化学学报 ›› 2026, Vol. 84 ›› Issue (4): 476-483.DOI: 10.6023/A25120419 上一篇 下一篇
研究论文
投稿日期:2025-12-25
发布日期:2026-01-09
通讯作者:
叶剑良, 黄培强
基金资助:
Guangsheng Lu, Zeng Han, Jianliang Ye*( ), Peiqiang Huang*(
), Peiqiang Huang*( )
)
Received:2025-12-25
Published:2026-01-09
Contact:
Jianliang Ye, Peiqiang Huang
Supported by:文章分享
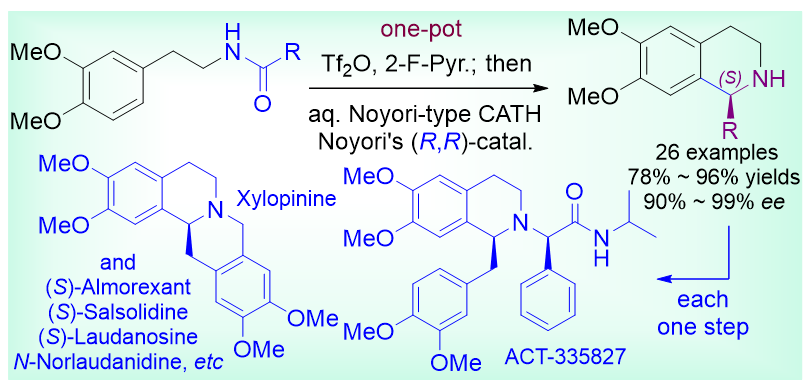
本工作采用先官能化再还原的酰胺不对称还原转化新策略实现了酰胺的“一瓶”催化不对称还原型Bischler-Napieralski反应. 该方法涉及串联进行的三氟甲磺酸酐/2-氟-吡啶(Tf2O/2-F-Pyr.)促进的Bischler-Napieralski脱水环化以及水相Noyori型催化不对称转移氢化(CATH)反应. 通过此一瓶法, 以高收率和优异的对映选择性合成了一系列四氢异喹啉衍生物. 该方法适用于酰基部分带有给电子或吸电子基团的N-芳乙基芳香酰胺, 以及N-芳乙基脂肪酰胺. 通过这一方法实现了四个生物碱, (S)-猪毛菜定碱、(S)-劳丹素、(S)-木防己碱、(S)-N-norlaudanidine, 以及药物分子ACT-335827的高效、催化对映选择性合成. 此外, 还实现了生物碱(S)-cryptostyline III及药物分子almorexant等的形式合成.
卢广生, 韩增, 叶剑良, 黄培强. 酰胺的“一瓶”催化不对称还原型Bischler-Napieralski反应: 生物活性1-取代四氢异喹啉的对映选择性合成[J]. 化学学报, 2026, 84(4): 476-483.
Guangsheng Lu, Zeng Han, Jianliang Ye, Peiqiang Huang. One-pot, Catalytic Asymmetric Reductive Bischler-Napieralski-Type Reaction of Amides: An Enantioselective Entrance to Biologically Active 1-Substituted Tetrahydroisoquinolines[J]. Acta Chimica Sinica, 2026, 84(4): 476-483.
