有机化学 ›› 2026, Vol. 46 ›› Issue (2): 653-663.DOI: 10.6023/cjoc202507038 上一篇 下一篇
研究论文
张云一a, 严含冰b, 朱先进c, 石永佳a, 李俊鑫a,*( ), 杨道山a,*(
), 杨道山a,*( ), 李旭锋c,*(
), 李旭锋c,*( )
)
收稿日期:2025-07-29
修回日期:2025-09-01
发布日期:2025-10-09
通讯作者:
李俊鑫, 杨道山, 李旭锋
基金资助:
Yunyi Zhanga, Hanbing Yanb, Xianjin Zhuc, Yongjia Shia, Junxin Lia,*( ), Daoshan Yanga,*(
), Daoshan Yanga,*( ), Xufeng Lic,*(
), Xufeng Lic,*( )
)
Received:2025-07-29
Revised:2025-09-01
Published:2025-10-09
Contact:
Junxin Li, Daoshan Yang, Xufeng Li
Supported by:文章分享
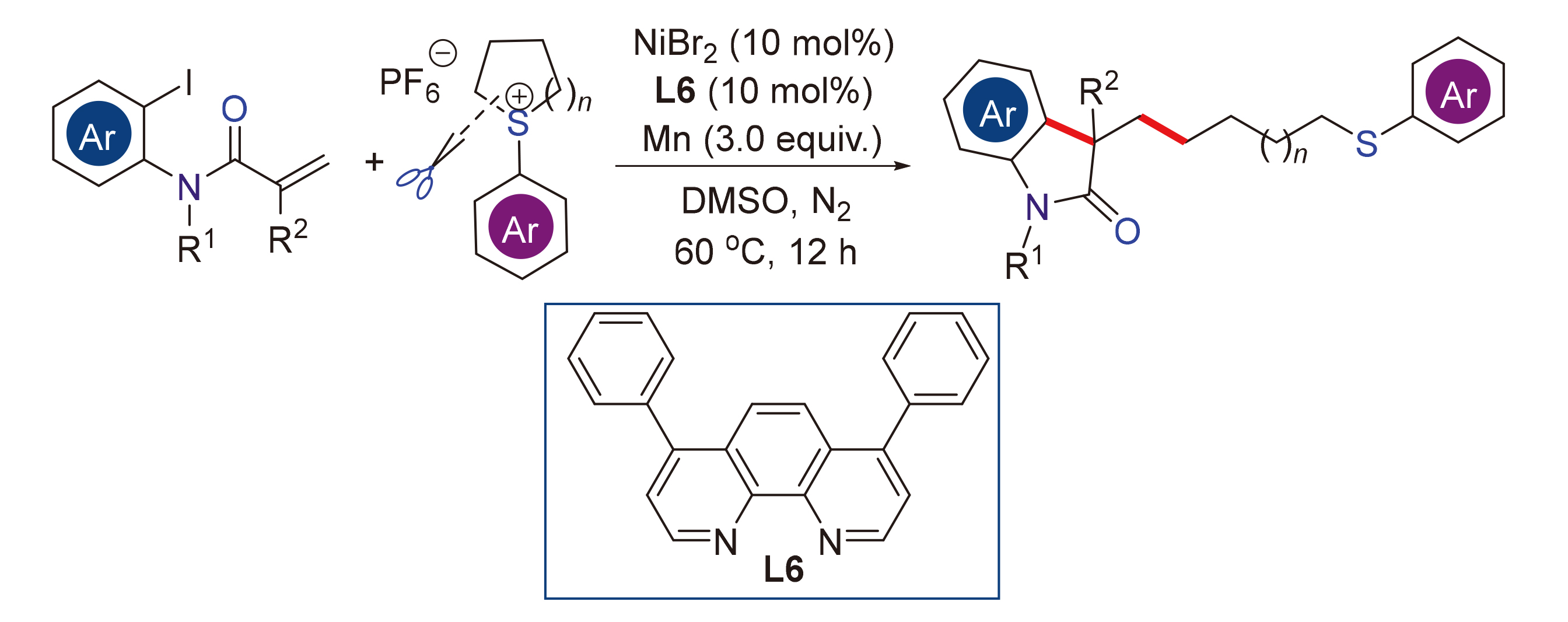
镍催化的烯烃与烷基亲电试剂之间的还原交叉偶联反应是构建杂环化合物的重要方法之一. 然而, 通过还原交叉偶联反应实现烯烃芳烷基化的烷基化试剂仍相当有限. 开发以稳定且易得的起始原料, 通过还原偶联策略合成杂环化合物的更为普适的方法仍具有挑战性. 发展了一种新的镍催化还原串联芳烷基化反应: 以环硫鎓盐作为C(sp³)亲电试剂, 通过镍催化的还原偶联策略实现烯烃的芳烷基化, 从而合成了一系列含硫醚片段的氧化吲哚类化合物. 该串联开环/环化/还原偶联策略可在温和条件下高效构建多种带有全碳季碳中心的氧化吲哚, 并具有优异的官能团兼容性. 值得注意的是, 许多药物衍生物均可利用该策略直接实现官能化.
