化学学报 ›› 2019, Vol. 77 ›› Issue (9): 814-831.DOI: 10.6023/A19050170 上一篇 下一篇
所属专题: 有机自由基化学
综述
投稿日期:2019-05-12
发布日期:2019-06-12
通讯作者:
吴劼
E-mail:Jie_wu@fudan.edu.cn
作者简介:叶盛青, 2008年在复旦大学化学系获学士学位; 2013年在复旦大学有机化学专业获理学博士学位(导师: 吴劼教授); 2013年至2015年在美国The Scripps Research Institute从事博士后工作(导师: 余金权教授); 2015年至2019年在苏州诺华制药科技有限公司担任工艺经理; 2019年至今在台州学院医药化工与材料工程学院任副教授. 主要从事类天然产物小分子骨架的有机方法学研究.|吴劼, 1991~1995年就读于江西师范大学化学系, 1995~2000年在中国科学院上海有机化学研究所进行研究生学习(2000年获得博士学位), 2000至2004 年先后在美国哈佛大学(博士后)、洛克菲勒大学艾伦·戴蒙德艾滋病研究中心(访问科学家)及VivoQuest, Inc.(研究员)从事有机合成、药物化学及相关研究工作, 2004年9月回国加入复旦大学化学系(副教授), 2006年4月晋升为教授. 2019年8月加入台州学院医药化工与材料工程学院. 目前主要研究领域: 构建类天然小分子化合物用于抗肿瘤及免疫类疾病、炎症疾病和神经退行性疾病新药研究.
基金资助:Received:2019-05-12
Published:2019-06-12
Contact:
Wu, Jie
E-mail:Jie_wu@fudan.edu.cn
Supported by:文章分享
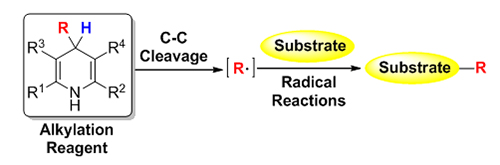
汉斯酯(Hantzsch Esters)在1881年被合成以来一直被用作还原剂参与加氢还原反应, 近年来科学家发现4-取代的汉斯酯可以发生碳碳键断裂而发生烷基迁移反应. 随后大量以4-取代的汉斯酯类化合物作为烷基化试剂的反应被报道出来, 科学家发现这类烷基化反应的历程主要是烷基自由基的迁移历程. 随着近年自由基化学的快速发展, 为这类新型烷基化试剂的应用发展提供了有力基础. 本文按照该烷基化试剂参与的反应类型分类, 进行简单介绍.
叶盛青, 吴劼. 4-取代的汉斯酯(Hantzsch Esters)作为烷基化试剂参与的有机反应[J]. 化学学报, 2019, 77(9): 814-831.
Ye, Shengqing, Wu, Jie. 4-Substituted Hantzsch Esters as Alkylation Reagents in Organic Synthesis[J]. Acta Chimica Sinica, 2019, 77(9): 814-831.
| [1] |
Hantzsch, A . Ber. Dtsch. Chem. Ges. 1881, 14, 1637.
doi: 10.1002/(ISSN)1099-0682 |
| [2] |
(a) Janis, R. A.; Triggle, D. J. J. Med. Chem. 1983, 25, 775.
doi: 10.1021/jm00159a007 |
|
(b) Bocker, R. H.; Guengerich, F. P. J. Med. Chem. 1986, 29, 1596.
doi: 10.1021/jm00159a007 |
|
|
(c) Xie, W.; Wu, Y.; Zhang, J.; Mei, Q.; Zhang, Y.; Zhu, N.; Liu, R.; Zhang, H. Eur. J. Med. Chem. 2018, 145, 35.
doi: 10.1021/jm00159a007 |
|
|
(d) Xie, W.; Zhang, H.; He, J.; Zhang, J.; Yu, Q.; Luo, C.; Li, S. Bioorg. Med. Chem. Lett. 2017, 27, 530.
doi: 10.1021/jm00159a007 |
|
| [3] |
Bergstrom, F. W. Chem. Rev. 1944, 35, 77.
doi: 10.1021/cr60111a001 |
| [4] |
Mauzerall, D.; Westheimer, F. H. J. Am. Chem. Soc. 1955, 77, 2261.