| Entry | Variation from standard condition | Yielda/% | eeb/% |
|---|---|---|---|
| 1 | none | 96 | 94 |
| 2 | 1 mol% (R,R)-NC | 86 | 93 |
| 3 | 0.5 mol% (R,R)-NC | 52 | 90 |
| 4 | 0.1 mol% (R,R)-NC | 34 | 92 |
| 5 | no Bi(OTf)3 | 40 | 89 |
| 6 | no AgSbF6 | 33 | 91 |
| 7 | 3 mol% AgSbF6, 6 mol% Bi(OTf)3 | 65 | 89 |
| 8 | 2 mol% AgSbF6, 4 mol% Bi(OTf)3 | 45 | 91 |
| 9 | 20 equiv. HCOONa | 73 | 90 |
| 10 | 10 equiv. HCOONa | 65 | 89 |
| 11 | 40 equiv. HCOONa | 93 | 92 |
| 12 | HCOOK instead of HCOONa | 13 | 90 |
| 13 | HCOOLi instead of HCOONa | 8 | 90 |
| 14 | HCOONH4 instead of HCOONa | 49 | 89 |
| 15 | r.t. for 18 h | 85 | 92 |
| Entry | Variation from standard condition | Yielda/% | eeb/% |
|---|---|---|---|
| 1 | none | 96 | 94 |
| 2 | 1 mol% (R,R)-NC | 86 | 93 |
| 3 | 0.5 mol% (R,R)-NC | 52 | 90 |
| 4 | 0.1 mol% (R,R)-NC | 34 | 92 |
| 5 | no Bi(OTf)3 | 40 | 89 |
| 6 | no AgSbF6 | 33 | 91 |
| 7 | 3 mol% AgSbF6, 6 mol% Bi(OTf)3 | 65 | 89 |
| 8 | 2 mol% AgSbF6, 4 mol% Bi(OTf)3 | 45 | 91 |
| 9 | 20 equiv. HCOONa | 73 | 90 |
| 10 | 10 equiv. HCOONa | 65 | 89 |
| 11 | 40 equiv. HCOONa | 93 | 92 |
| 12 | HCOOK instead of HCOONa | 13 | 90 |
| 13 | HCOOLi instead of HCOONa | 8 | 90 |
| 14 | HCOONH4 instead of HCOONa | 49 | 89 |
| 15 | r.t. for 18 h | 85 | 92 |
| [1] |
(a)
doi: 10.1021/ar700155p pmid: 28791118 |
|
(b)
doi: 10.1073/pnas.0804348105 pmid: 28791118 |
|
|
(c)
doi: 10.1039/b821200g pmid: 28791118 |
|
|
(d)
doi: 10.1039/c5sc02913a pmid: 28791118 |
|
|
(e)
pmid: 28791118 |
|
|
(f)
doi: 10.1021/acs.accounts.0c00727 pmid: 28791118 |
|
|
(g)
doi: 10.1021/acs.accounts.0c00803 pmid: 28791118 |
|
|
(h)
doi: 10.1021/acs.chemrev.1c00631 pmid: 28791118 |
|
|
(i)
doi: 10.6023/A25070247 pmid: 28791118 |
|
|
(赵雅宸, 俞飚, 化学学报, 2025, 83, 1397).
doi: 10.6023/A25070247 pmid: 28791118 |
|
|
(j)
doi: 10.1039/D4QO02276A pmid: 28791118 |
|
|
(k)
pmid: 28791118 |
|
|
(l)
doi: 10.1002/cjoc.v43.1 pmid: 28791118 |
|
|
(m)
doi: 10.6023/A25050159 pmid: 28791118 |
|
|
(左恒昕宇, 虎亚光, 乔霞, 张野, 王少华, 化学学报, 2025, 83, 981.)
doi: 10.6023/A25050159 pmid: 28791118 |
|
| [2] |
(a)
doi: 10.1002/anie.v20:10 |
|
(b)
doi: 10.1016/j.tet.2019.05.024 |
|
| [3] |
For selected examples, see: (a) Bechara, W. S.; Pelletier, G.; Charette, A. B. Nat. Chem. 2012, 4, 228.
doi: 10.1038/nchem.1268 |
|
(b)
|
|
|
(c)
doi: 10.1021/ja105194s |
|
|
(d)
doi: 10.1139/v01-150 |
|
| [4] |
For selected examples, see: (a) White, K. L.; Mewald, M.; Movassaghi, M. J. Org. Chem. 2015, 80, 7403.
doi: 10.1021/acs.joc.5b01023 pmid: 17076488 |
|
(b)
doi: 10.1021/ol801264u pmid: 17076488 |
|
|
(c)
pmid: 17076488 |
|
|
(d)
pmid: 17076488 |
|
|
(e)
doi: 10.1021/jo802355d pmid: 17076488 |
|
| [5] |
For reviews, see: (a) Pace, V.; Holzer, W. Aust. J. Chem. 2013, 66, 507.
