有机化学 ›› 2025, Vol. 45 ›› Issue (4): 1166-1177.DOI: 10.6023/cjoc202407039 上一篇 下一篇
综述与进展
乔秀秀, 李倩, 赵世娜, 魏瑞琪, 马桃, 何永辉*( ), 赵晓静*(
), 赵晓静*( )
)
收稿日期:2024-10-16
修回日期:2026-11-26
发布日期:2024-12-05
基金资助:
Xiu-Xiu Qiao, Qian Li, Shi'na Zhao, Ruiqi Wei, Tao Ma, Yong-Hui He( ), Xiao-Jing Zhao(
), Xiao-Jing Zhao( )
)
Received:2024-10-16
Revised:2026-11-26
Published:2024-12-05
Contact:
* E-mail: Supported by:文章分享
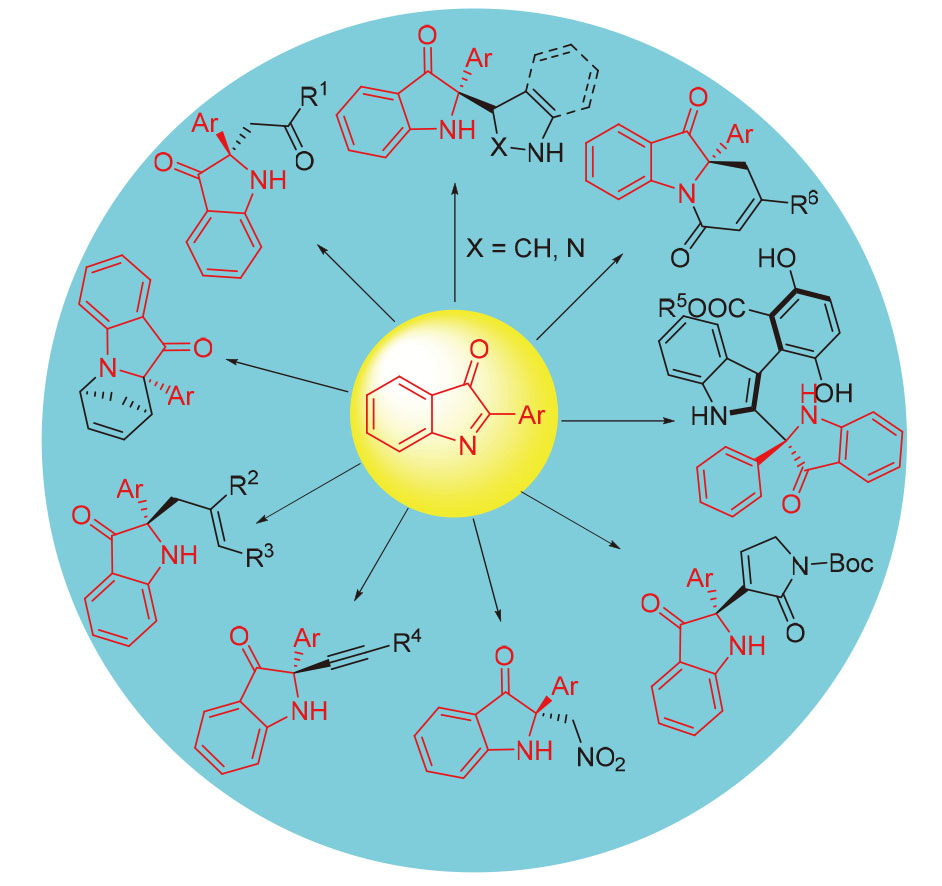
含有2,2-二取代吲哚啉-3-酮类骨架的化合物在天然产物、手性药物等诸多领域有着广泛应用. 通过不对称催化高效构建2,2-二取代吲哚啉-3--酮类化合物受到越来越广泛的关注. 综述了近些年国内外在以2-取代的3H-吲哚-3-酮类化合物为底物的不对称催化合成2,2-二取代吲哚啉-3-酮类化合物方面取得的研究进展, 主要从不对称aza- Friedel-Crafts反应、Mannich反应、aza-Diels-Alder反应、aza-Henry反应、aza-Morita-Baylis-Hillman反应以及其他反应等方面进行讨论, 为今后不对称合成C2季碳中心吲哚啉-3-酮类化合物研究提供参考.