doi: 10.1021/ja01613a070 |
| [5] |
For selected reviews see: (a) Ouellet, S. G.; Walji, A. M.; Macmillan, D. W. C. Acc. Chem. Res. 2007, 40, 1327.
doi: 10.1021/ar7001864 |
|
(b) de Vries, J. G.; Mrsic, N. Catal. Sci. Technol. 2011, 1, 727.
doi: 10.1021/ar7001864 |
|
|
(c) Zheng, C.; You, S.-L. Chem. Soc. Rev. 2012, 41, 2498.
doi: 10.1021/ar7001864 |
|
|
(d) Huang, W.; Cheng, X.. Synlett 2017, 28, 148.
doi: 10.1021/ar7001864 |
|
|
(e) Li, X.; Meng, Y.; Yi, P.; Stepień, M.; Chmielewski, P. J. Angew. Chem., Int. Ed. 2017, 56, 10810.
doi: 10.1021/ar7001864 |
|
| [6] | Loev, B.; Snader, K. M . J. Org. Chem., 1965, 30 1914. |
| [7] |
Wei, Z.; Li, J.; Wang, Z.; Li, P.; Wang, Y . Chin. J. Org. Chem. 2017, 37, 1835 (in Chinese).
doi: 10.6023/cjoc201612055 |
|
( 魏振中, 李江飞, 王泽云, 李品华, 王永秋 , 有机化学, 2017, 37, 1835.)
doi: 10.6023/cjoc201612055 |
|
| [8] |
For selected examples see:(a) Zou, Y.-Q.; Hörmann, F. M.; Bach, T. Chem. Soc. Rev. 2018, 47, 278.
doi: 10.1039/C7CS00509A |
|
(b) Wang, F.; Chen, P.; Liu, G. Acc. Chem. Res. 2018, 51, 2036.
doi: 10.1039/C7CS00509A |
|
|
(c) Wang, K.; Kong, W. Chin. J. Chem. 2018, 36, 247.
doi: 10.1039/C7CS00509A |
|
|
(d) Qiu, S.; Wang, C.; Xie, S.; Huang, X.; Chen, L.; Zhao, Y.; Zeng, Z. Chem. Commun. 2018, 54, 11383.
doi: 10.1039/C7CS00509A |
|
|
(e) Xie, L.-Y.; Peng, S.; Liu, F.; Chen, G.-R.; Xia, W.; Yu, X.; Li, W.-F.; Cao, Z.; He, W.-M. Org. Chem. Front. 2018, 5, 2604.
doi: 10.1039/C7CS00509A |
|
|
(f) Lu, L.-H.; Zhou, S.-J.; He, W.-B.; Xia, W.; Chen, P.; Yu, X.; Xu, X.; He, W.-M. Org. Biomol. Chem. 2018, 16, 9064.
doi: 10.1039/C7CS00509A |
|
|
(g) Zheng, Y.; Liu, M.; Qiu, G.; Xie, W.; Wu, J. Tetrahedron 2019, 75, 1663.
doi: 10.1039/C7CS00509A |
|
|
(h) Liu, K.-J.; Jiang, S.; Lu, L.-H.; Tang, L.-L.; Tang, S.-S.; Tang, H.-S.; Tang, Z.; He, W.-M.; Xu, X. Green Chem. 2018, 20, 3038.
doi: 10.1039/C7CS00509A |
|
|
(i) Xie, L.-Y.; Peng, S.; Liu, F.; Yi, J.-Y.; Wang, M.; Tang, Z.; Xu, X.; He, W.-M. Adv. Synth. Catal. 2018, 360, 4259.
doi: 10.1039/C7CS00509A |
|
|
(j) Xie, L.-Y.; Peng, S.; Liu, F.; Chen, G.-R.; Xia, W.; Yu, X.; Li, W.-F.; Cao, Z.; He, W.-M. Org. Chem. Front. 2018, 5, 2604.
doi: 10.1039/C7CS00509A |
|
|
(k) Guo, T.; Wei, X.-N.; Liu, Y.; Zhang, P.-K.; Zhao, Y.-H. Org. Chem. Front. 2019, 6, 1414. 1414.