doi: 10.1071/CH13042 |
|
(b)
doi: 10.1002/adsc.v356.18 |
|
|
(c)
doi: 10.1039/C6CS00244G |
|
|
(d)
doi: 10.1039/C8CS00335A |
|
|
(e)
doi: 10.1039/C8OB00733K |
|
|
(f)
doi: 10.1021/acscatal.0c02377 |
|
|
(g)
doi: 10.1016/j.tetlet.2019.151423 |
|
|
(h)
doi: 10.1016/j.trechm.2020.07.001 |
|
|
(i)
doi: 10.3389/fchem.2021.655849 |
|
|
(j)
doi: 10.1055/a-1679-8205 |
|
|
(k)
doi: 10.1360/SSC-2023-0203 |
|
|
(罗艳龙, 中国科学:化学, 2023, 53, 129, 勘误: 罗艳龙, 中国科学:化学, 2024, 54, 968).
|
|
|
(l)
doi: 10.1021/acs.accounts.5c00366 |
|
| [6] |
For selected examples, see: (a) Yasui, S.; Aoki, S.; Okamura, T.; Sato, T. Chem. Lett. 2025, 54, upaf094.
pmid: 20672260 |
|
(b)
doi: 10.1002/anie.v64.29 pmid: 20672260 |
|
|
(c)
doi: 10.1002/anie.v63.6 pmid: 20672260 |
|
|
(d)
doi: 10.1246/bcsj.20230088 pmid: 20672260 |
|
|
(e)
doi: 10.1246/cl.230245 pmid: 20672260 |
|
|
(f)
doi: 10.1021/jacs.7b10944 pmid: 20672260 |
|
|
(g)
doi: 10.1002/anie.201001127 pmid: 20672260 |
|
| [7] |
For selected examples, see: (a)
doi: 10.1021/acs.orglett.3c01390 pmid: 25399919 |
|
(b)
doi: 10.1002/anie.v63.2 pmid: 25399919 |
|
|
(c)
doi: 10.1002/anie.202107536 pmid: 25399919 |
|
|
(d)
doi: 10.1002/anie.201612367 pmid: 25399919 |
|
|
(e)
doi: 10.1002/chem.201405256 pmid: 25399919 |
|
| [8] |
For catalytic asymmetric reductive transformations, see: (a)
doi: 10.1002/anie.v62.25 pmid: 31479277 |
|
(b)
doi: 10.1038/s41467-023-41846-x pmid: 31479277 |
|
|
(c)
pmid: 31479277 |
|
|
(d)
doi: 10.1002/anie.v60.16 pmid: 31479277 |
|
|
(e)
doi: 10.1021/acs.orglett.9b02862 pmid: 31479277 |
|
|
For selected non-asymmetric transformations, see:(f)
pmid: 31479277 |
|
|
(g)
doi: 10.1007/s11426-025-2935-3 pmid: 31479277 |
|
|
(h)
doi: 10.6023/cjoc202408032 pmid: 31479277 |
|
|
(徐业鹏, 阮义, 郑剑峰, 黄培强, 有机化学, 2025, 45, 988.)
doi: 10.6023/cjoc202408032 pmid: 31479277 |
|
|
(i)
pmid: 31479277 |
|
|
(j)
doi: 10.1007/s11426-022-1501-y pmid: 31479277 |
|
|
(k)
doi: 10.6023/A23050226 pmid: 31479277 |
|
|
(何倩, 李杰, 喻思佳, 吴东坪, 叶剑良, 黄培强, 化学学报, 2023, 81, 1265.)