乔秀秀, 李倩, 赵世娜, 魏瑞琪, 马桃, 何永辉, 赵晓静. 2-取代的3H-吲哚-3-酮类化合物参与的C2位手性吲哚啉-3-酮类化合物的不对称合成研究进展[J]. 有机化学, 2025, 45(4): 1166-1177.
Xiu-Xiu Qiao, Qian Li, Shi'na Zhao, Ruiqi Wei, Tao Ma, Yong-Hui He, Xiao-Jing Zhao. Research Progress in Asymmetric Synthesis of C2 Chiral Indolin-3-ones Involving 2-Substituted-3H-indol-3-ones[J]. Chinese Journal of Organic Chemistry, 2025, 45(4): 1166-1177.
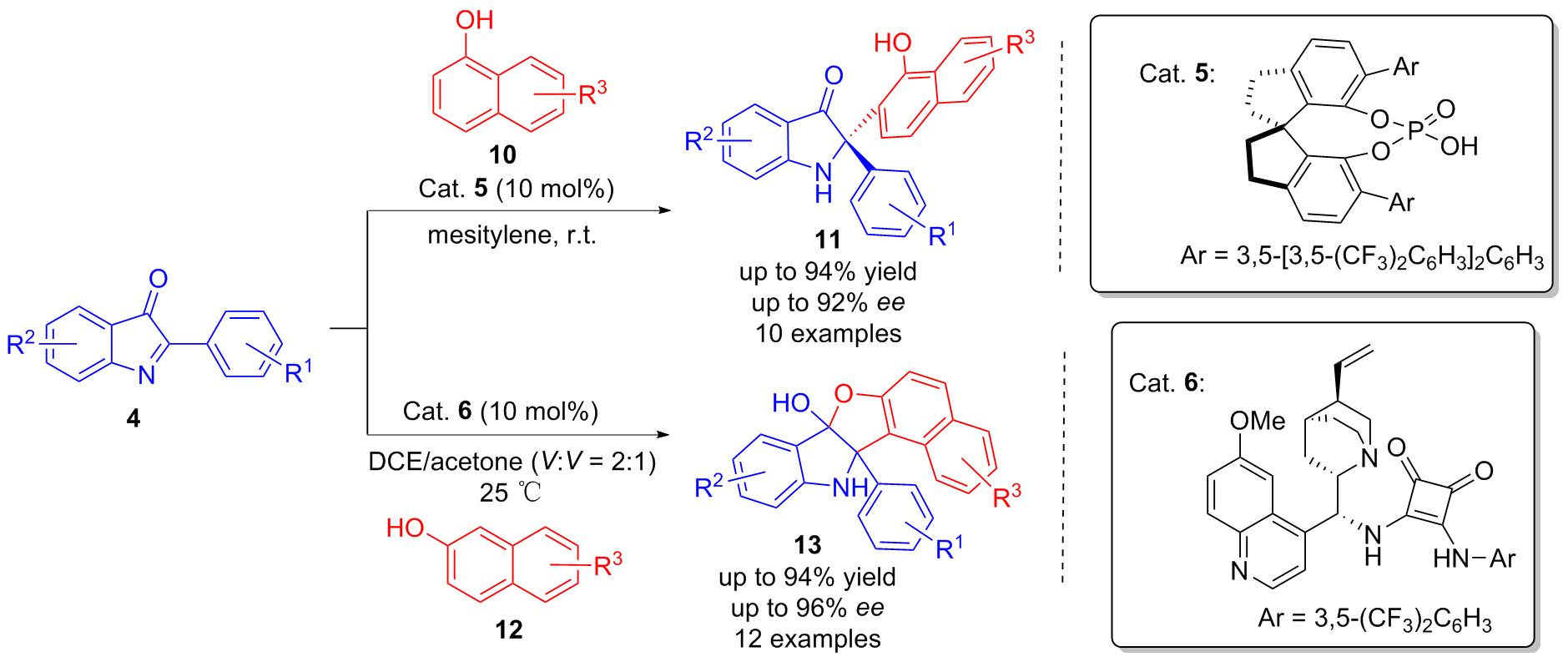
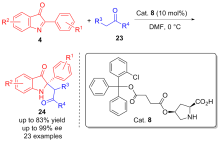

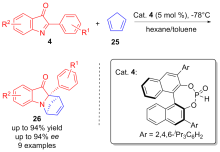
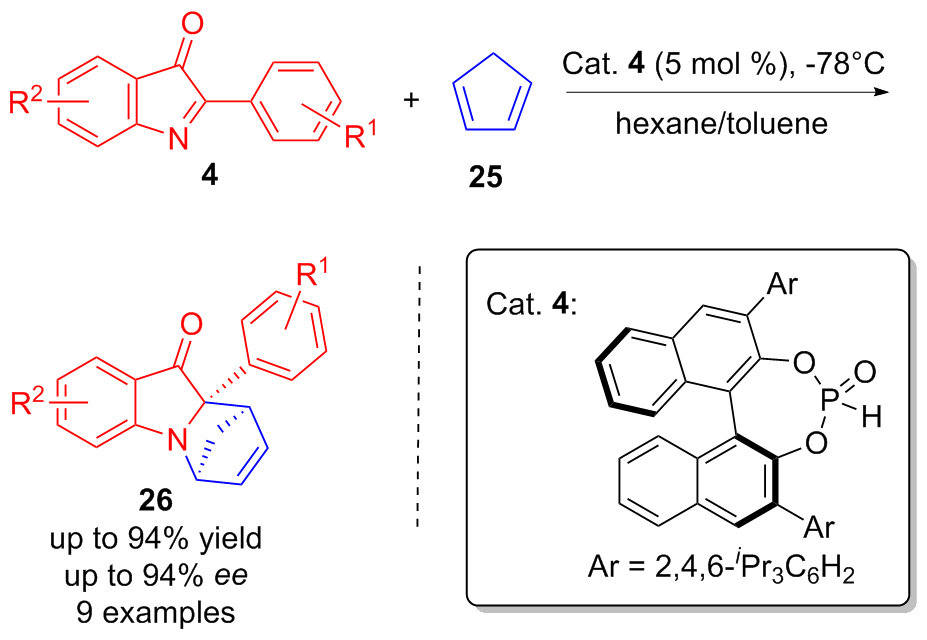
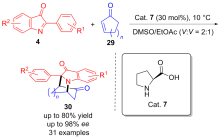
