有机化学 ›› 2026, Vol. 46 ›› Issue (2): 594-602.DOI: 10.6023/cjoc202507003 上一篇 下一篇
研究论文
收稿日期:2025-07-02
修回日期:2025-08-24
发布日期:2025-10-13
通讯作者:
王全瑞
基金资助:
Wentao Guo, Shuyu Meng, Quanrui Wang*( )
)
Received:2025-07-02
Revised:2025-08-24
Published:2025-10-13
Contact:
Quanrui Wang
Supported by:文章分享
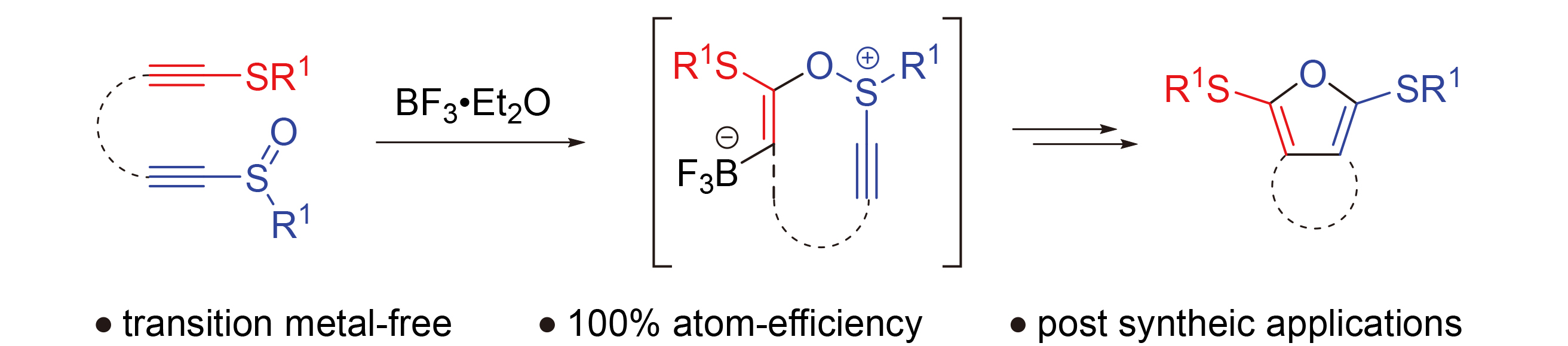
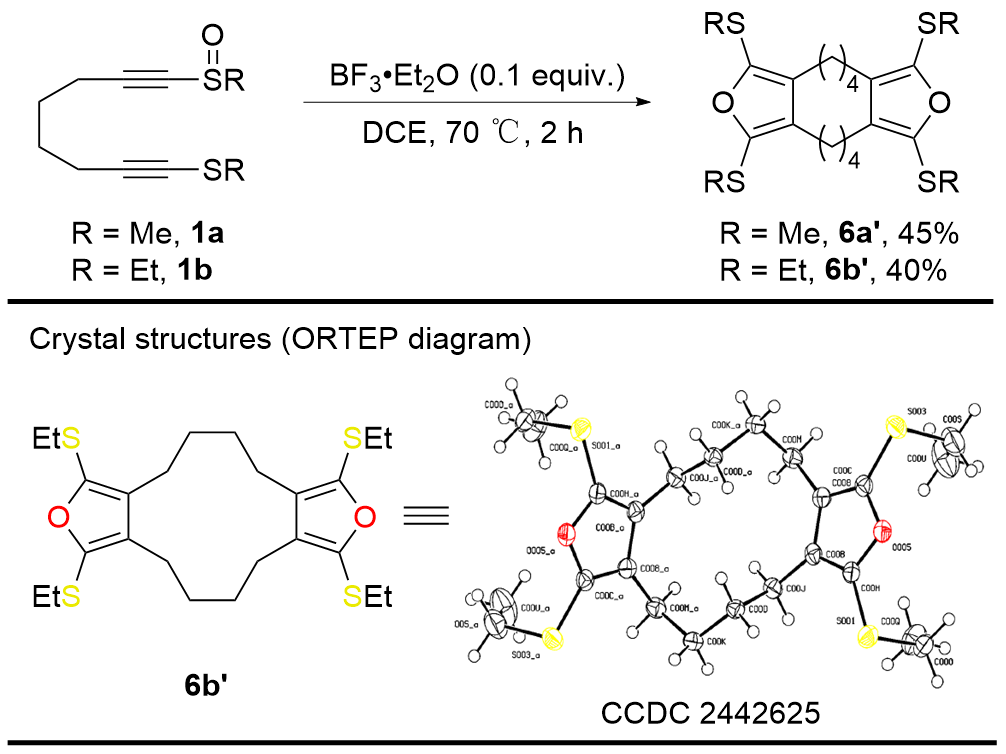
报道了一种基于分子内串联重排反应的双环呋喃类化合物的合成策略. 该串联反应由三氟化硼乙醚络合物催化的炔基硫醚与炔基亚砜的分子内交叉偶联引发, 随后经历[3,3]-σ硫鎓离子重排及5-exo-dig环化反应, 最终以中等产率以及100%原子经济性获得产物环烷并[c]呋喃. 所得呋喃类化合物的合成应用价值已通过对产物中烷硫基和呋喃环的后修饰反应得到验证.
