有机化学 ›› 2026, Vol. 46 ›› Issue (2): 399-419.DOI: 10.6023/cjoc202507029 上一篇 下一篇
综述与进展
刘小晨a,b,*( ), 雷英a, 唐楷a, 林昳c, 马昌期b,*(
), 雷英a, 唐楷a, 林昳c, 马昌期b,*( )
)
收稿日期:2025-07-21
修回日期:2025-09-26
发布日期:2025-11-05
通讯作者:
刘小晨, 马昌期
基金资助:
Xiaochen Liua,b,*( ), Ying Leia, Kai Tanga, Yi Linc, Changqi Mab,*(
), Ying Leia, Kai Tanga, Yi Linc, Changqi Mab,*( )
)
Received:2025-07-21
Revised:2025-09-26
Published:2025-11-05
Contact:
Xiaochen Liu, Changqi Ma
Supported by:文章分享
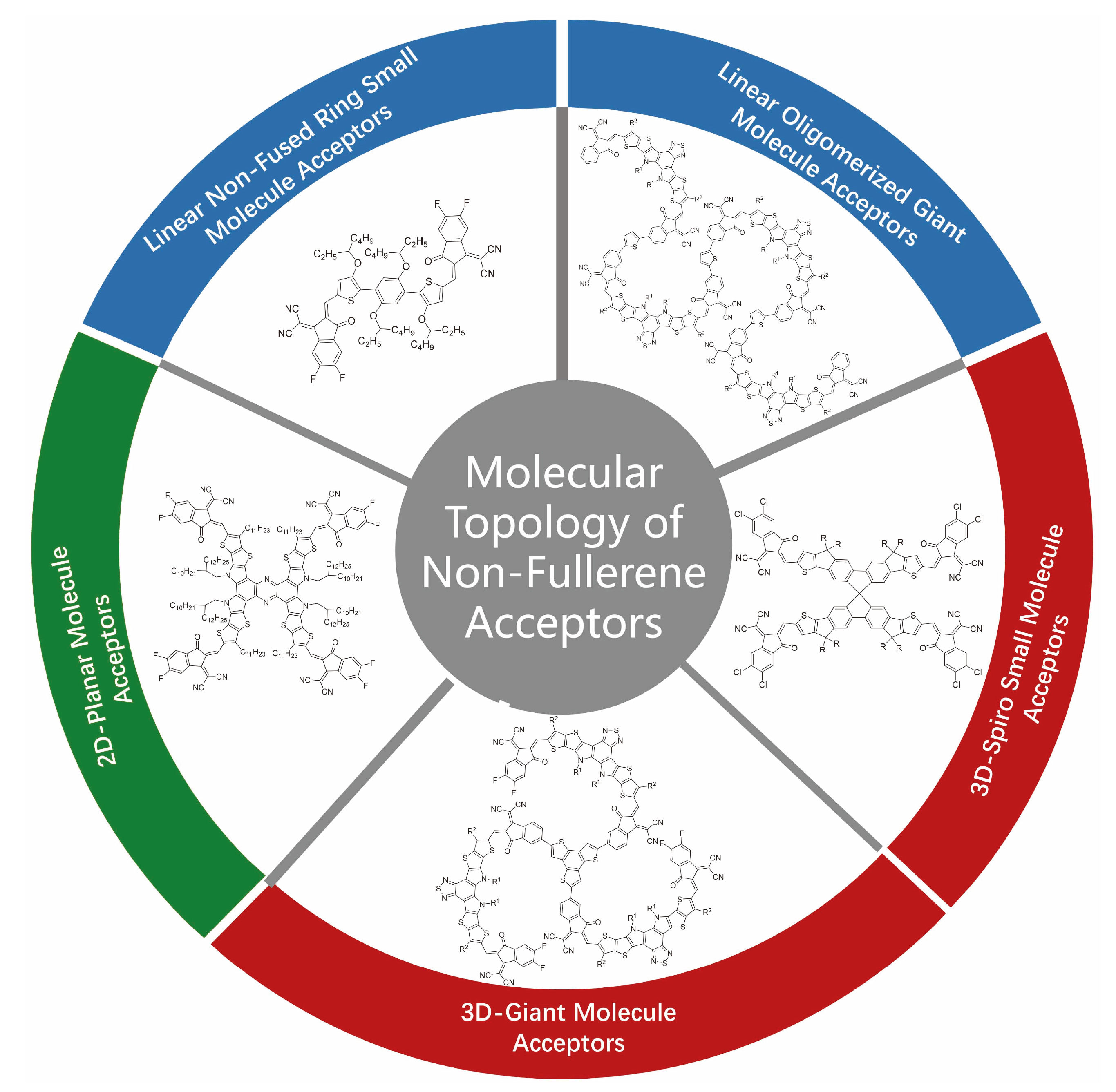
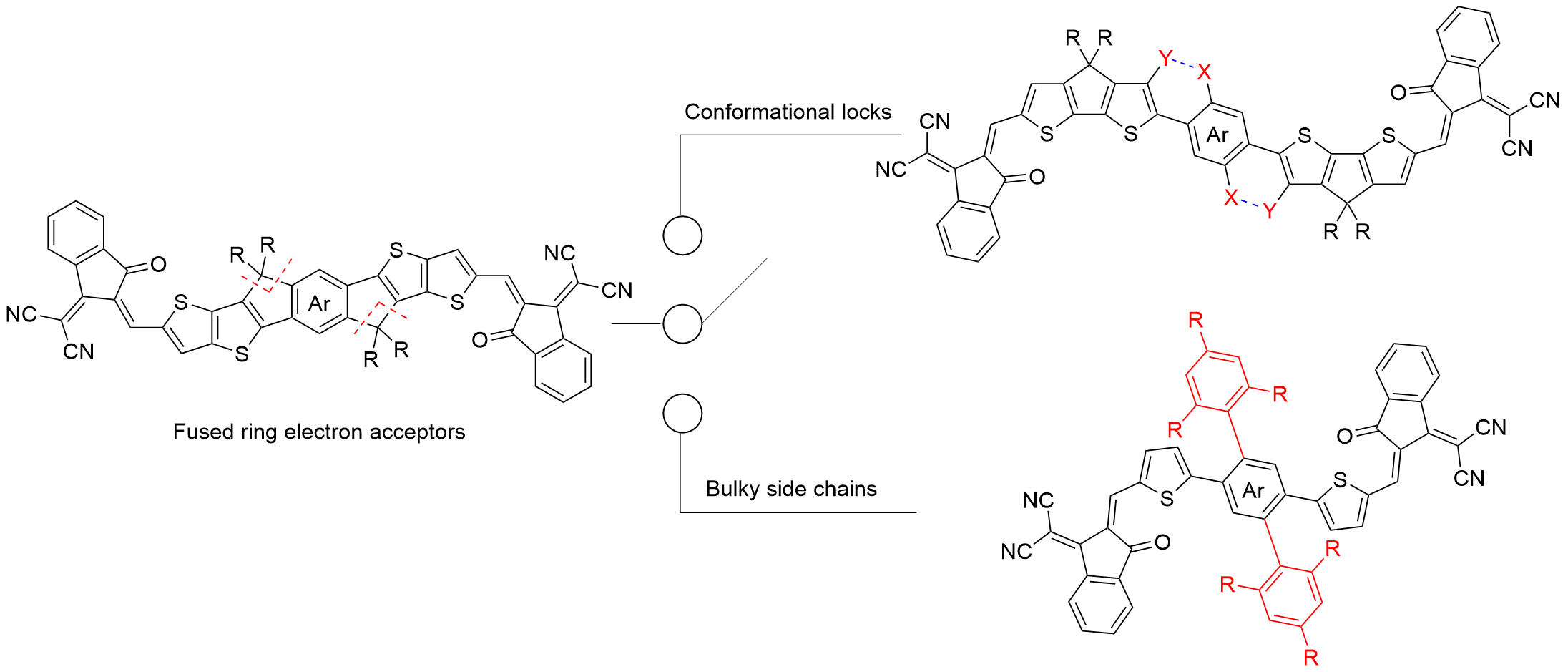
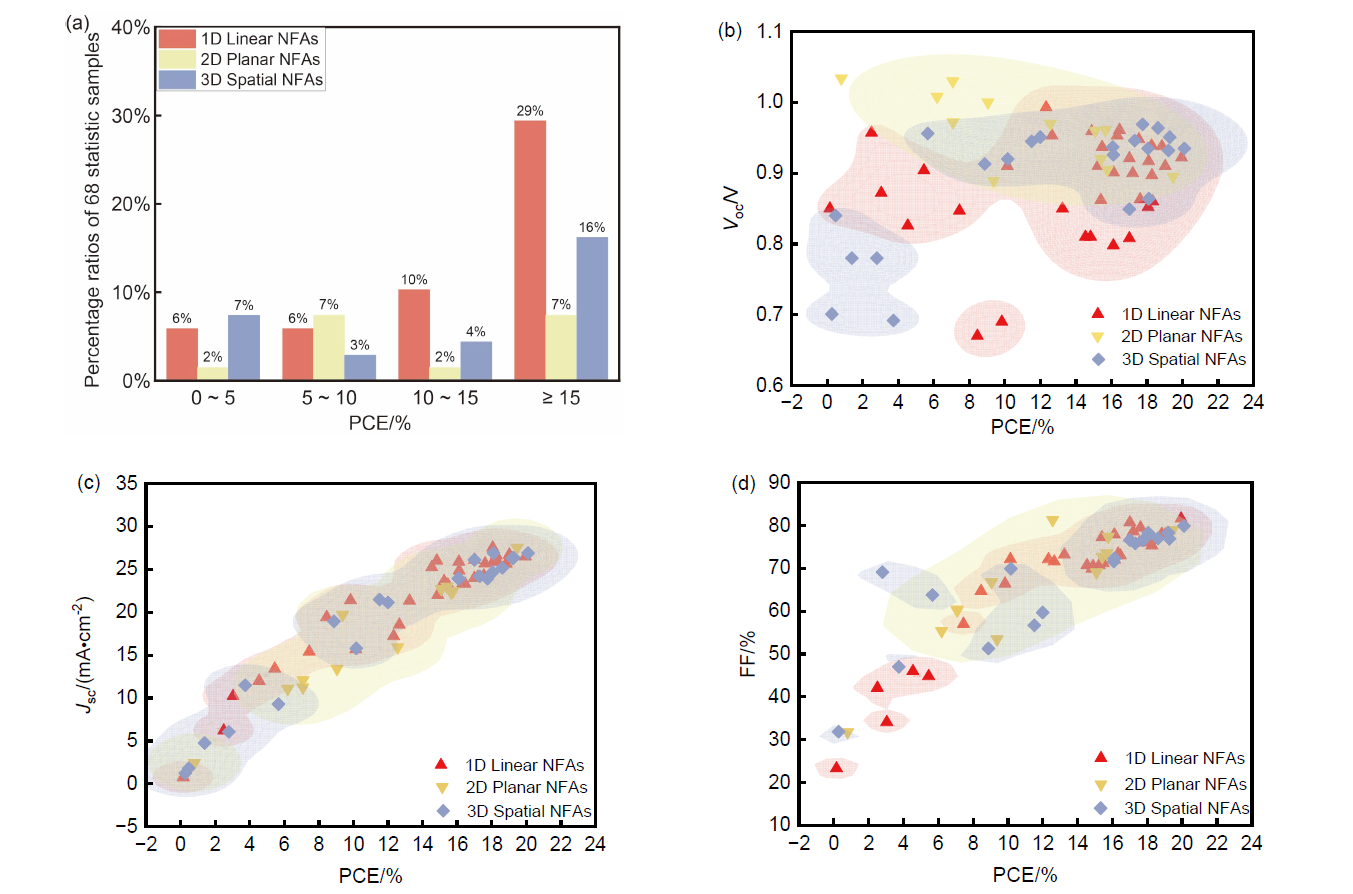
非富勒烯受体(NFAs)是有机太阳能电池活性层核心材料, 其分子结构空间拓扑创新推动器件效率与稳定性突破. 发展遵循问题驱动-结构优化-性能提升的科学逻辑. 早期苝二酰亚胺体系验证了非富勒烯受体可行性, 但刚性平面引发过度聚集, 限制载流子传输, 制约器件性能. 后续以茚并二噻吩[3,2-b]并噻吩(IDTT)为核心的3,9-双(2-亚甲基- (3-(1,1-二氰基亚甲基)-茚酮))-5,5,11,11-四(4-己基苯基)-二噻吩并[2,3-d:2',3'-d']-s-茚并[1,2-b:5,6-b']二噻吩(ITIC)系列采用A-D-A线性构型, 借刚性稠环抑制过度聚集, 首次将单结器件效率推至10%以上, 确立了以梯形稠环为核的设计思路. 双噻吩并[2'',3'':4',5']噻吩并[2',3':4,5]吡咯[3,2-e:2',3'-g][2,1,3]苯并噻二唑(BTP)为核的Y系列受体通过引入缺电子核和构建“C”型骨架拓宽光吸收并提升与给体相容性, 突破20%效率瓶颈并成为高效体系. 此后拓扑研究向多维拓展, 形成一维线性二维平面三维空间三类结构. 一维聚焦骨架优化与提升分子的刚性缓解电压损失; 二维借助共轭扩展增强π电子离域, 需进一步增强分子平面性并优化相分离; 三维通过立体空间的区域调控抑制过度聚集并促进电荷的各向同性传输. 梳理了三类受体特征设计及进展, 揭示拓扑对性能的调控机制, 展望了未来设计方向, 为有机太阳能电池商业化提供参考.
刘小晨, 雷英, 唐楷, 林昳, 马昌期. 有机太阳能电池非富勒烯受体的分子结构空间拓扑研究进展[J]. 有机化学, 2026, 46(2): 399-419.
