有机化学 ›› 2026, Vol. 46 ›› Issue (2): 624-632.DOI: 10.6023/cjoc202506014 上一篇 下一篇
研究论文
收稿日期:2025-06-06
修回日期:2025-09-09
发布日期:2025-10-09
通讯作者:
于凤丽
基金资助:
Jiahao Li, Bing Yuan, Fengli Yu*( )
)
Received:2025-06-06
Revised:2025-09-09
Published:2025-10-09
Contact:
Fengli Yu
Supported by:文章分享
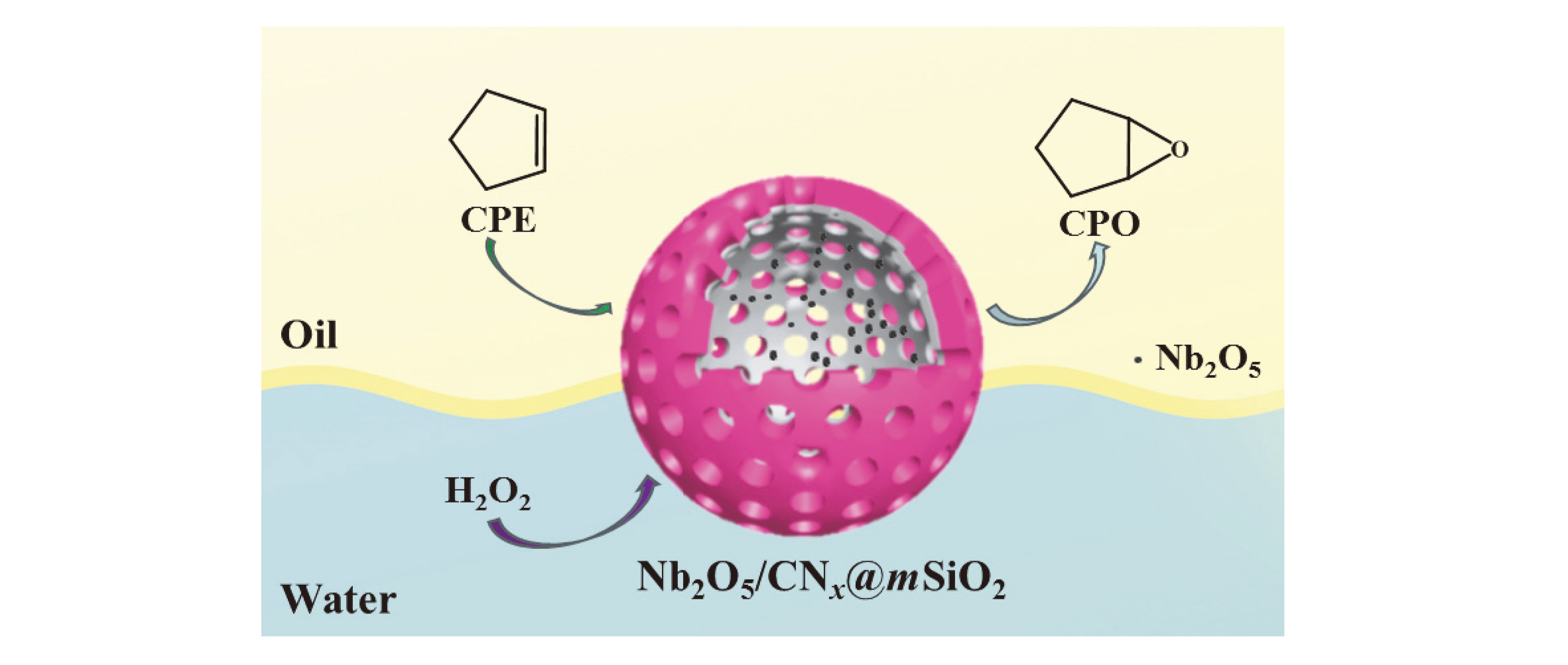
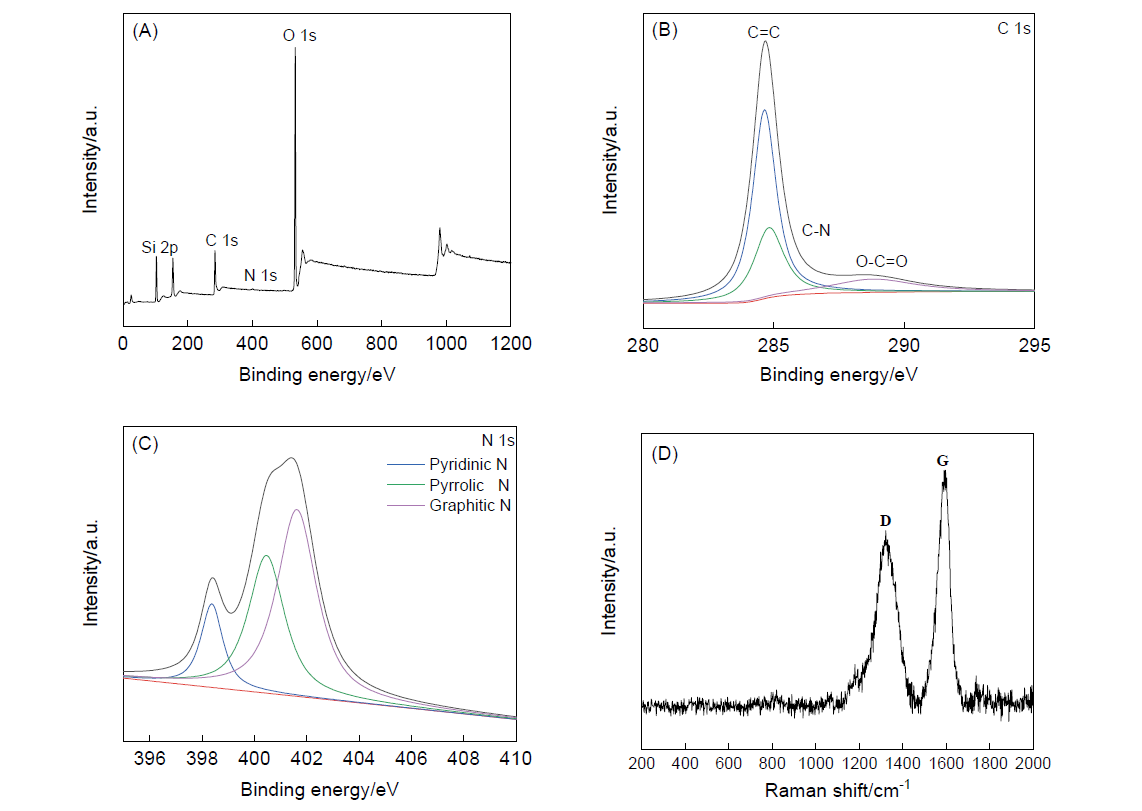
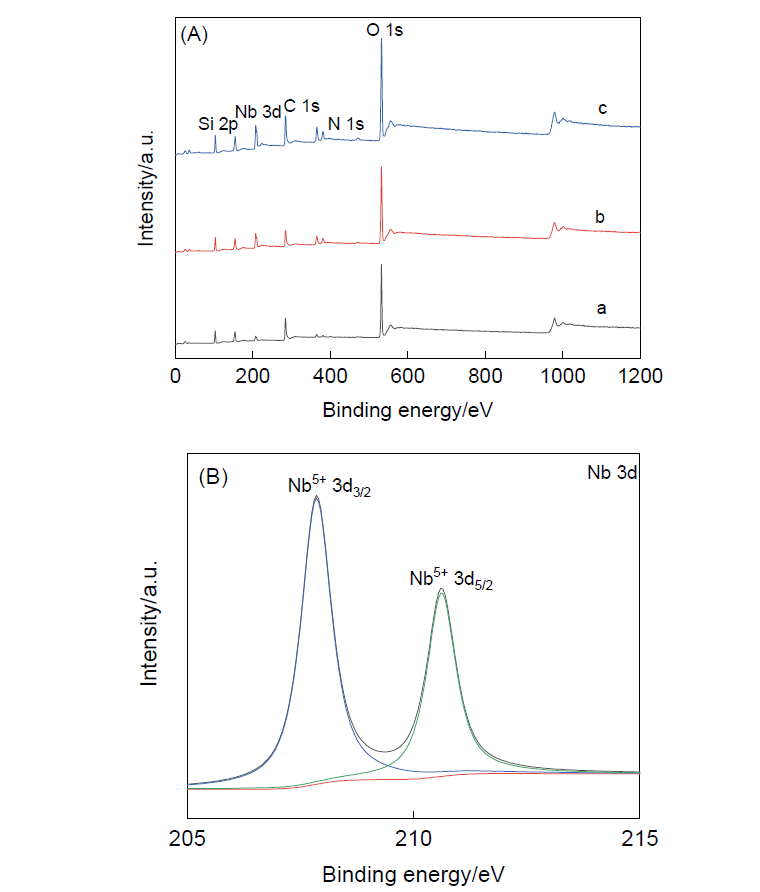
以聚苯并恶嗪为碳源和氮源, 硅酸四乙酯为硅源, 通过限域碳化法合成一种中空的氮化碳@二氧化硅复合材料. 以五氯化铌为铌源, 通过浸渍-煅烧的方法将Nb2O5负载在中空的氮化碳@二氧化硅复合材料上. 以H2O2(水溶液)为氧化剂, 将合成的两亲性催化剂Nb2O5/CNx@mSiO2用于催化环戊烯的环氧化反应. 催化活性中心Nb2O5主要通过氮原子锚定在内部中空的具有疏水性的氮化碳骨架上, 而内部的空腔结构可以富集更多的环戊烯; 外部亲水性的介孔二氧化硅壳不仅可以保护催化活性中心减少其流失, 而且有利于催化剂的分离回收. 在最优反应条件下, 环戊烯转化率为94.0%, 环氧环戊烷选择性为92.1%. 催化剂可以循环使用三次. 本研究为环氧环戊烷的制备提供了一种绿色、高效的新方法, 为两亲性催化剂催化烯烃的水相氧化反应提供了一种新思路.
李佳豪, 袁冰, 于凤丽. 碳@硅复合材料负载的五氧化二铌催化环戊烯环氧化反应研究[J]. 有机化学, 2026, 46(2): 624-632.
Jiahao Li, Bing Yuan, Fengli Yu. Study on Epoxidation of Cyclopentene Catalyzed by Diniobium Pentoxide Loaded on Carbon@Silica Composite Material[J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 624-632.


| Solvent | Conv./% | Selectivity/% | |||
|---|---|---|---|---|---|
| | | | | ||
| DMF | 56.7 | 42.7 | 54.8 | 1.4 | 1.1 |
| MeCN | 90.5 | 74.4 | 7.5 | 7.9 | 10.2 |
