有机化学 ›› 2026, Vol. 46 ›› Issue (2): 612-623.DOI: 10.6023/cjoc202506008 上一篇 下一篇
研究论文
樊良鑫a, 时梦瑶a, 刘兴源a, 潘振良a, 杨国玉a, 吴璐璐a, 薛晓平a,*( ), 代莉b,*(
), 代莉b,*( ), 徐翠莲a,*(
), 徐翠莲a,*( )
)
收稿日期:2025-06-04
修回日期:2025-08-18
发布日期:2025-10-09
通讯作者:
薛晓平, 代莉, 徐翠莲
基金资助:
Liangxin Fana, Mengyao Shia, Xingyuan Liua, Zhenliang Pana, Guoyu Yanga, Lulu Wua, Xiaoping Xuea,*( ), Li Daib,*(
), Li Daib,*( ), Cuilian Xua,*(
), Cuilian Xua,*( )
)
Received:2025-06-04
Revised:2025-08-18
Published:2025-10-09
Contact:
Xiaoping Xue, Li Dai, Cuilian Xu
Supported by:文章分享
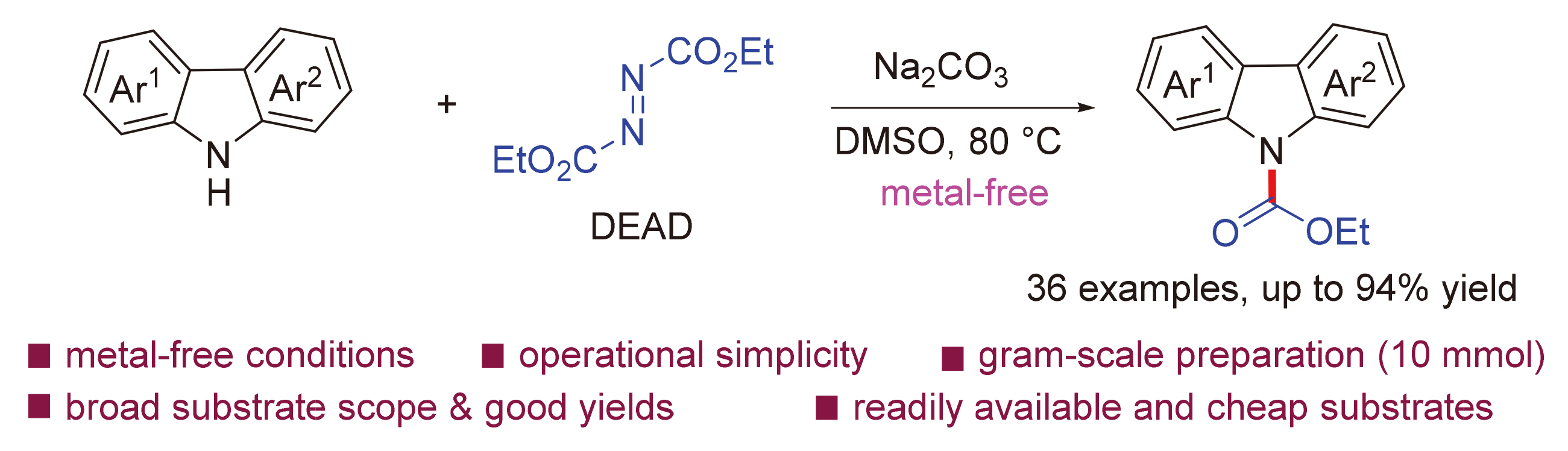
开发了一种在碱促进下以偶氮二甲酸二乙酯(DEAD)作为碳源的二级芳胺N-酰基化合成新策略, 使用咔唑化合物与DEAD为原料, 以15%~94%的收率合成了一系列结构多样的N-酯基咔唑化合物. 该方法无需过渡金属参与, 具有原料廉价易得、反应条件绿色环保、操作简单、底物范围宽泛和反应快速等优点, 为简单高效地合成N-取代咔唑提供了一种便捷的途径. 克级(10 mmol)规模的合成实验与产物衍生化研究, 进一步证实了该方法的实际应用价值. 反应机理研究表明, 该反应经历了咔唑在碱作用下的去质子化、氮负离子对DEAD的亲核进攻以及消除等过程.