doi: 10.1039/C7CS00509A |
|
| [9] |
For selected examples see: (a) Yoon, T. P.; Ischay, M. A.; Du, J . Nat. Chem. 2010, 2, 527.
doi: 10.1038/nchem.687 |
|
(b) Teplý, F. Collect. Czech. Chem. Commun. 2011, 76, 859.
doi: 10.1038/nchem.687 |
|
|
(c) Narayanam, J. M.; Stephenson, C. R. Chem. Soc. Rev. 2011, 40, 102.
doi: 10.1038/nchem.687 |
|
|
(d) Xuan, J.; Xiao, W. J. Angew. Chem., Int. Ed. 2012, 51, 6828.
doi: 10.1038/nchem.687 |
|
|
(e) Shi, L.; Xia, W. Chem. Soc. Rev. 2012, 41, 7687.
doi: 10.1038/nchem.687 |
|
|
(f) Prier, C. K.; Rankic, D. A.; MacMillan, D. W. Chem. Rev. 2013, 113, 5322.
doi: 10.1038/nchem.687 |
|
|
(g) Xi, Y.; Yi, H.; Lei, A. Org. Biomol. Chem. 2013, 11, 2387.
doi: 10.1038/nchem.687 |
|
|
(h) Xuan, J.; Lu, L. Q.; Chen, J. R.; Xiao, W. J. Eur. J. Org. Chem. 2013, 6755.
doi: 10.1038/nchem.687 |
|
|
(i) Hari, D. P.; König, B. Angew. Chem., Int. Ed. 2013, 52, 4734.
doi: 10.1038/nchem.687 |
|
|
(j) Hopkinson, M. N.; Sahoo, B.; Li, J. L.; Glorius, F. Chem. Eur. J. 2014, 20, 3874.
doi: 10.1038/nchem.687 |
|
|
(k) Peñ-López, M.; Rosas-Hernández, A.; Beller, M. Angew. Chem., Int. Ed. 2015, 54, 5006.
doi: 10.1038/nchem.687 |
|
|
(l) Shaw, M. H.; Twilton, J.; MacMillan, D. W. C. J. Org. Chem. 2016, 81, 6898.
doi: 10.1038/nchem.687 |
|
|
(m) Wang, D.; Zhang, L.; Luo, S. Acta Chim. Sinica 2017, 75, 22 (in Chinese)
doi: 10.1038/nchem.687 |
|
|
(王德红, 张龙, 罗三中, 化学学报, 2017, 75, 22.)
doi: 10.1038/nchem.687 |
|
| [10] |
Li, G.; Chen, R.; Wu, L.; Fu, Q.; Zhang, X.; Tang, Z. Angew. Chem., Int. Ed. 2013, 52, 8432.
doi: 10.1002/anie.v52.32 |
| [11] |
Zhang, H.-H.; Yu, S . J. Org. Chem. 2017, 82, 9995.
doi: 10.1021/acs.joc.7b01425 |
| [12] |
Gu, F.; Huang, W.; Liu, X.; Chen, W.; Cheng, X. Adv. Synth. Catal. 2017, 360, 925.
doi: 10.1002/adsc.v360.5 |
| [13] |
Wu, Q.-Y.; Min, Q.-Q.; Ao, G.-Z.; Liu, F. Org. Biomol. Chem. 2018, 16, 6391.
doi: 10.1039/C8OB01641K |
| [14] | Mcdonald, B. R.; Scheidt, K. A . Org. Lett. 2018, 20, 6881. |
| [15] |
Van Leeuwen, T.; Buzzetti, L.; Perego, L. A.; Melchiorre, P. Angew. Chem., Int. Ed. 2019, 58, 4953.
doi: 10.1002/anie.201814497 |
| [16] |
Milligan, J. A.; Phelan, J. P.; Polites, V. C.; Kelly, C. B.; Molander, G. A . Org. Lett. 2018, 20, 6840.
doi: 10.1021/acs.orglett.8b02968 |
| [17] |
Chen, H.; Anand, D.; Zhou, L . Asian J. Org. Chem. 2019, 8, 661.
doi: 10.1002/ajoc.v8.5 |
| [18] |
Chen, W.; Liu, Z.; Tian, J.; Li, J.; Ma, J.; Cheng, X.; Li, G. J. Am. Chem. Soc. 2016, 138, 12312.