张云一, 严含冰, 朱先进, 石永佳, 李俊鑫, 杨道山, 李旭锋. 镍催化烯烃与环硫鎓盐的还原串联芳烷基化反应[J]. 有机化学, 2026, 46(2): 653-663.
Yunyi Zhang, Hanbing Yan, Xianjin Zhu, Yongjia Shi, Junxin Li, Daoshan Yang, Xufeng Li. Nickel-Catalyzed Reductive Cascade Arylalkylation of Alkenes with Cyclosulfonium Salts[J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 653-663.
| Entry | Variation from the standard reaction conditions | Yieldb/% |
|---|---|---|
| 1 | None | 76 |
| 2 | Ligands other than L6 | Trace~69 |
| 3 | Catalysts other than NiBr2 | 21~65 |
| 4 | Solvents other than DMSO | Trace~73 |
| 5 | Zn instead of Mn | 17 |
| 6 | Room temperature instead of 60 ℃ | N.R. |
| Entry | Variation from the standard reaction conditions | Yieldb/% |
|---|---|---|
| 1 | None | 76 |
| 2 | Ligands other than L6 | Trace~69 |
| 3 | Catalysts other than NiBr2 | 21~65 |
| 4 | Solvents other than DMSO | Trace~73 |
| 5 | Zn instead of Mn | 17 |
| 6 | Room temperature instead of 60 ℃ | N.R. |
| [1] |
(c)
doi: 10.1016/j.cclet.2022.108036 |
|
(d)
doi: 10.6023/A24100329 |
|
|
(辛翠, 蒋俊, 邓紫微, 欧丽娟, 何卫民, 化学学报, 2024, 82, 1109.)
doi: 10.6023/A24100329 |
|
|
(e)
doi: 10.6023/cjoc202407017 |
|
|
(永波, 舒洪波, 黄华文, 有机化学, 2025, 45, 2086.)
|
|
|
(f)
doi: 10.6023/cjoc202206053 |
|
|
(李芳洁, 卢斌, 刘阳, 王晓明, 有机化学, 2022, 42, 3390.)
doi: 10.6023/cjoc202206053 |
|
|
(g)
doi: 10.6023/cjoc202003029 |
|
|
(郭欣, 郭亚军, 孔德志, 卢会杰, 华远照, 王敏灿, 有机化学, 2020, 40, 1999.)
doi: 10.6023/cjoc202003029 |
|
| [2] |
(a)
doi: 10.1021/acs.accounts.8b00097 |
|
(b)
doi: 10.1016/j.cclet.2021.01.021 |
|
|
(c)
|
|
| [1] |
(a)
doi: 10.6023/cjoc202301023 |
|
(岁丹丹, 岑南楠, 龚若蕖, 陈阳, 陈文博, 有机化学, 2023, 43, 3239.)
doi: 10.6023/cjoc202301023 |
|
|
(b)
|
|
| [2] |
(d)
doi: 10.1016/j.cclet.2024.110038 |
| [3] |
(a)
doi: 10.1021/acs.accounts.5b00057 |
|
(b)
doi: 10.1002/anie.v58.6 |
|
|
(c)
doi: 10.1016/j.cclet.2022.01.077 |
|
|
(d)
doi: 10.6023/cjoc202502007 |
|
|
(谢沈彤, 李文静, 刘钰, 陆熹, 师仁义, 有机化学, 2025, 45, 2121.)
doi: 10.6023/cjoc202502007 |
|
|
(e)
doi: 10.1039/D1GC00562F |
|
|
(f)
doi: 10.1039/D3GC02575F |
|
|
(g)
doi: 10.6023/cjoc202404019 |
|
|
(张鑫伟, 林水侦, 黄晓雷, 有机化学, 2024, 44, 3456 )
doi: 10.6023/cjoc202404019 |
|
| [4] |
(a)
doi: 10.1021/acscatal.0c01842 pmid: 32905517 |
|
(b)
doi: 10.1021/jacs.8b08190 pmid: 32905517 |
|
| [5] |
doi: 10.1021/acs.orglett.9b02870 |
| [6] |
(a)
doi: 10.1002/anie.v58.20 |
|
(b)
doi: 10.1021/jacs.4c03745 |
|
|
(c)
doi: 10.1021/acs.orglett.1c01871 |
|
| [7] |
(a)
doi: 10.1021/acs.orglett.2c00599 |
|
(b)
doi: 10.1021/acs.accounts.2c00771 |
|
| [8] |
(a)
doi: 10.1055/a-1677-5971 pmid: 32742188 |
|
(b)
doi: 10.1002/ejic.202000249 pmid: 32742188 |
|
|
(c)
doi: 10.1016/j.cclet.2020.06.003 pmid: 32742188 |
|
| [9] |
(a)
doi: 10.1038/s41586-019-0982-0 |
|
(b)
doi: 10.1021/acscatal.3c03096 |
|
|
(c)
doi: 10.1039/D2SC04507A |
|
|
(d)
doi: 10.1039/D2GC04843D |
|
|
(e)
|
|
| [10] |
doi: 10.1021/jacs.1c03459 pmid: 34028272 |
| [11] |
(a)
doi: 10.1039/C9CS00837C |
|
(b)
doi: 10.1016/j.cclet.2021.09.068 |
|
|
(c)
doi: 10.1021/acs.joc.5c01659 |
|
|
(d)
doi: 10.1016/j.cclet.2021.06.068 |
|
|
(e)
doi: 10.1021/acssuschemeng.1c07636 |
|
|
(f)
doi: 10.1021/acssuschemeng.5c03350 |
|
| [12] |
(a)
doi: 10.1021/acs.orglett.1c01050 |
|
(b)
doi: 10.1039/D1QO01570B |
|
|
(c)
doi: 10.1039/D1GC04184C |
|
|
(d)
doi: 10.1016/j.cclet.2023.108403 |
|
|
(e)
doi: 10.1021/acs.orglett.4c00017 |
|
|
(f)
doi: 10.6023/cjoc202209004 |
|
|
(徐浩, 张杰, 左峻泽, 王丰晓, 吕健, 混旭, 杨道山, 有机化学, 2022, 42, 4037.)