| EtOH | 89.5 | 81.5 | 1.8 | 6.6 | 10.1 |
| t-BuOH | 72.3 | 83.2 | 2.5 | 5.7 | 8.6 |
| Acetone | 95.4 | 89.0 | 3.5 | 3.5 | 4.0 |
| Solvent | Conv./% | Selectivity/% | |||
|---|---|---|---|---|---|
| | | | | ||
| DMF | 56.7 | 42.7 | 54.8 | 1.4 | 1.1 |
| MeCN | 90.5 | 74.4 | 7.5 | 7.9 | 10.2 |
| EtOH | 89.5 | 81.5 | 1.8 | 6.6 | 10.1 |
| t-BuOH | 72.3 | 83.2 | 2.5 | 5.7 | 8.6 |
| Acetone | 95.4 | 89.0 | 3.5 | 3.5 | 4.0 |
| Entry | Catalyst | Conv./% | Sel./% |
|---|---|---|---|
| 1 | Nb2O5 | 17.1 | 58.6 |
| 2 | Nb2O5/CNx | 56.6 | 84.4 |
| 3 | Nb2O5/mSiO2 | 84.1 | 89.2 |
| 4 | Nb2O5/CNx@mSiO2b | 94.0 | 92.1 |
| 5 | Nb2O5/CNx@mSiO2c | 76.2 | 94.9 |
| 6 | Nb2O5/CNx@mSiO2d | 66.7 | 94.7 |
| Entry | Catalyst | Conv./% | Sel./% |
|---|---|---|---|
| 1 | Nb2O5 | 17.1 | 58.6 |
| 2 | Nb2O5/CNx | 56.6 | 84.4 |
| 3 | Nb2O5/mSiO2 | 84.1 | 89.2 |
| 4 | Nb2O5/CNx@mSiO2b | 94.0 | 92.1 |
| 5 | Nb2O5/CNx@mSiO2c | 76.2 | 94.9 |
| 6 | Nb2O5/CNx@mSiO2d | 66.7 | 94.7 |
| Entry | Catalyst | Experiment condition | Conv./% | Sel./% | Recycle number | Ref. |
|---|---|---|---|---|---|---|
| 1 | Co@MCM(F) | MeCN, 60 ℃, 3 h | 45.2 | 80.7 | 1 | [ |
| 2 | R-Ti-MWW-PI | MeCN, 60 ℃, 2 h | 97.8 | 99.9 | 6 | [ |
| 3 | TS-1_P-0.05K | MeOH, 40 ℃, 2 h | 52.0 | 98.2 | 5 | [ |