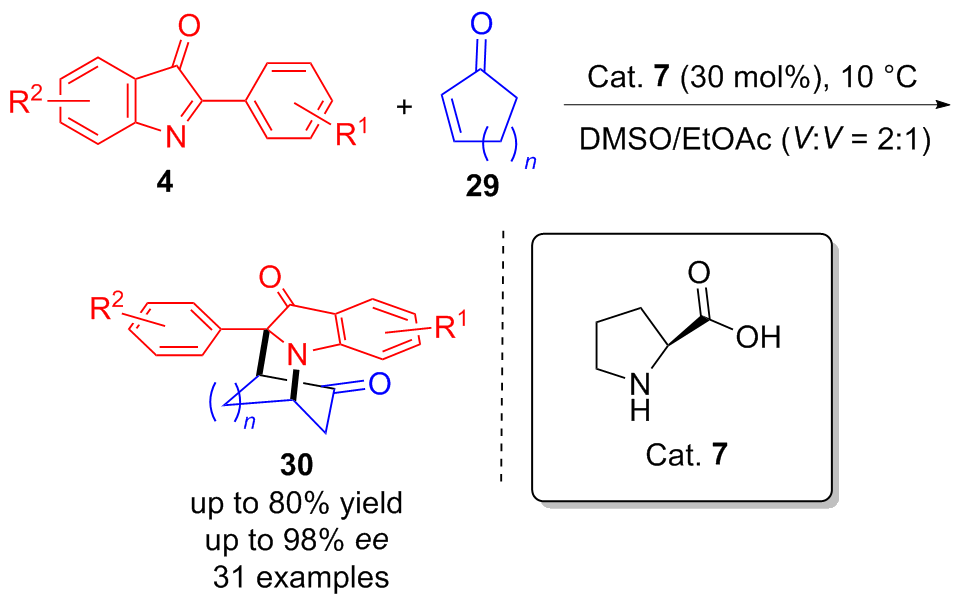




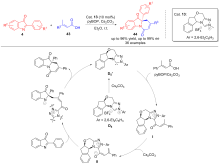

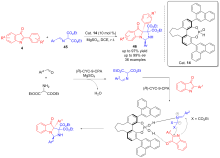
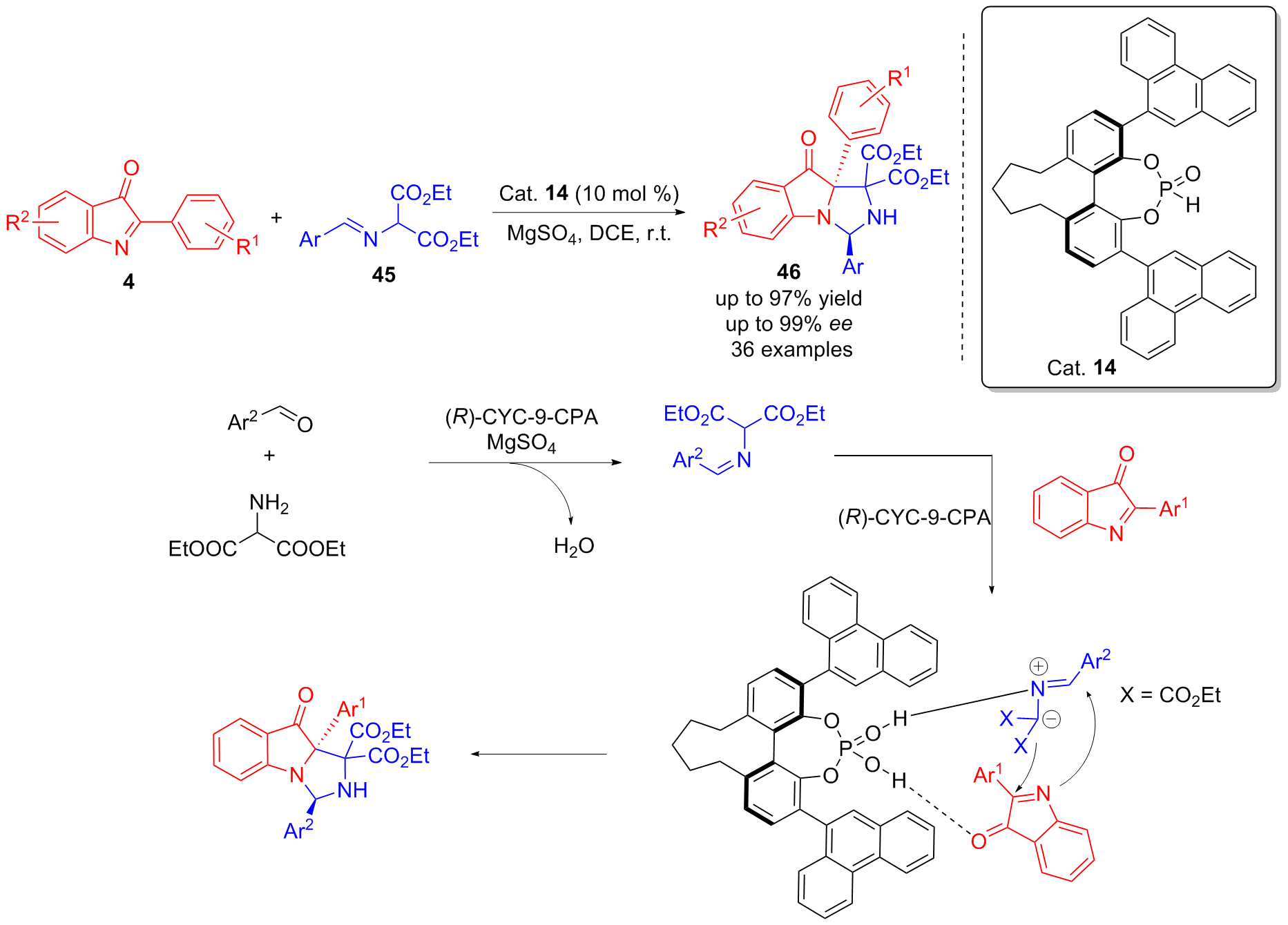
| [1] |
Dhote, P. S.; Patel, P.; Vanka, K.; Ramana, C. V. Org. Biomol. Chem. 2021, 19, 7970.