Xiaochen Liu, Ying Lei, Kai Tang, Yi Lin, Changqi Ma. Progress in Molecular Structure Topology of Non-Fullerene Acceptors for Organic Solar Cells[J]. Chinese Journal of Organic Chemistry, 2026, 46(2): 399-419.
| NFAs | Donor | HOMO/LUMO/eV | Voc/V | Jsc/(mA•cm-2) | FF/% | PCE/% | Ref. |
|---|---|---|---|---|---|---|---|
| BTT-FIC | PM6 | -3.87/-5.64 | 0.953 | 18.54 | 71.6 | 12.65 | [ |
| MC7F3 | PM1 | -4.06/-5.69 | 0.863 | 25.68 | 79.5 | 17.61 | [ |
| M34 | PM6 | -3.90/-5.61 | 0.910 | 23.63 | 70.7 | 15.24 | [ |
| SB16 | PM6 | -4.11/-5.54 | 0.850 | 0.740 | 23.3 | 0.15 | [ |
| CB16 | PM6 | -3.97/-5.73 | 0.860 | 25.98 | 76.9 | 18.32 | [ |
| S-F | D18 | -3.87/-5.78 | 0.862 | 23.20 | 77.2 | 15.40 | [ |
| C-F | D18 | -3.86/-5.78 | 0.921 | 24.00 | 77.1 | 17.00 | [ |
| S-Cl46-Cl | D18 | -4.01/-5.68 | 0.872 | 10.23 | 34.1 | 3.04 | [ |
| M-Cl46-Cl | D18 | -3.97/-5.85 | 0.904 | 13.43 | 44.8 | 5.44 | [ |
| ?-Cl46-Cl | D18 | -3.95/-5.91 | 0.993 | 17.21 | 72.1 | 12.32 | [ |
| C-Cl46-Cl | D18 | -3.99/-5.79 | 0.922 | 26.50 | 81.6 | 19.94 | [ |
| DF-PCIC | PBDB-T | -5.49/-3.77 | 0.910 | 15.66 | 72.1 | 10.14 | [ |
| DOC2C6-2F | PBDB-T | -5.49/-3.83 | 0.850 | 21.35 | 73.2 | 13.24 | [ |
| NoCA-5 | J52 | -5.43/-3.83 | 0.810 | 26.02 | 69.9 | 14.82 | [ |
| TCOR2 | P1 | -5.59/-4.02 | 0.870 | 24.22 | 72.0 | 15.17 | [ |