郭闻涛, 孟书玉, 王全瑞. 炔基亚砜的分子内交叉偶联/[3,3]-硫鎓离子重排策略构建环烷并[c]呋喃[J]. 有机化学, 2026, 46(2): 594-602.
Wentao Guo, Shuyu Meng, Quanrui Wang. Intramolecular Cross-Coupling/[3,3]-Sulfonium Rearrangement of Alkynyl Sulfoxides for the Construction of Cycloalkano[c]furans[J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 594-602.
| Entry | c(1h)/(mol•L-1) | Yieldb/% | |
|---|---|---|---|
| 6a | 6a' | ||
| 1 | 0.2 | Trace | 45 |
| 2 | 0.1 | 18 | 27 |
| 3 | 0.01 | 48 | Trace |
| 4 | 0.005 | 46 | Trace |
| Entry | c(1h)/(mol•L-1) | Yieldb/% | |
|---|---|---|---|
| 6a | 6a' | ||
| 1 | 0.2 | Trace | 45 |
| 2 | 0.1 | 18 | 27 |
| 3 | 0.01 | 48 | Trace |
| 4 | 0.005 | 46 | Trace |
| Entry | Cat. (equiv.) | Solv. | T/℃ | t/h | Conv.b/% |
|---|---|---|---|---|---|
| 1 | BF3•Et2O (0.1) | DCE | 70 | 2 | 50 (48)c |
| 2 | BF3•Et2O (0.1) | DCE | 70 | 6 | 52 |
| 3 | BF3•Et2O (0.1) | DCE | 70 | 12 | 57 |
| 4 | BF3•Et2O (0.1) | DCE | 25 | 6 | 46 |
| 5 | BF3•Et2O (0.1) | DCE | 25 | 12 | 49 |
| 6d | BF3•Et2O (0.1) | DCE | 70 | 12 | 65 (62)c |
| 7d | BF3•Et2O (0.1) | DCE | 70 | 12 | 35 |
| 8d | BF3•Et2O (0.1) | DCE | 70 | 12 | 29 |
| 9d | BF3•Et2O (0.1) | DCE | 70 | 12 | 27 |
| 10d | TfOH (0.1) | DCE | 70 | 12 | 43 |
| 11d | Tf2NH (0.1) | DCE | 70 | 6 | 41 |
| 12d | AgSbF6 (0.1) | DCE | 70 | 6 | 26 |
| 13d | TiCl4 (0.1) | DCE | 70 | 12 | 13 |
| 14d | AuCl3 (0.1) | DCE | 70 | 12 | 18 |
| 15d | SnCl4 (0.1) | DCE | 70 | 12 | 31 |
| 16d | In(OTf)3 (0.1) | DCE | 70 | 12 | 28 |
| 17d | Eu(OTf)3 (0.1) | DCE | 70 | 12 | 35 |
| 18d | SmI2 (0.1) | DCE | 70 | 12 | <5 |
| 19d | BuBOTf (0.1) | DCE | 70 | 12 | 41 |
| 20d | BF3•Et2O (0.1) | Toluene | 70 | 6 | 32 |
| 21d | BF3•Et2O (0.1) | MeCN | 70 | 8 | 41 |
| 22d | BF3•Et2O (0.1) | MeNO2 | 70 | 4 | 27 |
| 23d | BF3•Et2O (0.1) | CCl4 | 70 | 12 | 46 |
| Entry | Cat. (equiv.) | Solv. | T/℃ | t/h | Conv.b/% |