doi: 10.1021/jacs.6b06379 |
| [19] |
Nakajima, K.; Nojima, S.; Nishibayashi, Y. Angew. Chem., Int. Ed. 2016, 55, 14106.
doi: 10.1002/anie.v55.45 |
| [20] |
Gutiérrez-Bonet, Á.; Tellis, J. C.; Matsui, J. K.; Vara, B. A.; Molander, G. A. ACS Catal. 2016, 6, 8004.
doi: 10.1021/acscatal.6b02786 |
| [21] |
Dumoulin, A.; Matsui, J. K.; Gutiérrez-Bonet, Á.; Molander, G. A. Angew. Chem., Int. Ed. 2018, 57, 6614.
doi: 10.1002/anie.201802282 |
| [22] |
Badir, S. O.; Dumoulin, A.; Matsui, J. K.; Molander, G. A. Angew. Chem., Int. Ed. 2018, 57, 6610.
doi: 10.1002/anie.201800701 |
| [23] |
Nakajima, K.; Guo, X.; Nishibayashi, Y . Chem. Asian J. 2018, 13, 3653.
doi: 10.1002/asia.v13.23 |
| [24] |
Buzzetti, L.; Prieto, A.; Roy, S. R.; Melchiorre, P. Angew. Chem., Int. Ed. 2017, 56, 15039.
doi: 10.1002/anie.201709571 |
| [25] |
For selected examples see: (a) Wu, C.; Lu, L.-H.; Peng, A.-Z.; Jia, G.-K.; Peng, C.; Cao, Z.; Tang, Z.; He, W.-M.; Xu, X . Green Chem. 2018, 20, 3683.
doi: 10.1039/C8GC00491A |
|
(b) Lu, L.-H.; Zhou, S.-J.; Sun, M.; Chen, J.-L.; Xia, W.; Yu, X.; Xu, X.; He, W.-M. ACS Sustainable Chem. Eng. 2019, 7, 1574.
doi: 10.1039/C8GC00491A |
|
|
(c) Wu, C.; Xiao, H.-J.; Wang, S.-W.; Tang, M.-S.; Tang, Z.-L.; Xia, W.; Li, W.-F.; Zhong, C.; He, W.-M. ACS Sustainable Chem. Eng.. 2019, 7, 2169.
doi: 10.1039/C8GC00491A |
|
|
(d) Wu, C.; Wang, Z.; Hu, Z.; Zeng, F.; Zhang, X.-Y.; Cao, Z.; Tang, Z.; He, W.-M.; Xu, X. Org. Biomol. Chem.. 2018, 16, 3177.
doi: 10.1039/C8GC00491A |
|
|
(e) Wang, Z.; Yang, L.; Liu, H.-L.; Tan, Y.-Z.; Bao, W.-H.; Wang, M.; Tang, Z.; He, W.-M. Chin. J. Org. Chem. 2018, 38, 2639 (in Chinese)
doi: 10.1039/C8GC00491A |
|
|
(王峥, 杨柳, 刘慧兰, 谭英芝, 包文虎, 汪明, 唐子龙, 何卫民, 有机化学, 2018, 38, 2639.)
doi: 10.1039/C8GC00491A |
|
| [26] |
Liu, X.; Liu, R.; Dai, J.; Cheng, X.; Li, G . Org. Lett. 2018, 20, 6906.
doi: 10.1021/acs.orglett.8b03050 |
| [27] |
Song, Z.-Y.; Zhang, C.-L.; Ye, S. Org. Biomol. Chem. 2019, 17, 181.
doi: 10.1039/C8OB02912A |
| [28] |
Li, G.; Wu, L.; Lv, G.; Liu, H.; Fu, Q.; Zhang, X.; Tang, Z . Chem. Commun. 2014, 50, 6246.
doi: 10.1039/C4CC01119H |
| [29] |
Nakajima, K.; Nojima, S.; Sakata, K.; Nishibayashi, Y . ChemCatChem 2016, 8, 1028.
doi: 10.1002/cctc.v8.6 |
| [30] |
Wang, Z.-J.; Zheng, S.; Matsui, J. K.; Liu, Z.; Molander, G. A. Chem. Sci. 2019, 10, 4389.