|
| [2] |
Liu, J.-F.; Jiang, Z.-Y.; Wang, R.-R.; Zheng, Y.-T.; Chen, J.-J.; Zhang, X.-M.; Ma, Y.-B.; Isatisine, A. Org. Lett. 2007, 9, 4127.
pmid: 17850153 |
| [3] |
Váradi, A.; Marrone, G. F.; Palmer, T. C.; Narayan, A.; Szabó, M. R.; Le Rouzic, V.; Grinnell, S. G.; Subrath, J. J.; Warner, E.; Kalra, S.; Hunkele, A.; Pagirsky, J.; Eans, S. O.; Medina, J. M.; Xu, J.; Pan, Y.-X.; Borics, A.; Pasternak, G. W.; McLaughlin, J. P.; Majumdar, S. J. Med. Chem. 2016, 59, 8381.
|
| [4] |
(a) Wang, S.-G.; You, S.-L. Angew. Chem.,Int. Ed. 2014, 53, 2194.
|
|
(b) You, S.-L.; Cai, Q.; Zeng, M. Chem. Soc. Rev. 2009, 38, 2190.
|
|
|
(c) Zhou, D.; Huang, Z.; Yu, X.; Wang, Y.; Li, J.; Wang, W.; Xie, H. Org. Lett. 2015, 17, 5554.
|
|
|
(d) Ling, J.; Lam, S. K.; Lo, B.; Lam, S.; Wong, W.-T.; Sun, J.; Chen, G.; Chiu, P. Org. Chem. Front. 2016, 3, 457.
|
|
|
(e) Svestka, D.; Otevrel, J.; Bobal, P. Adv. Synth. Catal. 2022, 364, 2174.
|
|
| [5] |
Yin, Q.; You, S.-L. Chem. Sci. 2011, 2, 1344.
|
| [6] |
Rueping, M.; Raja, S.; Núñez, A. Adv. Synth. Catal. 2011, 353, 563.
|
| [7] |
Nakamura, S.; Matsuda, N.; Ohara, M. Chem.-Eur. J. 2016, 22, 9478.
doi: 10.1002/chem.201601573 pmid: 27124556 |
| [8] |
(a) Yarlagadda, S.; Sridhar, B.; Subba Reddy, B. V. Chem. Asian J. 2018, 13, 1327.
|
|
(b) Dong, C.-L.; Ding, X.; Huang, L.-Q.; He, Y.-H.; Guan, Z. Org. Lett. 2020, 22, 1076.
|
|
|
(c) Lu, F.-Y.; Chen, Y.-J.; Chen, Y.; Ding, X.; Guan, Z.; He, Y.-H. Chem. Commun. 2020, 56, 623.
|
|
| [9] |
Ma, T.; He, Y.; Qiao, X.-X.; Zou, C.-P.; Wu, X.-X.; Li, G.; Zhao, X.-J. Org. Biomol. Chem. 2023, 21, 489.
|
| [10] |
Qiao, X.-X.; He, Y.; Ma, T.; Zou, C.-P.; Wu, X.-X.; Li, G.; Zhao, X.-J. Chem. Eur. J. 2023, 29, e202203914.