| BT-IC4F | PBDB-T | -5.89/-4.27 | 0.690 | 21.40 | 66.4 | 9.83 | [ |
| BT2F-IC4F | PBDB-T | -5.98/-4.31 | 0.670 | 19.43 | 64.7 | 8.45 | [ |
| TBT-10 | PBQx-TF | -5.68/-3.91 | 0.826 | 12.00 | 46.0 | 4.54 | [ |
| TBT-11 | PBQx-TF | -5.62/-3.93 | 0.847 | 15.40 | 57.0 | 7.44 | [ |
| TBT-13 | PBQx-TF | -5.49/-3.97 | 0.798 | 25.90 | 77.9 | 16.10 | [ |
| TBT-26 | BBQx-TF | -5.54/-4.16 | 0.808 | 26.10 | 80.7 | 17.00 | [ |
| 2BTh-2F-C2 | D18 | -5.55/-3.98 | 0.910 | 26.71 | 77.9 | 19.02 | [ |
| BM-2F | J52 | -5.39/-3.99 | 0.810 | 25.25 | 70.7 | 14.53 | [ |
| 3TT-C2-F | D18 | -5.56/-3.95 | 0.900 | 24.13 | 78.6 | 17.19 | [ |
| EV-i | PM6 | -5.77/-3.91 | 0.897 | 26.60 | 76.6 | 18.27 | [ |
| EV-o | PM6 | -5.80/-3.71 | 0.957 | 6.20 | 42.1 | 2.50 | [ |
| DYA-I | D18 | -5.51/-3.99 | 0.938 | 25.67 | 78.00 | 18.83 | [ |
| DYA-O | D18 | -5.47/-3.93 | 0.948 | 24.29 | 76.00 | 17.54 | [ |
| DYA-IO | D18 | -5.49/-3.96 | 0.961 | 23.32 | 73.00 | 16.45 | [ |
| DIBP3F-S | PM6 | -6.03/-4.29 | 0.901 | 24.86 | 72.00 | 16.11 | [ |
| DIPB-3F-Se | PM6 | -6.00/-4.27 | 0.917 | 25.92 | 76.10 | 18.09 | [ |
| DOY-TVT | D18 | -5.80/-3.66 | 0.852 | 27.53 | 77.01 | 18.06 | [ |
| DYF-TF | D18 | -5.59/-3.63 | 0.939 | 25.82 | 75.3 | 18.26 | [ |
| DY | PM6 | -5.66/-3.74 | 0.959 | 22.01 | 70.5 | 14.88 | [ |