doi: 10.1039/C9SC00776H |
| [31] |
Cao, L.; Zheng, L.; Huang, Q. J. Organomet. Chem. 2014, 768, 56.
doi: 10.1016/j.jorganchem.2014.06.021 |
| [32] |
For selected examples see: (a) Xie, L.-Y.; Peng, S.; Tan, J.-X.; Sun, R.-X.; Yu, X.; Dai, N.-N.; Tang, Z.-L.; Xu, X.; He, W.-M. ACS Sustainable Chem. Eng. 2018, 6, 16976.
doi: 10.1021/acssuschemeng.8b04339 |
|
(b) Xie, L.-Y.; Peng, S.; Lu, L.-H.; Hu, J.; Bao, W.-H.; Zeng, F.; Tang, Z.; Xu, X.; He, W.-M. ACS Sustainable Chem. Eng. 2018, 6, 7989.
doi: 10.1021/acssuschemeng.8b04339 |
|
|
(c) Xie, L.-Y.; Peng, S.; Jiang, L.-L.; Peng, X.; Xia, W.; Yu, X.; Wang, X.-X.; Cao, Z.; He, W.-M. Org. Chem. Front. 2019, 6, 167.
doi: 10.1021/acssuschemeng.8b04339 |
|
| [33] |
Gutiérrez-Bonet, Á.; Remeur, C.; Matsui, J. K.; Molander, G. A. J. Am. Chem. Soc. 2017, 139, 12251.
doi: 10.1021/jacs.7b05899 |
| [34] |
Matsui, J. K.; Gutiérrez-Bonet, Á.; Rotella, M.; Alam, R.; Gutierrez, O.; Molander, G. A. Angew. Chem., Int. Ed. 2018, 57, 15847.
doi: 10.1002/anie.201809919 |
| [35] |
For selected examples see: (a) Gong, X.; Wang, M.; Ye, S.; Wu, J . Org. Lett. 2019, 21, 1156.
doi: 10.1021/acs.orglett.9b00100 |
|
(b) Ye, S.; Qiu, G.; Wu, J . Chem. Commun. 2019, 55, 1013.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(c) Ye, S.; Zheng, D.; Wu, J.; Qiu, G. Chem. Commun. 2019, 55, 2214.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(d) Ye, S.; Li, Y.; Wu, J.; Li, Z. Chem. Commun. 2019, 55, 2489.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(e) Gong, X.; Li, X.; Xie, W.; Wu, J.; Ye, S. Org. Chem. Front. 2019, 6, 1863.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(f) Zhang, J.; Xie, W.; Ye, S.; Wu, J. Org. Chem. Front. 2019, 6, 2254.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(g) Ye, S.; Xiang, T.; Li, X.; Wu, J. Org. Chem. Front. 2019, 6, 2183.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(h) Ye, S.; Li, X.; Xie, W.; Wu, J. Asian J. Org. Chem. 2019, 8, 893.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(i) Ye, S.; Li, X.; Xie, W.; Wu, J. Eur. J. Org. Chem. 2019, 10.1002/ejoc.201900396.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(j) Zhang, J.; Li, X.; Xie, W.; Ye, S.; Wu, J. Org. Lett. 2019, 21, DOI: 10.1021/acs.orglett.9b01323.
doi: 10.1021/acs.orglett.9b00100 |
|
|
(k) Zong, Y.; Lang, Y.; Yang, M.; Li, X.; Fan, X.; Wu, J. Org. Lett. 2019, 21, 1935.
doi: 10.1021/acs.orglett.9b00100 |
|
| [36] |
Wang, X.; Li, H.; Qiu, G.; Wu, J . Chem. Commun. 2019, 55, 2062.
doi: 10.1039/C8CC10246E |
| [37] |
Wang, X.; Yang, M.; Xie, W.; Fan, X.; Wu, J . Chem. Commun. 2019, 55, 6010.
doi: 10.1039/C9CC03004B |
| [38] |
Verrier, C.; Alandini, N.; Pezzetta, C.; Moliterno, M.; Buzzetti, L.; Hepburn, H. B.; Vega-Penaloza, A.; Silvi, M.; Melchiorre, P . ACS Catal. 2018, 8, 1062.