樊良鑫, 时梦瑶, 刘兴源, 潘振良, 杨国玉, 吴璐璐, 薛晓平, 代莉, 徐翠莲. 以偶氮二甲酸二乙酯为碳源合成N-酯基咔唑[J]. 有机化学, 2026, 46(2): 612-623.
Liangxin Fan, Mengyao Shi, Xingyuan Liu, Zhenliang Pan, Guoyu Yang, Lulu Wu, Xiaoping Xue, Li Dai, Cuilian Xu. Synthesis of N-Ester Carbazoles Using Diethyl Azodicarboxylate as Carbon Source[J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 612-623.
| Entry | Substrate | Base | Solvent | T/℃ | Yieldb/% |
|---|---|---|---|---|---|
| 1 | 1~6 | Na2CO3 | DMSO | 80 | NR |
| 2 | 7 | Na2CO3 | DMSO | 80 | 44 |
| 3 | 8 | Na2CO3 | DMSO | 80 | NR |
| 4 | 9 | Na2CO3 | DMSO | 80 | 15 |
| 5 | 10 | Na2CO3 | DMSO | 80 | NR |
| 6 | 11 | Na2CO3 | DMSO | 80 | 23 |
| 7 | 12 | Na2CO3 | DMSO | 80 | 32 |
| 8 | 13 | Na2CO3 | DMSO | 80 | 92 |
| 9 | 13 | Li2CO3 | DMSO | 80 | 75 |
| 10 | 13 | NaOAc | DMSO | 80 | 39 |
| 11 | 13 | Na3PO4 | DMSO | 80 | 73 |
| 12 | 13 | NaHCO3 | DMSO | 80 | 54 |
| 13 | 13 | Na2HPO4 | DMSO | 80 | 10 |
| 14 | 13 | KHCO3 | DMSO | 80 | 63 |
| 15 | 13 | K2CO3 | DMSO | 80 | 58 |
| 16 | 13 | K3PO4 | DMSO | 80 | 67 |
| 17 | 13 | tBuONa | DMSO | 80 | 51 |
| 18 | 13 | Et3N | DMSO | 80 | NR |
| 19 | 13 | Na2CO3 | DCE | 80 | 46 |
| 20 | 13 | Na2CO3 | Toluene | 80 | 51 |
| 21 | 13 | Na2CO3 | 1,4-dioxane | 80 | 67 |
| 22 | 13 | Na2CO3 | THF | 80 | Trace |
| 23 | 13 | Na2CO3 | DMF | 80 | 73 |
| 24 | 13 | Na2CO3 | MeCN | 80 | 21 |
| 25 | 13 | Na2CO3 | Sulfolane | 80 | 63 |
| 26 | 13 | Na2CO3 | DMSO | 70 | 65 |
| 27 | 13 | Na2CO3 | DMSO | 90 | 91 |
| 28c | 13 | Na2CO3 | DMSO | 80 | 78 |
| 29d | 13 | Na2CO3 | DMSO | 80 | 70 |
| 30e | 13 | Na2CO3 | DMSO | 80 | Trace |
| Entry | Substrate | Base | Solvent | T/℃ | Yieldb/% |
|---|---|---|---|---|---|
| 1 | 1~6 | Na2CO3 | DMSO | 80 | NR |
| 2 | 7 | Na2CO3 | DMSO | 80 | 44 |
| 3 | 8 | Na2CO3 | DMSO | 80 | NR |
| 4 | 9 | Na2CO3 | DMSO | 80 | 15 |
| 5 | 10 | Na2CO3 | DMSO | 80 | NR |
| 6 | 11 | Na2CO3 | DMSO | 80 | 23 |
| 7 | 12 | Na2CO3 | DMSO | 80 | 32 |
| 8 | 13 | Na2CO3 | DMSO | 80 | 92 |
| 9 | 13 | Li2CO3 | DMSO | 80 | 75 |