| TY | PM6 | -5.64/-3.75 | 0.953 | 23.35 | 73.4 | 16.32 | [ |
| QY | PM6 | -5.64/-3.76 | 0.937 | 23.20 | 71.2 | 15.47 | [ |
| NFAs | Donor | HOMO/LUMO/eV | Voc/V | Jsc/(mA•cm-2) | FF/% | PCE/% | Ref. |
|---|---|---|---|---|---|---|---|
| BTT-FIC | PM6 | -3.87/-5.64 | 0.953 | 18.54 | 71.6 | 12.65 | [ |
| MC7F3 | PM1 | -4.06/-5.69 | 0.863 | 25.68 | 79.5 | 17.61 | [ |
| M34 | PM6 | -3.90/-5.61 | 0.910 | 23.63 | 70.7 | 15.24 | [ |
| SB16 | PM6 | -4.11/-5.54 | 0.850 | 0.740 | 23.3 | 0.15 | [ |
| CB16 | PM6 | -3.97/-5.73 | 0.860 | 25.98 | 76.9 | 18.32 | [ |
| S-F | D18 | -3.87/-5.78 | 0.862 | 23.20 | 77.2 | 15.40 | [ |
| C-F | D18 | -3.86/-5.78 | 0.921 | 24.00 | 77.1 | 17.00 | [ |
| S-Cl46-Cl | D18 | -4.01/-5.68 | 0.872 | 10.23 | 34.1 | 3.04 | [ |
| M-Cl46-Cl | D18 | -3.97/-5.85 | 0.904 | 13.43 | 44.8 | 5.44 | [ |
| ?-Cl46-Cl | D18 | -3.95/-5.91 | 0.993 | 17.21 | 72.1 | 12.32 | [ |
| C-Cl46-Cl | D18 | -3.99/-5.79 | 0.922 | 26.50 | 81.6 | 19.94 | [ |
| DF-PCIC | PBDB-T | -5.49/-3.77 | 0.910 | 15.66 | 72.1 | 10.14 | [ |
| DOC2C6-2F | PBDB-T | -5.49/-3.83 | 0.850 | 21.35 | 73.2 | 13.24 | [ |
| NoCA-5 | J52 | -5.43/-3.83 | 0.810 | 26.02 | 69.9 | 14.82 | [ |
| TCOR2 | P1 | -5.59/-4.02 | 0.870 | 24.22 | 72.0 | 15.17 | [ |
| BT-IC4F | PBDB-T | -5.89/-4.27 | 0.690 | 21.40 | 66.4 | 9.83 | [ |
