465237 |
|
|
(g) Xia, X.-F.; He, W.; Zhang, G.-W.; Wang, D. Org. Chem. Front. 2019, 6, 342.
doi: 10.1039/C8QO01190G pmid: 31465237 |
|
|
(h) Chen, F.; Huang, X.; Yang, C.; Jiang, H.; Zeng, W. J. Org. Chem. 2021, 86, 14572.
doi: 10.1021/acs.joc.1c01437 pmid: 31465237 |
|
|
(i) Xia, X.-F.; Yu, J.; Wang, D. Adv. Synth. Catal. 2018, 360, 562.
doi: 10.1002/adsc.201701258 pmid: 31465237 |
|
|
(j) Wang, L.; Qi, C.; Cheng, R.; Liu, H.; Xiong, W.; Jiang, H. Org. Lett. 2019, 21, 7386.
doi: 10.1021/acs.orglett.9b02698 pmid: 31465237 |
|
|
(k) Zhang, P.; Wang, C.; Cui, M.; Du, M.; Li, W.; Jia, Z.; Zhao, Q. Org. Lett. 2020, 22, 1149.
doi: 10.1021/acs.orglett.9b04681 pmid: 31465237 |
|
|
(l) Zhang, P.; Yu, G.; Zhao, N.; Zhang, S.; Zhang, M.; Wang, L.; Li, Z.; Ying, J.; Gao, X. J. Org. Chem. 2021, 86, 10105.
doi: 10.1021/acs.joc.1c00828 pmid: 31465237 |
|
|
(m) Zhao, M.; He, W.; Zou, L.-H.; Wang, D.; Sun, T.-Y.; Xia, X.-F. Org. Chem. Front. 2021, 8, 643.
doi: 10.1039/D0QO01341B pmid: 31465237 |
|
|
(n) Li, X.; Xu, Z.; Wang, L.; Wang, F.; Yang, J.; Li, P. ChemPhotoChem 2021, 5, 142.
doi: 10.1002/cptc.202000197 pmid: 31465237 |
|
|
(o) Ji, X.-S.; Zuo, H.-D.; Shen, Y.-T.; Hao, W.-J.; Tu, S.-J.; Jiang, B. Chem. Commun. 2022, 58, 10420.
doi: 10.1039/D2CC03922B pmid: 31465237 |
|
| [13] |
(a) Xuan, J.; He, X.-K.; Xiao, W.-J. Chem. Soc. Rev. 2020, 49, 2546.
doi: 10.1039/c9cs00523d pmid: 28585948 |
|
(b) Chen, J.-R.; Hu, X.-Q.; Lu, L.-Q.; Xiao, W.-J. Chem. Soc. Rev. 2016, 45, 2044.
doi: 10.1039/C5CS00655D pmid: 28585948 |
|
|
(c) Mondal, S.; Dumur, F.; Gigmes, D.; Sibi, M.-P.; Bertrand, M.-P.; Nechab, M. Chem. Rev. 2022, 122, 5842.
doi: 10.1021/acs.chemrev.1c00582 pmid: 28585948 |
|
|
(d) Melchiorre, P. Chem. Rev. 2022, 122, 1483.
doi: 10.1021/acs.chemrev.1c00993 pmid: 28585948 |
|
|
(e) Xuan, J.; Studer, A. Chem. Soc. Rev. 2017, 46, 4329.
doi: 10.1039/c6cs00912c pmid: 28585948 |
|
| [14] |
(a) Yang, D.; Yan, Q.; Zhu, E.; Lv, J.; He, W.-M. Chin. Chem. Lett. 2021, 33, 1798.
doi: 10.1016/j.cclet.2021.09.068 |
|
(b) Wang, Z.; Liu, Q.; Liu, R.; Ji, Z.; Li, Y.; Zhao, X.; Wei, W. Chin. Chem. Lett. 2022, 33, 1479.
doi: 10.1016/j.cclet.2021.08.036 |
|
|
(c) Qu, Z.; Wang, P.; Chen, X.; Deng, G.-J.; Huang, H. Chin. Chem. Lett. 2021, 32, 2582.
doi: 10.1016/j.cclet.2021.02.047 |
|
|
(d) Zhao, X.; Wei, W. Chin. Chem. Lett. 2021, 32, 258.
doi: 10.1016/j.cclet.2020.11.034 |
|
|
(e) Wan, Y.; Liu, Q.; Wu, H.; Zhang, Z.; Zhang, G. Org. Chem. Front. 2022, 9, 1634.
doi: 10.1039/D1QO01914G |
|
|
(f) Wang, L.; Xu, T.; Rao, Q.; Zhang, T.-S.; Hao, W.-J.; Tu, S.-J.; Jiang, B. Org. Lett. 2021, 23, 7845.
doi: 10.1021/acs.orglett.1c02865 |
|
|
(g) Cao, X.; Cheng, X.; Xuan, J. Org. Lett. 2018, 20, 449.
doi: 10.1021/acs.orglett.7b03794 |
|
|
(h) Liu, H.; Fan, X.; Hu, J.; Ma, T.; Wang, F.; Yang, J.; Li, D. J. Org. Chem. 2022, 87, 12877.
doi: 10.1021/acs.joc.2c01453 |
|
|
(i) Wang, S.-W.; Yu, J.; Zhou, Q.-Y.; Chen, S.-Y.; Xu, Z.-H.; Tang, S. ACS Sustainable Chem. Eng. 2019, 7, 10154.