| 10 | 13 | NaOAc | DMSO | 80 | 39 |
| 11 | 13 | Na3PO4 | DMSO | 80 | 73 |
| 12 | 13 | NaHCO3 | DMSO | 80 | 54 |
| 13 | 13 | Na2HPO4 | DMSO | 80 | 10 |
| 14 | 13 | KHCO3 | DMSO | 80 | 63 |
| 15 | 13 | K2CO3 | DMSO | 80 | 58 |
| 16 | 13 | K3PO4 | DMSO | 80 | 67 |
| 17 | 13 | tBuONa | DMSO | 80 | 51 |
| 18 | 13 | Et3N | DMSO | 80 | NR |
| 19 | 13 | Na2CO3 | DCE | 80 | 46 |
| 20 | 13 | Na2CO3 | Toluene | 80 | 51 |
| 21 | 13 | Na2CO3 | 1,4-dioxane | 80 | 67 |
| 22 | 13 | Na2CO3 | THF | 80 | Trace |
| 23 | 13 | Na2CO3 | DMF | 80 | 73 |
| 24 | 13 | Na2CO3 | MeCN | 80 | 21 |
| 25 | 13 | Na2CO3 | Sulfolane | 80 | 63 |
| 26 | 13 | Na2CO3 | DMSO | 70 | 65 |
| 27 | 13 | Na2CO3 | DMSO | 90 | 91 |
| 28c | 13 | Na2CO3 | DMSO | 80 | 78 |
| 29d | 13 | Na2CO3 | DMSO | 80 | 70 |
| 30e | 13 | Na2CO3 | DMSO | 80 | Trace |
| [1] |
(a)
doi: 10.1021/cr0306790 |
|
(b)
doi: 10.1021/ar900059r |
|
|
(c)
|
|
|
(d)
doi: 10.1021/acscatal.5c01292 |
|
| [2] |
(a)
doi: 10.1021/cr200447s pmid: 22480243 |
|
(b)
pmid: 22480243 |
|
|
(c)
pmid: 22480243 |
|
|
(d)
doi: 10.1021/cr020059j pmid: 22480243 |
|
| [3] |
doi: 10.1016/j.tet.2012.05.127 |
| [4] |
doi: S0923-7534(19)31884-8 pmid: 30215676 |
| [5] |
doi: S0223-5234(17)30345-8 pmid: 28477443 |
| [6] |
doi: 10.3390/molecules16087155 |
| [7] |
pmid: 3377684 |
| [8] |
(a)
doi: 10.1016/j.jacc.2018.02.049 |
|
(b)
doi: 10.1038/s41467-022-34765-w |
|
| [9] |
doi: 10.1021/acsmedchemlett.0c00420 pmid: 33335665 |
| [10] |
pmid: 16417354 |
| [11] |
(a)
doi: 10.1002/anie.v56.47 |
|
(b)
doi: 10.1039/C9QO00017H |
|
|
(c)
doi: 10.1002/ajoc.v10.5 |
|
| [12] |
doi: 10.1021/ja710555g |
| [13] |
doi: 10.1039/c3ra40972d |
| [14] |
doi: 10.1039/c3qo00069a |
| [15] |
doi: 10.1039/C5OB01264C |
| [16] |
doi: 10.1039/C7QO00945C |
| [17] |
doi: 10.1039/C6RA22108D |
| [18] |
doi: 10.1039/C6OB02627C |
| [19] |
doi: 10.1039/D3OB01437A |
| [20] |
doi: 10.1021/ol048564l |
| [21] |
|
| [22] |
doi: 10.3762/bjoc.20.2 pmid: 38213840 |
| [23] |