| BT2F-IC4F | PBDB-T | -5.98/-4.31 | 0.670 | 19.43 | 64.7 | 8.45 | [ |
| TBT-10 | PBQx-TF | -5.68/-3.91 | 0.826 | 12.00 | 46.0 | 4.54 | [ |
| TBT-11 | PBQx-TF | -5.62/-3.93 | 0.847 | 15.40 | 57.0 | 7.44 | [ |
| TBT-13 | PBQx-TF | -5.49/-3.97 | 0.798 | 25.90 | 77.9 | 16.10 | [ |
| TBT-26 | BBQx-TF | -5.54/-4.16 | 0.808 | 26.10 | 80.7 | 17.00 | [ |
| 2BTh-2F-C2 | D18 | -5.55/-3.98 | 0.910 | 26.71 | 77.9 | 19.02 | [ |
| BM-2F | J52 | -5.39/-3.99 | 0.810 | 25.25 | 70.7 | 14.53 | [ |
| 3TT-C2-F | D18 | -5.56/-3.95 | 0.900 | 24.13 | 78.6 | 17.19 | [ |
| EV-i | PM6 | -5.77/-3.91 | 0.897 | 26.60 | 76.6 | 18.27 | [ |
| EV-o | PM6 | -5.80/-3.71 | 0.957 | 6.20 | 42.1 | 2.50 | [ |
| DYA-I | D18 | -5.51/-3.99 | 0.938 | 25.67 | 78.00 | 18.83 | [ |
| DYA-O | D18 | -5.47/-3.93 | 0.948 | 24.29 | 76.00 | 17.54 | [ |
| DYA-IO | D18 | -5.49/-3.96 | 0.961 | 23.32 | 73.00 | 16.45 | [ |
| DIBP3F-S | PM6 | -6.03/-4.29 | 0.901 | 24.86 | 72.00 | 16.11 | [ |
| DIPB-3F-Se | PM6 | -6.00/-4.27 | 0.917 | 25.92 | 76.10 | 18.09 | [ |
| DOY-TVT | D18 | -5.80/-3.66 | 0.852 | 27.53 | 77.01 | 18.06 | [ |
| DYF-TF | D18 | -5.59/-3.63 | 0.939 | 25.82 | 75.3 | 18.26 | [ |
| DY | PM6 | -5.66/-3.74 | 0.959 | 22.01 | 70.5 | 14.88 | [ |
| TY | PM6 | -5.64/-3.75 | 0.953 | 23.35 | 73.4 | 16.32 | [ |
| QY | PM6 | -5.64/-3.76 | 0.937 | 23.20 | 71.2 | 15.47 | [ |