doi: 10.1021/acscatal.7b03788 |
| [39] |
Goti, G.; Bieszczad, B.; Vega-Penaloza, A.; Melchiorre, P. Angew. Chem., Int. Ed. 2019, 58, 1213.
doi: 10.1002/anie.201810798 |
| [40] |
de Assis, F. F.; Huang, X.; Akiyama, M.; Pilli, R. A.; Meggers, E. J. Org. Chem. 2018, 83, 10922.
doi: 10.1021/acs.joc.8b01588 |
| [41] |
Zhang, H.-H.; Zhao, J.-J.; Yu, S. J. Am. Chem. Soc. 2018, 140, 16914.
doi: 10.1021/jacs.8b10766 |
| [42] | Li, F.; Tian, D.; Fan, Y.; Lee, R.; Lu, G.; Yin, Y.; Qiao, B.; Zhao, X.; Xiao, Z.; Jiang, Z . Nat. Commun. 2019, DOI: 10.1038/s41467-019- 09857-9. |
| [1] | 邓沈娜, 彭常春, 牛云宏, 许云, 张云霄, 陈祥, 王红敏, 刘珊珊, 沈晓. 自由基Brook重排调控的α-氟烷基-α-硅基甲醇参与的烯烃双官能团化反应[J]. 化学学报, 2024, 82(2): 119-125. |
| [2] | 陈健强, 朱钢国, 吴劼. 镍催化氮杂环丙烷的开环偶联反应研究[J]. 化学学报, 2024, 82(2): 190-212. |
| [3] | 李雅宁, 王晓艳, 唐勇. 自由基聚合的立体选择性调控★[J]. 化学学报, 2024, 82(2): 213-225. |
| [4] | 李珊, 路俊欣, 刘杰, 蒋绿齐, 易文斌. 氟烷基亚磺酸钠盐电化学合成α-氟烷基酮[J]. 化学学报, 2024, 82(2): 110-114. |
| [5] | 易敬霖, 陈茂. 三氟氯乙烯与甲基异丙烯基醚的光诱导共聚反应★[J]. 化学学报, 2024, 82(2): 126-131. |
| [6] | 任妍妍, 李欣, 韩英锋. 基于氮杂环卡宾蓝光有机自由基的合成及其光学性质研究★[J]. 化学学报, 2023, 81(7): 735-740. |
| [7] | 刘坜, 郑刚, 范国强, 杜洪光, 谭嘉靖. 4-酰基/氨基羰基/烷氧羰基取代汉斯酯参与的有机反应研究进展[J]. 化学学报, 2023, 81(6): 657-668. |
| [8] | 杨洁, 凌琳, 李玉学, 吕龙. 高氯酸铵热分解机理的密度泛函理论研究[J]. 化学学报, 2023, 81(4): 328-337. |
| [9] | 赵亚婷, 刘帆, 汪秋安, 夏吾炯. 可见光促进(氮杂)芳香胺与重氮乙酸乙酯的N-烷基化反应[J]. 化学学报, 2023, 81(2): 111-115. |
| [10] | 陈健强, 朱钢国, 吴劼. 草酸酯类化合物在自由基脱羟基化反应中的研究进展[J]. 化学学报, 2023, 81(11): 1609-1623. |
| [11] | 张红丹, 兰欣雨, 程鹏. 羟基自由基辅助沸石分子筛合成的研究进展[J]. 化学学报, 2023, 81(1): 100-110. |
| [12] | 李小娟, 叶梓瑜, 谢书涵, 王永净, 王永好, 吕源财, 林春香. 氮氯共掺杂多孔碳活化过一硫酸盐降解苯酚的性能及机理研究[J]. 化学学报, 2022, 80(9): 1238-1249. |
| [13] | 岳广禄, 魏婧瑶, 邱頔, 莫凡洋. 芳基锡烷的合成研究进展[J]. 化学学报, 2022, 80(7): 956-969. |
| [14] | 吴波, 王冲, 李宝林, 王春儒. 光驱动的金属富勒烯分子磁开关※[J]. 化学学报, 2022, 80(2): 101-104. |
| [15] | 杨民, 叶柏柏, 陈健强, 吴劼. 可见光催化烷基磺酰自由基启动芳酰肼的烷基磺酰化反应[J]. 化学学报, 2022, 80(1): 11-15. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