doi: 10.1021/ja102758v pmid: 20614936 |
| [24] |
doi: 10.1016/j.tet.2009.08.075 |
| [25] |
doi: 10.1039/c2cc33695b |
| [26] |
doi: 10.1021/acscatal.7b02682 |
| [27] |
doi: 10.1002/anie.v51.33 |
| [28] |
pmid: 15651780 |
| [29] |
doi: 10.1021/acscentsci.7b00195 |
| [30] |
doi: 10.1016/j.tet.2006.10.071 |
| [31] |
|
| [1] | 莫百川, 李婷婷, 王芳, 李晓杰, 巴达日夫. 咔唑醌天然产物的合成研究进展[J]. 有机化学, 2025, 45(8): 2746-2766. |
| [2] | 张文博, 苏思铭, 杨鹏. 新型杯[3]咔唑衍生物的合成及其对羟喜树碱识别与检测研究[J]. 有机化学, 2025, 45(4): 1395-1401. |
| [3] | 任志明, 谢立娟, 晋静, 吴玉洁, 刘迪, 李久艳. 基于吲哚并[3,2,1-jk]咔唑的多重共振型发光分子研究进展[J]. 有机化学, 2025, 45(11): 4108-4127. |
| [4] | 姜晓琳, 王超洋, 武利园, 李跃辉. 含咔唑结构的小分子及聚合物催化二氧化碳转化研究进展[J]. 有机化学, 2024, 44(5): 1423-1444. |
| [5] | 孙静, 樊志杰, 杜纪宽, 李硕, 苗艳勤, 赵波, 董海亮, 王华. 一种Y型噻吨酮-咔唑分子的设计、合成及其蓝光和黄光有机发光二极管应用[J]. 有机化学, 2024, 44(4): 1210-1217. |
| [6] | 韩阳, 姜为超, 张靖, 彭进松, 陈春霞. 可见光促进钯催化C—H键胺化反应合成咔唑醌衍生物的研究[J]. 有机化学, 2022, 42(1): 266-276. |
| [7] | 李英俊, 林乐弟, 刘季红, 高立信, 盛丽, 靳焜, 刘雪洁, 杨鸿境, 李佳. 新型含咔唑环和芳环/芳稠杂环的N-酰腙衍生物的合成及蛋白酪氨酸磷酸酶1B (PTP1B)抑制活性评价[J]. 有机化学, 2021, 41(9): 3593-3607. |
| [8] | 李英俊, 林乐弟, 靳焜, 高立信, 盛丽, 刘季红, 李佳. 新型含咔唑环芳氨基乙酰腙衍生物的合成及其蛋白酪氨酸磷酸酶1B (PTP1B)抑制活性评价[J]. 有机化学, 2021, 41(8): 3157-3170. |
| [9] | 赵雨, 刘阳, 王鑫鑫, 洪莹莹, 满英秀, 王进军, 李家柱. 3,6-二烃基咔唑-1-甲醛和3,6-二烃基咔唑-1,8-二甲醛的高效合成[J]. 有机化学, 2021, 41(6): 2435-2444. |
| [10] | 杜玉英, 肖业元, 田福利, 韩利民, 顾彦龙, 竺宁. 吲哚多样化环化合成咔唑衍生物研究进展[J]. 有机化学, 2021, 41(2): 521-528. |
| [11] | 李雪, 宋子锐, 陈鑫, 蔡轶超, 刘雅婕, 陈春霞, 彭进松. 钯催化双芳基化反应合成咔唑醌衍生物的研究[J]. 有机化学, 2020, 40(4): 950-958. |
| [12] | 宋文越, 饶小峰, 卜庆青, 刘宁. 咔唑桥连NCN齿形钯配合物催化的唑类C-H键直接芳基化反应[J]. 有机化学, 2020, 40(2): 489-500. |
| [13] | 李英俊, 刘雪洁, 刘季红, 高立信, 靳焜, 盛丽, 杨鸿境, 林乐弟, 李佳. 基于咔唑的新型碳酰腙衍生物的合成及蛋白酪氨酸磷酸酶1B(PTP1B)抑制活性评价[J]. 有机化学, 2020, 40(2): 478-488. |
| [14] | 李英俊, 杨凯栋, 靳焜, 高立信, 盛丽, 刘雪洁, 杨鸿境, 林乐弟, 李佳. 基于咔唑的单-/双-硫代碳酰腙衍生物的合成及Cdc25B/PTP1B抑制活性评价[J]. 有机化学, 2020, 40(1): 162-174. |
| [15] | 李英俊, 赵月, 靳焜, 高立信, 盛丽, 刘雪洁, 杨鸿境, 林乐弟, 李佳. 含咔唑/苯并咪唑环的2,5-二取代-1,3,4-噻二唑酰胺衍生物的合成及对PTP1B/TCPTP抑制活性的评价[J]. 有机化学, 2019, 39(9): 2599-2608. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||