| NFAs | Donor | HOMO/LUMO/eV | Voc/V | Jsc/(mA•cm-2) | FF/% | PCE/% | Ref. |
|---|---|---|---|---|---|---|---|
| F-2PDI | PBDB-T | -5.52/-3.76 | 1.030 | 11.23 | 60.4 | 7.07 | [ |
| F-2PDI-4F | PBDB-T | -5.55/-3.81 | 1.000 | 13.42 | 66.8 | 9.05 | [ |
| anti-PDFC | PM6 | -5.57/-3.85 | 0.970 | 15.93 | 81.3 | 12.56 | [ |
| anti-PDFC | BTR-Cl | -5.57/-3.84 | 0.972 | 12.09 | 60.2 | 7.08 | [ |
| syn-PDFC | BTR-Cl | -5.58/-3.83 | 1.008 | 11.09 | 55.4 | 6.19 | [ |
| PDFC-Ph | BTR-Cl | -5.61/-3.93 | 1.034 | 2.43 | 31.8 | 0.798 | [ |
| CH8 | PM6 | -5.64/-3.75 | 0.889 | 19.70 | 53.5 | 9.37 | [ |
| 4A-DFIC | D18-Cl | -5.72/-4.03 | 0.905 | 22.47 | 77.4 | 15.76 | [ |
| DP-BTP | D18 | -5.67/-3.92 | 0.960 | 22.73 | 69.1 | 15.08 | [ |
| QD-1 | PM6 | -5.73/-3.80 | 0.895 | 27.52 | 79.0 | 19.46 | [ |
| DT-6IC | D18 | -5.65/-3.90 | 0.961 | 22.31 | 73.5 | 15.66 | [ |
| CH26 | PM6 | -3.59/-5.28 | 0.920 | 22.98 | 72.7 | 15.41 | [ |
| NFAs | Donor | HOMO/LUMO/eV | Voc/V | Jsc/(mA•cm-2) | FF/% | PCE/% | Ref. |
|---|---|---|---|---|---|---|---|
| F-2PDI | PBDB-T | -5.52/-3.76 | 1.030 | 11.23 | 60.4 | 7.07 | [ |
| F-2PDI-4F | PBDB-T | -5.55/-3.81 | 1.000 | 13.42 | 66.8 | 9.05 | [ |
| anti-PDFC | PM6 | -5.57/-3.85 | 0.970 | 15.93 | 81.3 | 12.56 | [ |