|---|---|---|---|---|---|
| 1 | BF3•Et2O (0.1) | DCE | 70 | 2 | 50 (48)c |
| 2 | BF3•Et2O (0.1) | DCE | 70 | 6 | 52 |
| 3 | BF3•Et2O (0.1) | DCE | 70 | 12 | 57 |
| 4 | BF3•Et2O (0.1) | DCE | 25 | 6 | 46 |
| 5 | BF3•Et2O (0.1) | DCE | 25 | 12 | 49 |
| 6d | BF3•Et2O (0.1) | DCE | 70 | 12 | 65 (62)c |
| 7d | BF3•Et2O (0.1) | DCE | 70 | 12 | 35 |
| 8d | BF3•Et2O (0.1) | DCE | 70 | 12 | 29 |
| 9d | BF3•Et2O (0.1) | DCE | 70 | 12 | 27 |
| 10d | TfOH (0.1) | DCE | 70 | 12 | 43 |
| 11d | Tf2NH (0.1) | DCE | 70 | 6 | 41 |
| 12d | AgSbF6 (0.1) | DCE | 70 | 6 | 26 |
| 13d | TiCl4 (0.1) | DCE | 70 | 12 | 13 |
| 14d | AuCl3 (0.1) | DCE | 70 | 12 | 18 |
| 15d | SnCl4 (0.1) | DCE | 70 | 12 | 31 |
| 16d | In(OTf)3 (0.1) | DCE | 70 | 12 | 28 |
| 17d | Eu(OTf)3 (0.1) | DCE | 70 | 12 | 35 |
| 18d | SmI2 (0.1) | DCE | 70 | 12 | <5 |
| 19d | BuBOTf (0.1) | DCE | 70 | 12 | 41 |
| 20d | BF3•Et2O (0.1) | Toluene | 70 | 6 | 32 |
| 21d | BF3•Et2O (0.1) | MeCN | 70 | 8 | 41 |
| 22d | BF3•Et2O (0.1) | MeNO2 | 70 | 4 | 27 |
| 23d | BF3•Et2O (0.1) | CCl4 | 70 | 12 | 46 |
| [1] |
(a)
doi: 10.1016/S0040-4020(97)10303-9 |
|
(b)
|
|
| [2] |
(a)
doi: 10.1246/cl.1987.1687 pmid: 22766766 |
|
(b)
doi: 10.7164/antibiotics.56.931 pmid: 22766766 |
|
|
(c)
doi: 10.1007/s11010-012-1372-1 pmid: 22766766 |
|
| [3] |
doi: 10.1021/ja8088185 |
| [4] |
(a)
pmid: 10964373 |
|
(b)
pmid: 10964373 |
|
| [5] |
|
| [6] |
(a)
doi: 10.1021/jacs.9b08510 |
|
(b)
|
|
|
(c)
doi: 10.6023/cjoc202400006 |
|
|
(金良, 史炳锋, 有机化学, 2024, 44, 657.)
doi: 10.6023/cjoc202400006 |
|
| [7] |
|
| [8] |
(a)
doi: 10.1002/anie.v51.17 pmid: 22515738 |
|
(b)
doi: 10.1021/ja302868s pmid: 22515738 |
|
| [9] |
(a)
doi: 10.1021/ja310002m |
|
(b)
doi: 10.1002/anie.v51.23 |
|
|
(c)
|
|
| [10] |
(a)
doi: 10.1021/acs.orglett.1c04242 |
|
(b)
doi: 10.6023/cjoc202401016 |
|
|
(孟书玉, 郭闻涛, 王全瑞, 有机化学, 2024, 44, 2274.)