doi: 10.6023/A23050226 pmid: 31479277 |
|
|
(l)
doi: 10.1039/C6CC05318A pmid: 31479277 |
|
|
(m)
doi: 10.1002/anie.v49:17 pmid: 31479277 |
|
| [9] |
For selected examples, see: (a)
doi: 10.1021/acs.orglett.6b01226 |
|
(b)
doi: 10.1002/adsc.v366.10 |
|
|
(c)
doi: 10.1055/s-0037-1611549 |
|
|
(d)
doi: 10.1002/adsc.v362.22 |
|
| [10] |
For selected examples, see: (a)
doi: 10.1016/j.tet.2014.01.039 |
|
(b)
doi: 10.1021/acs.joc.5b00335 |
|
| [11] |
For selected examples, see: (a)
doi: 10.1002/anie.v60.51 |
|
(b)
doi: 10.1002/anie.v60.31 |
|
|
(c)
doi: 10.1039/D3GC03193D |
|
| [12] |
For selected examples, see: (a)
doi: 10.1002/anie.v63.14 pmid: 22770123 |
|
(b)
doi: 10.1021/jacs.3c14517 pmid: 22770123 |
|
|
(c)
doi: 10.1002/anie.v62.37 pmid: 22770123 |
|
|
(d)
doi: 10.1002/anie.v61.36 pmid: 22770123 |
|
|
(e)
doi: 10.1021/acs.joc.2c00131 pmid: 22770123 |
|
|
(f)
doi: 10.1002/anie.201807735 pmid: 22770123 |
|
|
(g)
doi: 10.1021/ja304547s pmid: 22770123 |
|
|
(h)
pmid: 22770123 |
|
|
For a recent application, see:(i)
doi: 10.1021/acs.joc.3c02366 pmid: 22770123 |
|
|
For the reaction of vinylogous amides, see:(j)
doi: 10.1021/acs.joc.3c02108 pmid: 22770123 |
|
| [13] |
For selected examples, see: (a)
doi: 10.1002/anie.v60.35 |
|
(b)
doi: 10.1021/jacs.1c04363 |
|
|
(c)
doi: 10.1016/j.chempr.2019.05.006 |
|
| [14] |
(a)
doi: 10.1021/acs.chemrev.3c00054 pmid: 29324343 |
|
(b)
pmid: 29324343 |
|
|
(c)
doi: S0223-5234(17)31129-7 pmid: 29324343 |
|
| [15] |
(a)
doi: 10.1021/ar9502341 pmid: 15250744 |
|
(b)
doi: 10.1021/ja960364k pmid: 15250744 |
|
|
(c)
doi: 10.1016/j.tet.2011.11.031 pmid: 15250744 |
|
|
(d)
doi: 10.1021/op3000543 pmid: 15250744 |
|
|
(e)
pmid: 15250744 |
|
|
(f)
pmid: 15250744 |
|
|
(g)
doi: 10.1007/s11244-012-9854-7 pmid: 15250744 |
|
|
(h)
doi: 10.1002/adsc.v355.18 pmid: 15250744 |
|
|
(i)
doi: 10.3390/catal14120884 pmid: 15250744 |
|
| [16] |
(a)
doi: 10.1016/j.tetasy.2005.10.007 |
|
(b)
doi: 10.1002/ejoc.v2015.29 |
|
|
(c)
doi: 10.1021/acs.oprd.2c00243 |
|
| [17] |
|
| [18] |
(a)
doi: 10.1021/om011059x pmid: 16268617 |
|
(b)
pmid: 16268617 |
|
|
(c)
pmid: 16268617 |
|
|
(d)
doi: 10.1039/b403627a pmid: 16268617 |
|
|
(e)
doi: 10.1021/ja8050958 pmid: 16268617 |
|
|
(f)
doi: 10.1002/chem.v15:47 pmid: 16268617 |