| anti-PDFC | BTR-Cl | -5.57/-3.84 | 0.972 | 12.09 | 60.2 | 7.08 | [ |
| syn-PDFC | BTR-Cl | -5.58/-3.83 | 1.008 | 11.09 | 55.4 | 6.19 | [ |
| PDFC-Ph | BTR-Cl | -5.61/-3.93 | 1.034 | 2.43 | 31.8 | 0.798 | [ |
| CH8 | PM6 | -5.64/-3.75 | 0.889 | 19.70 | 53.5 | 9.37 | [ |
| 4A-DFIC | D18-Cl | -5.72/-4.03 | 0.905 | 22.47 | 77.4 | 15.76 | [ |
| DP-BTP | D18 | -5.67/-3.92 | 0.960 | 22.73 | 69.1 | 15.08 | [ |
| QD-1 | PM6 | -5.73/-3.80 | 0.895 | 27.52 | 79.0 | 19.46 | [ |
| DT-6IC | D18 | -5.65/-3.90 | 0.961 | 22.31 | 73.5 | 15.66 | [ |
| CH26 | PM6 | -3.59/-5.28 | 0.920 | 22.98 | 72.7 | 15.41 | [ |
| NFAs | Donor | HOMO/LUMO (eV) | Voc/V | Jsc/(mA•cm-2) | FF/% | PCE/% | Ref. |
|---|---|---|---|---|---|---|---|
| SFTTIC | PBDB-T | -5.46/-3.65 | 0.956 | 9.28 | 63.8 | 5.66 | [ |
| SFIC-Cl | PBDB-T | -5.85/-4.07 | 0.920 | 15.79 | 69.9 | 10.16 | [ |
| SCT-(TID)4 | PTB7-Th | -5.40/-3.93 | 0.701 | 1.21 | 31.9 | 0.27 | [ |
| SCT-(TIC)4 | PTB7-Th | -5.45/-4.04 | 0.780 | 6.06 | 69.1 | 2.79 | [ |
| SCT-(TFIC)4 | PM6 | -5.57/-4.18 | 0.692 | 11.49 | 47.0 | 3.73 | [ |
| DSOCT-(TIC)6 | PM6 | -5.58/-4.12 | 0.84 | 1.82 | 0.31 | 0.47 | [ |
| DSOCT-(TFIC)6 | PM6 | -5.67/-4.27 | 0.78 | 4.75 | 0.48 | 1.39 | [ |
| GTs | PM6 | -5.78/-3.95 | 0.935 | 24.66 | 78.3 | 18.05 | [ |
| TYT-S | D18 | -5.66/-4.24 | 0.964 | 25.18 | 77.0 | 18.61 | [ |