doi: 10.6023/cjoc202401016 |
|
| [11] |
doi: 10.1126/science.aat5883 |
| [12] |
|
| [13] |
|
| [14] |
doi: 10.1016/S0040-4039(01)90298-9 |
| [15] |
|
| [16] |
(a)
doi: 10.1021/ar00064a001 |
|
(b)
|
|
|
(c)
doi: 10.1021/acs.chemrev.0c00793 |
|
| [17] |
doi: 10.1055/s-00000083 |
| [18] |
doi: 10.1016/S0040-4020(97)00747-3 |
| [19] |
(a)
pmid: 12946131 |
|
(b)
doi: 10.1021/jacs.0c12194 pmid: 12946131 |
| [1] | 罗艳, 刘章伟, 胡朝蕾, 闭红艳, 莫冬亮. 十元氮杂环化合物合成最新研究进展[J]. 有机化学, 2026, 46(2): 420-442. |
| [2] | 陈晓培, 王清龙, 侯学会, 王川川, 马志伟, 张京玉. CF3-亚胺酰基亚砜叶立德在环化反应中的应用研究进展[J]. 有机化学, 2025, 45(8): 2796-2814. |
| [3] | 范玉兰, 邹小颖, 朱小青, 郑绿茵, 郭维. N-邻位C(sp3)—H键官能团化合成含氮杂环化合物研究进展[J]. 有机化学, 2025, 45(4): 1047-1096. |
| [4] | 张旭, 邹佩森, 刘小清, 闭红艳, 莫冬亮. 金催化炔烃分子内环化反应及应用研究进展[J]. 有机化学, 2025, 45(10): 3755-3776. |
| [5] | 诸雨楠, 矫伟康, 韦雪白, 陈春华, 莫冬亮. 二芳基碘盐参与的自由基芳基化反应研究进展[J]. 有机化学, 2025, 45(10): 3672-3690. |
| [6] | 董道青, 王怡萌, 朱耕宁, 王艳丽, 于曙光, 刘娟, 冯健新, 徐鑫明, 颜世强, 王祖利. N-(2-乙炔基苯基)丙烯酰胺的环化反应用于杂多环化合物的合成[J]. 有机化学, 2025, 45(10): 3741-3754. |
| [7] | 王文贵, 王守锋. 水溶液中的Minisci反应研究进展[J]. 有机化学, 2024, 44(7): 2136-2146. |
| [8] | 孟书玉, 郭闻涛, 王全瑞. 基于炔基亚砜与炔酰胺/炔醚的交叉偶联/[3,3]-硫鎓离子重排串联策略构筑四取代呋喃[J]. 有机化学, 2024, 44(7): 2274-2285. |
| [9] | 刘雯娟, 陈品红. 钯催化1,6-烯炔的环化反应研究[J]. 有机化学, 2024, 44(7): 2077-2091. |
| [10] | 刘杰, 韩峰, 李双艳, 陈天煜, 陈建辉, 徐清. 无过渡金属参与甲基杂环化合物与醇的选择性有氧烯基化反应[J]. 有机化学, 2024, 44(2): 573-583. |
| [11] | 陈玉琢, 孙红梅, 王亮, 胡方芝, 李帅帅. 基于α-氢迁移策略构建杂环骨架的研究进展[J]. 有机化学, 2023, 43(7): 2323-2337. |
| [12] | 孔德亮, 戴闻, 赵怡玲, 陈艺林, 朱红平. 脒基胺硼基硅宾与单酮和二酮的氧化环加成反应研究[J]. 有机化学, 2023, 43(5): 1843-1851. |
| [13] | 蒙玲, 汪君. 硫代黄烷酮类衍生物的合成研究进展[J]. 有机化学, 2023, 43(3): 873-891. |
| [14] | 段康慧, 唐俊龙, 伍婉卿. 稠杂环化合物的合成及其抗肿瘤活性研究进展[J]. 有机化学, 2023, 43(3): 826-854. |
| [15] | 郝二军, 丁笑波, 王珂新, 周红昊, 杨启亮, 石磊. 氮杂环丙烷与不饱和化合物发生[3+2]扩环反应的研究进展[J]. 有机化学, 2023, 43(12): 4057-4074. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||