doi: 10.1021/acssuschemeng.9b02178 |
|
|
(j) Qi, X.-K.; Zhang, H.; Pan, Z.-T.; Liang, R.-B.; Zhu, C.-M.; Li, J.-H.; Tong, Q.-X.; Gao, X.-W.; Wu, L.-Z.; Zhong, J.-J. Chem. Commun. 2019, 55, 10848.
doi: 10.1039/C9CC04977K |
|
|
(k) Xiao, Q.; Lu, M.; Deng, Y.; Jian, J.-X.; Tong, Q.-X.; Zhong, J.-J. Org. Lett. 2021, 23, 9303.
doi: 10.1021/acs.orglett.1c03700 |
|
| [15] |
(a) Gui, Q.-W.; Teng, F.; Li, Z.-C.; Xiong, Z.-Y.; Jin, X.-F.; Liu, H.-Y.; Lin, Y.-W.; Cao, Z.; He, W.-M. Chin. Chem. Lett. 2021, 32, 1907.
doi: 10.1016/j.cclet.2021.01.021 |
|
(b) Luo, S.-S.; Shen, H.; Li, S.-J.; Cao, T.; Luo, Y.-P.; Zhang, S.; Zhou, T.; Liu, X.-W. Org. Chem. Front. 2022, 9, 2627.
doi: 10.1039/D2QO00392A |
|
|
(c) Zheng, M.; Gao, K.; Zhang, Y.; Lu, H. Org. Chem. Front. 2021, 8, 1830.
doi: 10.1039/D1QO00099C |
|
|
(d) Zhu, Y.; Huang, H.-Y.; He, Y.-Q.; Wang, M.; Wang, X.-Y.; Song, X.-R.; Mao, Z.-J.; Tian, W.-F.; Xiao, Q. Org. Chem. Front. 2022, 9, 1924.
doi: 10.1039/D1QO01841H |
|
|
(e) Song, K.-X.; Qin, X.-Y.; Ma, Z.-X.; Geng, F.-Z.; Hao, W.-J.; Tu, S.-J.; Jiang, B. Org. Chem. Front. 2021, 8, 5681.
doi: 10.1039/D1QO00994J |
|
|
(f) Wu, D.; Hao, W.-J.; Rao, Q.; Lu, Y.; Tu, S.-J.; Jiang, B. Chem. Commun. 2021, 57, 1911.
doi: 10.1039/D0CC07880H |
|
| [16] |
(a) Ji, W.-Z.; Shi, H.-N.; Wei, P.; Hao, W.-J.; Tu, S.-J.; Jiang, B. Adv. Synth. Catal. 2021, 363, 838.
doi: 10.1002/adsc.202001125 |
|
(b) Zheng, J.-L.; Wu, D.; Lin, N.; Liu, Y.-P.; Wang, L.; Zhu, X.-T.; Hao, W.-J.; Wang, S.-L. Jiang, B. Tetrahedron Lett. 2021, 85, 153485.
doi: 10.1016/j.tetlet.2021.153485 |
|
|
(c) Geng, F.-Z.; Wang, S.-C.; Song, K.-X.; Hao, W.-J.; Jiang, B. Chin. J. Org. Chem. 2021, 41, 4815. (in Chinese)
doi: 10.6023/cjoc202106014 |
|
|
( 耿芳洲, 王世超, 宋克贤, 郝文娟, 姜波, 有机化学, 2021, 41, 4815.)
doi: 10.6023/cjoc202106014 |
|
|
(d) Zhu, C.-F.; Zhang, J.; Zhu, Y.-L.; Hao, W.-J.; Tu, S.-J.; Wang, D.-C.; Jiang, B. Org. Chem. Front. 2021, 8, 1952.
doi: 10.1039/D1QO00124H |
|
|
(f) Matsuo, K.; Yamaguchi, E.; Itoh, A. Asian J. Org. Chem. 2018, 7, 2435.
doi: 10.1002/ajoc.201800533 |
|
|
(g) Bag, D.; Kour, H.; Sawant, S. D. Org. Biomol. Chem. 2020, 18, 8278.
doi: 10.1039/D0OB01454K |
| [1] | 赵瑜, 段玉荣, 史时辉, 白育斌, 黄亮珠, 杨晓军, 张琰图, 冯彬, 张建波, 张秋禹. 可见光促进高价碘(III)试剂参与反应的研究进展[J]. 有机化学, 2023, 43(12): 4106-4140. |
| [2] | 耿芳洲, 王世超, 宋克贤, 郝文娟, 姜波. 可见光催化的1,6-烯炔参与的Kharasch加成反应[J]. 有机化学, 2021, 41(12): 4815-4824. |
| [3] | 刘涛, 屈川华, 谢劲, 朱成建. 光诱导原子经济的迭代型末端炔烃氢三氟甲基化和远程C(sp3)-H键官能团化[J]. 有机化学, 2019, 39(6): 1613-1622. |
| [4] | 闫溢哲, 崔畅, 李政. 低价碘催化的氧化偶联反应研究进展[J]. 有机化学, 2018, 38(10): 2501-2518. |
| [5] | 王剑, 崔冬梅. 非贵金属催化的烯烃和炔烃的氢胺化反应研究进展[J]. 有机化学, 2016, 36(6): 1163-1183. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||