|
| [11] |
Zheng, J.; Rong, M.-Y.; Feng, F.-F.; Zhang, F.-G.; Cheung, C. W.; Ma, J.-A. Asian J. Org. Chem. 2023, 12, e202300204.
|
| [12] |
Merad, J.; Lalli, C.; Bernadat, G.; Maury, J.; Masson, G. Chem. Eur. J. 2018, 24, 3925.
|
| [13] |
(a) Reddy, K. N.; Rao, M. V. K.; Sridhar, B.; Subba Reddy, B. V. Chem. Asian J. 2019, 14, 2958.
pmid: 25203602 |
|
(b) Wu, H.-C.; Wang, C.; Chen, Y.-H.; Liu, Y.-K. Chem. Commun. 2021, 57, 1762.
pmid: 25203602 |
|
|
(c) Parmar, D.; Sugiono, E.; Raja, S.; Rueping, M. Chem. Rev. 2014, 114, 9047.
doi: 10.1021/cr5001496 pmid: 25203602 |
|
|
(d) Zhu, Y.; Li, Y.; Meng, Q.; Li, X. Org. Chem. Front. 2016, 3, 709.
pmid: 25203602 |
|
|
(e) Ting, A.; Schaus, S. E. Eur. J. Org. Chem. 2007, 2007, 5797.
pmid: 25203602 |
|
|
(f) Córdova, A. Acc. Chem. Res. 2004, 37, 102.
pmid: 25203602 |
|
|
(g) List, B. J. Am. Chem. Soc. 2000, 122, 9336.
pmid: 25203602 |
|
| [14] |
Li, L.; Han, M.; Xiao, M.; Xie, Z. Synlett 2011, 2011, 1727.
|
| [15] |
Rueping, M.; Rasappan, R.; Raja, S. Helv. Chim. Acta 2012, 95, 2296.
|
| [16] |
Li, J.-S.; Liu, Y.-J.; Zhang, G.-W.; Ma, J.-A. Org. Lett., 2017, 19, 6364.
|
| [17] |
Li, J.-S.; Liu, Y.-J.; Li, S.; Ma, J.-A. Chem. Commun. 2018, 54, 9151.
|
| [18] |
An, J.-X.; Yang, F.-F.; Wang, P.; Gu, Z.-C.; Li, Y.; Chen, L.; Zhao, Y.-L.; He, B. RSC Adv. 2022, 12, 7040.
|
| [19] |
(a) Masson, G.; Lalli, C.; Benohoud, M.; Dagousset, G. Chem. Soc. Rev. 2013, 42, 902.
pmid: 27748978 |
|
(b) Eschenbrenner-Lux, V.; Kumar, K.; Waldmann, H. Angew. Chem., Int. Ed. 2014, 53, 11146.
pmid: 27748978 |
|
|
(c) Cao, M.-H.; Green, N. J.; Xu, S.-Z. Org. Biomol. Chem. 2017, 15, 3105.
pmid: 27748978 |
|
|
(d) Hatanaka, Y.; Nantaku, S.; Nishimura, Y.; Otsuka, T.; Sekikaw, T. Chem. Commun. 2017, 53, 8996.
pmid: 27748978 |
|
|
(e) Wang, C.; Li, Y.; Wu, Y.; Wang, Q.; Shi, W.; Yuan, C.; Zhou, L.; Xiao, Y.; Guo, H. Org. Lett. 2018, 20, 2880.
pmid: 27748978 |
|
|
(f) Tomifuji, R.; Kurahashi, T.; Matsubara, S. Chem.-Eur. J. 2019, 25, 8987.
doi: 10.1002/chem.201901563 pmid: 27748978 |
|
|
(g) Weilbeer, C.; Sickert, M.; Naumov, S.; Schneider, C. Chem.-Eur. J. 2017, 23, 513.
doi: 10.1002/chem.201604356 pmid: 27748978 |
|
| [20] |
Rueping, M.; Raja, S. Beilstein J. Org. Chem. 2012, 8, 1819.