| 4 | Mg0.5Co0.5Fe2O4 | 1,4-Dioxane, 60 ℃, 8 h | 95.2 | 96.3 | 5 | [ |
| 5 | SBA-15-ImCl-PF6-W | No solvent, 35 ℃, 3 h | 53.0 | 99.0 | 7 | [ |
| 6 | Nb2O5/CNx@mSiO2 | Acetone, 35 ℃, 4 h | 94.0 | 92.1 | 3 | This work |
| Entry | Catalyst | Experiment condition | Conv./% | Sel./% | Recycle number | Ref. |
|---|---|---|---|---|---|---|
| 1 | Co@MCM(F) | MeCN, 60 ℃, 3 h | 45.2 | 80.7 | 1 | [ |
| 2 | R-Ti-MWW-PI | MeCN, 60 ℃, 2 h | 97.8 | 99.9 | 6 | [ |
| 3 | TS-1_P-0.05K | MeOH, 40 ℃, 2 h | 52.0 | 98.2 | 5 | [ |
| 4 | Mg0.5Co0.5Fe2O4 | 1,4-Dioxane, 60 ℃, 8 h | 95.2 | 96.3 | 5 | [ |
| 5 | SBA-15-ImCl-PF6-W | No solvent, 35 ℃, 3 h | 53.0 | 99.0 | 7 | [ |
| 6 | Nb2O5/CNx@mSiO2 | Acetone, 35 ℃, 4 h | 94.0 | 92.1 | 3 | This work |
| [1] |
doi: 10.1134/S0965544112050088 |
| [2] |
doi: 10.1016/j.apcata.2004.12.029 |
| [3] |
doi: 10.1016/j.jorganchem.2019.121073 |
| [4] |
doi: 10.3389/fchem.2020.585347 |
| [5] |
doi: 10.3390/catal12101241 |
| [6] |
doi: 10.1016/j.apcatb.2022.121461 |
| [7] |
doi: 10.1016/j.cattod.2018.04.002 |
| [8] |
doi: 10.1002/chem.v25.16 |
| [9] |
doi: 10.1021/acs.langmuir.1c00893 |
| [10] |
|
| [11] |
|
| [12] |
doi: 10.1016/j.catcom.2023.106754 |
| [13] |
doi: 10.1016/j.jcis.2024.03.076 |