|
| [19] |
pmid: 2296014 |
| [20] |
doi: 10.1039/c0cc01487g |
| [21] |
|
| [22] |
doi: 10.1111/bph.1994.113.issue-4 |
| [23] |
doi: 10.1097/00003643-200207000-00002 |
| [24] |
(a)
doi: 10.1021/acs.joc.1c00602 pmid: 21563765 |
|
(b)
doi: 10.1021/jo2007237 pmid: 21563765 |
|
| [25] |
doi: 10.1002/cmdc.201300003 pmid: 23589487 |
| [26] |
(a)
doi: 10.1002/cjoc.v36.2 |
|
(b)
doi: 10.1016/j.bmc.2007.05.059 |
|
| [27] |
(a)
doi: 10.1016/s0092-8674(00)80949-6 pmid: 9491897 |
|
(b)
doi: 10.1016/j.tetlet.2014.03.130 pmid: 9491897 |
|
| [28] |
(a)
doi: 10.3891/acta.chem.scand.23-0244 |
|
(b)
doi: 10.1002/hlca.v54:6 |
|
| [29] |
pmid: 7996543 |
| [30] |
|
| [1] | 刘洪昤, 刘艺婷, 王新华, 何福生, 吴劼. 碱促进次磺酰胺与高价碘(III)试剂的硫-氟烷基化反应[J]. 化学学报, 2026, 84(3): 287-292. |
| [2] | 杜云哲, 李曙懿, 程思柳, 贾慧劼, 王政, 韩利民, 竺宁. H2S促使环脒开环与醛发生三组分串联反应高效构建硫代酰胺类衍生物[J]. 化学学报, 2026, 84(1): 93-100. |
| [3] | 林健, 李恒渊, 周波, 叶龙武. 无过渡金属参与的炔酰胺与吲哚酮的多样性环化反应[J]. 化学学报, 2025, 83(6): 551-556. |
| [4] | 杨雪, 刘妍伶, 陈霞, 周晓玉, 王爱玲, 刘海龙. 草酰单胺配体促进的低剂量钯催化水相Suzuki偶联反应[J]. 化学学报, 2025, 83(4): 354-359. |
| [5] | 李国凯, 朱滨锋, 胡涛, 樊瑞峰, 孙蔚青, 和振秀, 陈景超, 樊保敏. 光催化叔胺的脱烷基酰化反应[J]. 化学学报, 2025, 83(3): 199-205. |
| [6] | 孟凡荣, 李国锋, 赵杰, 肖文精, 石德清, 陈加荣. 光催化氮自由基参与的乙烯基环丙烷加成/环化反应合成四氢吡啶★[J]. 化学学报, 2025, 83(12): 1472-1479. |
| [7] | 雷平, 苏秦, 王栋, Shahid Ali Khan, 阿布拉江•克依木. 无催化剂光化学合成吲哚酮: 电子供体-受体复合物介导的N-芳基丙烯酰胺环化反应[J]. 化学学报, 2025, 83(11): 1379-1385. |
| [8] | 卡迪尔亚•阿布都外力, 热孜古丽•玉努斯, 李佳佳, 罗时玮, 阿布都热西提•阿布力克木. N-烷氧基酰胺无溶剂氧化脱氢N—N自偶联反应研究[J]. 化学学报, 2024, 82(7): 731-735. |
| [9] | 翟彤仪, 葛畅, 钱鹏程, 周波, 叶龙武. Brønsted酸催化炔酰胺分子内氢烷氧化/Claisen重排反应★[J]. 化学学报, 2023, 81(9): 1101-1107. |
| [10] | 王瑞祥, 赵庆如, 顾庆, 游书力. 金/铱接力催化炔基酰胺环化/不对称烯丙基苄基化串联反应★[J]. 化学学报, 2023, 81(5): 431-434. |
| [11] | 何倩, 李杰, 喻思佳, 吴东坪, 叶剑良, 黄培强. 铱催化叔酰胺与呋喃硅醚间的类插烯Aldol缩合反应: γ-亚苄基-丁烯酸内酯的合成★[J]. 化学学报, 2023, 81(10): 1265-1270. |
| [12] | 李善武, 朱陈宇杰, 罗尹豪, 张亚茹, 滕汉明, 王宗瑞, 甄永刚. 酰胺与酰亚胺类n型有机半导体材料的研究进展[J]. 化学学报, 2022, 80(12): 1600-1617. |
| [13] | 卢小彪, 肖茜, 万常峰, 汪志勇, 刘晋彪. 无金属条件下的肉桂酸类和酰胺的脱羧氧化偶联反应构建碳-碳键[J]. 化学学报, 2021, 79(6): 751-754. |
| [14] | 方婧, 赵文娟, 张明浩, 方千荣. 一种新型酰胺功能化的共价有机框架用于选择性染料吸附[J]. 化学学报, 2021, 79(2): 186-191. |
| [15] | 杜重阳, 陈耀峰. 二乙基锌促进CO2的硅氢化反应以及CO2为C1合成子的有机胺甲酰化或脲化反应[J]. 化学学报, 2020, 78(9): 938-944. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||