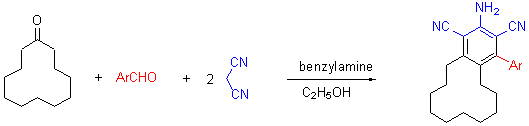
| [1] Murata, H.; Ishitani, H.; Iwamoto, M. Org. Biomol. Chem. 2010, 8, 1202. [2] Moafi, L.; Ahadi, S.; Khavasi, H. S.; Bazgir, A. Synthesis 2011, 1399. [3] Wang, H. Y.; Zou, Y.; Tao, C. Z. Chin. J. Org. Chem 2011, 31, 2161 (in Chinese). (王慧彦, 邹毅, 陶传洲, 有机化学, 2011, 31, 2161.) [4] Dumur, F.; Gautier, N.; Gallego-Planas, N.; Sahin, Y.; Levillain, E.; Mercier, N.; Hudhomme, P. J. Org. Chem. 2004, 69, 2164. [5] Xiao, Y.; Qian, X. H. Tetrahedron Lett. 2003, 44, 2087. [6] Sepiol, J.; Milart, P. Tetrahedron 1985, 41, 5261. [7] Griffiths, J.; Lockwood, M.; Roozpeikar, B. J. Chem. Soc., Perkin Trans. 1 1977, 1608. [8] Rong, L. C.; Han, H. X.; Jiang, H.; Tu, S. J. Synth. Commun. 2008, 38, 3530. [9] Rong, L. C.; Tao, S. M.; Xia, S.; Liu, L. H.; Yin, S.; Shi, Y. H. Res. Chem. Intermed. 2012, 38, 1647. [10] Rong, L. C.; Gao, L. J.; Han, H. X.; Jiang, H.; Dai, Y. S.; Tu, S. J. Synth. Commun. 2010, 40, 289. [11] Wang, X. S.; Zhang, M. M.; Li, Q.; Yao, C. S.; Tu, S. J. Tetrahedron 2007, 63, 5265. [12] Rong, L. C.; Han, H. X.; Jiang, H.; Tu, S. J. Synth. Commun. 2009, 9, 3493. [13] Cui, S. L.; Lin, X. F.; Wang, Y. G. J. Org. Chem. 2005, 70, 2866. [14] Shaterian, H. R.; Honarmand, M.; Oveisi, A. R. Monatsh. Chem. 2010, 141, 557. [15] Adib, M.; Mohammadi, B.; Ansari, S.; Bijanzadeh, H. R.; Zhu, L. Synthesis 2010, 1526. [16] Subhash, B.; Alissa, H.; Hari, K.; Grigoriy, S. Tetrahedron Lett. 2011, 52, 1878. [17] Yi, C.; Blum, C.; Liu, S.; Frei, G.; Neels, A.; Renaud, P.; Leutwyler, S.; Decurtins, S. J. Org. Chem. 2008, 73, 3596. [18] Xin, X.; Wang, Y.; Xu, W.; Lin, Y.; Duan, H.; Dong, D. Green Chem. 2010, 12, 893. |