| [14] |
doi: 10.1039/C7GC00298J |
| [15] |
doi: 10.1002/cctc.v9.23 |
| [16] |
doi: 10.1039/C9CY01398A |
| [17] |
doi: 10.1016/j.cattod.2022.08.021 |
| [18] |
doi: 10.1016/j.jcis.2023.12.069 |
| [19] |
doi: 10.1039/C8CC03267J |
| [20] |
doi: 10.1039/D1NR05695F |
| [21] |
doi: 10.1016/j.apsusc.2021.151829 |
| [22] |
doi: 10.1016/j.indcrop.2022.115140 |
| [23] |
doi: 10.1016/j.cej.2023.143110 |
| [24] |
doi: 10.1039/D3TA07853A |
| [25] |
doi: 10.1021/acs.chemrev.6b00374 |
| [26] |
doi: 10.1021/acscatal.7b02335 |
| [27] |
|
| [28] |
doi: 10.15376/biores |
| [29] |
doi: 10.1016/j.fuel.2022.124400 |
| [30] |
doi: 10.1039/D1NJ02427B |
| [31] |
doi: 10.1016/j.cjche.2022.07.004 |
| [32] |
|
| [33] |
doi: 10.1016/j.materresbull.2024.112927 |
| [34] |
doi: 10.1002/slct.v1.20 |
| [35] |
doi: 10.1002/slct.v6.9 |
| [1] | 高源, 刘岩, 李师伍, 唐天胜, 孟钰坤, 赵志飞. 氮杂环卡宾(NHC)催化[3+2]环加成反应高非对映选择性地构建环戊酮-螺环氧化吲哚化合物[J]. 有机化学, 2026, 46(3): 1008-1016. |
| [2] | 刘涛, 周永博, 郎同庆, 牛海波, 陈飞, 杜智宏, 薄春博, 李敏, 刘宁. 席夫碱型Fe(III)配合物催化二氧化碳和环氧化物制备环状碳酸酯[J]. 有机化学, 2026, 46(1): 233-240. |
| [3] | 郭聪颖, 高睿, 李倩, 王辉, 张丹维, 周伟, 黎占亭. 高稳定两亲性甘脲纳米组装体构筑及细胞内短链DNA高效递送[J]. 有机化学, 2025, 45(8): 2945-2952. |
| [4] | 马惠敏, 赵宇含, 黄业伟, 孔令斌. 水介质中氰基环氧化物的化学选择性绿色合成研究[J]. 有机化学, 2025, 45(8): 2867-2875. |