doi: 10.3762/bjoc.8.208 pmid: 23209517 |
| [21] |
Liu, J.-X.; Zhou, Q.-Q.; Deng, J.-G.; Chen, Y.-C. Org. Biomol. Chem. 2013, 11, 8175.
|
| [22] |
Yadav, J.; Dolas, A. J.; Iype, E.; Rangan, K.; Ohshita, J.; Kumar, D.; Kumar, I. J. Org. Chem. 2021, 86, 17213.
|
| [23] |
Zhao, Q.; Li, Y.; Zhang, Q.-X.; Cheng, J.-P.; Li, X. Angew. Chem., Int. Ed. 2021, 60, 17608.
|
| [24] |
(a) Liu, X.; Zheng, K.; Feng, X. Synthesis 2014, 46, 2241.
|
|
(b) Hack, D.; Blümel, M.; Chauhan, P.; Philipps, A. R.; Enders, D. Chem. Soc. Rev. 2015, 44, 6059.
|
|
|
(c) Saha, P.; Saikia, A. K. Org. Biomol. Chem. 2018, 16, 2820.
|
|
|
(d) Bakhtiari, A.; Safaei-Ghomi, J. Synlett 2019, 30, 1738.
doi: 10.1055/s-0037-1611875 |
|
| [25] |
Zhang, Q.-X.; Li, Y.; Wang, J.; Yang, C.; Liu, C.-J.; Li, X.; Cheng, J.-P. Angew. Chem., Int. Ed. 2020, 59, 4550.
|
| [26] |
(a) Blay, G.; Brines, A.; Monleón, A.; Pedro, J. R. Chem.-Eur. J. 2012, 18, 2440.
|
|
(b) Zhang, F.-G.; Ma, H.; Nie, J.; Zheng, Y.; Gao, Q.; Ma, J.-A. Adv. Synth. Catal. 2012, 354, 1422.
|
|
|
(c) Liu, T.-L.; Zhang, H.-X.; Zheng, Y.; Yao, Q.; Ma, J.-A. Chem. Commun. 2012, 48, 12234.
|
|
|
(d) De Munck, L.; Monleón, A.; Vila, C.; Pedro, J. R. Adv. Synth. Catal. 2017, 359, 1582.
|
|
| [27] |
Wu, X.-X.; Ma, T.; Qiao, X.-X.; Zou, C.-P.; Li, G.; He, Y.; Zhao, X.-J. Chem. Asian. J. 2023, 18, e202300526.
|
| [28] |
(a) Fang, X.; Wang, C.-J. Chem. Commun. 2015, 51, 1185.
|
|
(b) Otocka, S.; Kwiatkowska, M.; Madalińska, L.; Kiełbasiński, P. Chem. Rev. 2017, 117, 4147.
|
|
|
(c) Ping, X.-N.; Wei, P.-S.; Zhu, X.-Q.; Xie, J.-W. J. Org. Chem. 2017, 82, 2205.
|
|
|
(d) Devannah, V.; Sharma, R.; Watson, D. A. J. Am. Chem. Soc. 2019, 141, 8436.
|
|
|
(e) Bär, A.; Bär, S. I.; Schobert, R. Org. Biomol. Chem. 2020, 18, 7565.
|
|
| [29] |
Parra, A.; Alfaro, R.; Marzo, L.; Moreno-Carrasco, A.; García Ruano, J. L.; Alemán, J. Chem. Commun. 2012, 48, 9759.
|
| [30] |
Wei, Y.; Shi, M. Chem. Rev. 2013, 113, 6659.
|
| [31] |
Wu, X.-X.; He, Y.; Qiao, X.-X.; Ma, T.; Zou, C.-P.; Li, G.; Zhao, X.-J. J. Org. Chem. 2023, 88, 6599.
|
| [32] |
(a) Cheng, J. K.; Xiang, S.-H.; Li, S.; Ye, L.; Tan, B. Chem. Rev. 2021, 121, 4805.
|
|
(b) Da, B.-C.; Xiang, S.-H.; Li, S.; Tan, B. Chin. J. Chem. 2021, 39, 1787.