doi: 10.6023/cjoc202209004 |
|
| [13] |
doi: 10.1002/cjoc.v42.14 |
| [1] | 孙雨, 朱延涛, 张东, 倪春节. 镍催化炔基与三氟乙酰亚胺氯和水的环化反应: 高效合成2-(三氟甲基)吲哚衍生物[J]. 有机化学, 2026, 46(3): 1017-1026. |
| [2] | 范润东, 陈冰然, 黎富奇, 陈锦秀, 严琼姣, 汪伟, 周慧. 无光敏剂光诱导α-三氟甲基烯烃的氢二氟烷基化反应[J]. 有机化学, 2026, 46(2): 641-652. |
| [3] | 高山, 解鑫, 刘元红. 镍/可见光协同催化的溴代芳烃与醇的C—O键偶联反应[J]. 有机化学, 2026, 46(1): 266-278. |
| [4] | 梁媛, 桂超, 王雯雯, 褚雪强, 徐浩, 沈志良. 镍催化的芳基全氟丁基磺酸酯通过碳-氧键官能化的硼化反应高效合成芳基硼酸酯[J]. 有机化学, 2026, 46(1): 207-214. |
| [5] | 张元贺, 沈运杰, 谈东兴, 韩福社. Cryptoquinonemethides合成中螺五环非对映异构体环丙烷化反应活性及选择性研究[J]. 有机化学, 2026, 46(1): 96-105. |
| [6] | 赵倩倩, 魏培垚, 刘孙典, 张博鑫, 梁承远. 钯催化串联反应在含季碳中心的复杂天然产物全合成中的应用[J]. 有机化学, 2025, 45(9): 3289-3300. |
| [7] | 刘丽强, 南军, 段兴宇, 王昊龙, 蔡巷, 王梓诚, 张恩泽, 孙彦民. 三齿P配体钌络合物催化脂肪醇胺N-烷基化反应研究[J]. 有机化学, 2025, 45(8): 2983-2988. |
| [8] | 马海宸, 周钧岍, 王嘉利, 王优, 朱少林. 镍催化的迁移偶联反应研究进展[J]. 有机化学, 2025, 45(6): 1871-1904. |
| [9] | 田江艳, 李驰, 陈剑飞, 吕辉. 镍和钴催化卤代烃与醛酮/亚胺的还原偶联反应研究进展[J]. 有机化学, 2025, 45(6): 1946-1960. |
| [10] | 谢沈彤, 李文静, 刘钰, 陆熹, 师仁义. 镍催化多氟芳烃与烷基卤化物的还原烷基化反应[J]. 有机化学, 2025, 45(6): 2121-2127. |
| [11] | 周强, 杨宝臻, 郝贵林, 罗木鹏, 曹石, 赵蓓, 袁华, 王守国. 铑(III)催化的非活化烯烃与α-重氮羰基化合物的对映选择性烯丙位C—H键烷基化反应[J]. 有机化学, 2025, 45(6): 2109-2120. |
| [12] | 高根伟, 李震, 李炎, 陆熹. 光/镍协同催化C(sp2)—C(sp3)键构建研究进展[J]. 有机化学, 2025, 45(6): 1905-1919. |
| [13] | 牛丽菁, 吴成娟, 梁文静, 耿琰, 董育斌. 光催化串联反应构建共价有机框架[J]. 有机化学, 2025, 45(5): 1707-1715. |
| [14] | 谭芳芳, 史孟欣, 张文敏, 李洋. 光催化生物质相关转化[J]. 有机化学, 2025, 45(5): 1523-1547. |
| [15] | 张明美, 沙风, 伍新燕. β-二羰基化合物与靛红衍生的α,β-不饱和酮之间的Michael加成/半缩酮化串联反应构建3,4'-吡喃-螺环氧化吲哚类化合物[J]. 有机化学, 2025, 45(4): 1369-1378. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||