| [5] | 张明美, 沙风, 伍新燕. β-二羰基化合物与靛红衍生的α,β-不饱和酮之间的Michael加成/半缩酮化串联反应构建3,4'-吡喃-螺环氧化吲哚类化合物[J]. 有机化学, 2025, 45(4): 1369-1378. |
| [6] | 曹博, 温翔, 陈洁, 王斌. 四齿氮配体的点击合成及其在非血红素铁催化不对称环氧化反应中的应用[J]. 有机化学, 2025, 45(12): 4490-4496. |
| [7] | 王涛, 陶晟, 陈飞, 杜智宏, 薄春博, 李敏, 刘宁. 双功能钼配合物催化二氧化碳和环氧化物制备环状碳酸酯[J]. 有机化学, 2025, 45(12): 4354-4361. |
| [8] | 刘岩, 王晓梅, 何林, 李师伍, 赵志飞. 氮杂环卡宾(NHC)催化[3+2]环加成反应高非对映选择性地构建螺氧吲哚二氢呋喃稠合吡唑啉酮化合物[J]. 有机化学, 2024, 44(4): 1301-1310. |
| [9] | 韩彪, 张瑶瑶, 陈舒晗, 赵梦鸽, 李楠, 李维双, 朱磊. 轴向嫁接型温度响应性手性salen MnIII聚合物的制备及在纯水相烯烃不对称环氧化反应中的应用[J]. 有机化学, 2023, 43(1): 244-253. |
| [10] | 王卫伟, 赵宇, 刘鑫磊, 蒋家珍, 王明安. 3-芳基-7-甲基-7-羟基-2-辛烯-6-内酯类化合物的合成及杀菌活性[J]. 有机化学, 2021, 41(6): 2343-2353. |
| [11] | 董宏波, 王卫伟, 赵宇, 刘鑫磊, 王明安. 3,7-二甲基-7-羟基-2-辛烯-6-内酯类似物的合成及杀菌活性[J]. 有机化学, 2021, 41(4): 1646-1657. |
| [12] | 袁谷城, 刘嘉威, 于士航, 原超楠, 边庆花, 王敏, 钟江春. 美国白蛾性信息素(3Z,6Z,9S,10R)-9,10-环氧-3,6-二十一碳二烯的不对称合成[J]. 有机化学, 2021, 41(11): 4437-4443. |
| [13] | 祁连山, 王涛, 魏永梅, 田恒水. 共还原剂醛类对金属卟啉催化丙烯环氧化的作用研究[J]. 有机化学, 2020, 40(5): 1305-1309. |
| [14] | 刘铨瑶, 石磊, 刘宁. 吡啶桥连双功能有机催化剂催化二氧化碳、环氧化物和芳香胺合成3-芳基-2-噁唑烷酮的研究[J]. 有机化学, 2019, 39(10): 2882-2891. |
| [15] | 孙默然, 代磊, 杨华, 刘宏民, 于德泉. Hydramicromelins A,B和C母核的对映选择性合成[J]. 有机化学, 2018, 38(9): 2443-2449. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||