|
|
|
(c) Wang, Y. B.; Tan, B. Acc. Chem. Res. 2018, 51, 534.
|
|
|
(d) Kitagawa, O. Acc. Chem. Res. 2021, 54, 719.
|
|
| [33] |
Yuan, X.; Wu, X.; Peng, F.; Yang, H.; Zhu, C.; Fu, H. Chem. Commun. 2020, 56, 12648.
|
| [34] |
Fang, S.; Jin, S.; Ma, R.; Lu, T.; Du, D. Org. Lett. 2019, 21, 5211.
|
| [35] |
Yuan, X.; Wu, X.; Zhang, P.; Peng, F.; Liu, C.; Yang, H.; Zhu, C.; Fu, H. Org. Lett. 2019, 21, 2498.
doi: 10.1021/acs.orglett.8b04012 pmid: 30942603 |
| [1] | 王霜, 毛羊杰, 娄绍杰, 许丹倩. 基于氧化型导向基团的不对称C—H键官能团化反应研究进展[J]. 有机化学, 2025, 45(6): 1961-1994. |
| [2] | 唐梦瑶, 杨晓瑜. 手性磷酸催化不对称亲电胺化反应研究进展[J]. 有机化学, 2025, 45(6): 1785-1818. |
| [3] | 杜一鸣, 贾均松, 李玉龙, 舒伟. 手性α-芳基酮的催化合成研究进展[J]. 有机化学, 2025, 45(6): 1838-1870. |
| [4] | 苏雷, 杨熙, 闫捷, 蒋元力, 陈丽娟, 郑庆舒, 刘家旺. 不对称羰基化偶联反应研究进展[J]. 有机化学, 2025, 45(6): 2007-2047. |
| [5] | 蒋晨阳, 尹艳丽, 江智勇. 光酶催化不对称自由基加成反应研究进展[J]. 有机化学, 2025, 45(5): 1614-1633. |
| [6] | 杨之同, 宋恒谦, 雷盼, 闫嘉航, 谢卫青. Communesin生物碱核心五环骨架的高效合成[J]. 有机化学, 2025, 45(3): 1003-1008. |
| [7] | 林恩泽, 李必杰. 基于碳氢键断裂的金属催化的内烯烃不对称氢芳基化进展[J]. 有机化学, 2025, 45(2): 546-558. |
| [8] | 邹瑜, 郭伟聪, 汪君. 不含手性取代基的平面手性环戊二烯基铑催化剂在不对称碳氢键活化中的应用[J]. 有机化学, 2025, 45(2): 466-476. |
| [9] | 王家晟, 王泽树, 何卫民, 叶龙武. 邻炔基苯胺氢胺化合成轴手性吲哚研究进展[J]. 有机化学, 2024, 44(6): 1786-1792. |
| [10] | 刘晨光. 含氮芳香性杂环化合物的不对称氢化反应研究进展[J]. 有机化学, 2024, 44(5): 1403-1422. |
| [11] | 高淳, 刘欣, 王明慧, 刘淑贤, 朱婷婷, 张怡康, 郝二军, 杨启亮. 电化学不对称合成反应的研究进展[J]. 有机化学, 2024, 44(3): 673-727. |
| [12] | 陈宛婷, 钟雄威, 邢佳乐, 吴昌书, 高杨. C—N轴手性化合物的不对称催化合成研究进展[J]. 有机化学, 2024, 44(2): 349-377. |
| [13] | 杨爽, 房新强. 氮杂环卡宾催化实现的动力学拆分近期研究进展[J]. 有机化学, 2024, 44(2): 448-480. |
| [14] | 孙婷珈, 孙国银, 孙威, 彭雪松, 廖娟, 游勇, 袁伟成. 碳-碳不饱和键的催化不对称氢-膦(磷)官能化反应研究进展[J]. 有机化学, 2024, 44(12): 3647-3677. |
| [15] | 李晖, 殷亮. 铜催化的手性膦化合物不对称合成研究进展[J]. 有机化学, 2024, 44(12): 3575-3586. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||