| T-Qx-15Cl | PM6 | -5.56/-3.86 | 0.935 | 26.90 | 79.9 | 20.10 | [ |
| T-Qx-15F | PM6 | -5.54/-3.84 | 0.946 | 24.20 | 75.8 | 17.30 | [ |
| T-Qx-12Cl3F | PM6 | -5.57/-3.86 | 0.932 | 26.30 | 78.3 | 19.20 | [ |
| 3BY | PM6 | -5.65/-3.80 | 0.969 | 23.92 | 76.6 | 17.75 | [ |
| 3QY | PM6 | -5.67/-3.83 | 0.951 | 26.36 | 76.9 | 19.27 | [ |
| SP1-Ph | PM6 | -5.74/-3.89 | 0.951 | 21.13 | 59.7 | 11.99 | [ |
| SP2-Ph | PM6 | -5.78/-3.89 | 0.945 | 21.47 | 56.7 | 11.51 | [ |
| SP3-Ph | PM6 | -5.73/-3.89 | 0.937 | 23.95 | 71.6 | 16.07 | [ |
| SP4-Ph | PM6 | -5.74/-3.91 | 0.926 | 23.90 | 72.3 | 16.10 | [ |
| SP6-Ph | PM6 | -5.71/-3.89 | 0.913 | 18.95 | 51.3 | 8.87 | [ |
| TDY-α | PM6 | -5.69/-3.92 | 0.864 | 26.90 | 78.0 | 18.10 | [ |
| TDY-β | PM6 | -5.76/-3.96 | 0.849 | 26.10 | 76.6 | 17.00 | [ |
| NFAs | Donor | HOMO/LUMO (eV) | Voc/V | Jsc/(mA•cm-2) | FF/% | PCE/% | Ref. |
|---|---|---|---|---|---|---|---|
| SFTTIC | PBDB-T | -5.46/-3.65 | 0.956 | 9.28 | 63.8 | 5.66 | [ |
| SFIC-Cl | PBDB-T | -5.85/-4.07 | 0.920 | 15.79 | 69.9 | 10.16 | [ |
| SCT-(TID)4 | PTB7-Th | -5.40/-3.93 | 0.701 | 1.21 | 31.9 | 0.27 | [ |
| SCT-(TIC)4 | PTB7-Th | -5.45/-4.04 | 0.780 | 6.06 | 69.1 | 2.79 | [ |
| SCT-(TFIC)4 | PM6 | -5.57/-4.18 | 0.692 | 11.49 | 47.0 | 3.73 | [ |
| DSOCT-(TIC)6 | PM6 | -5.58/-4.12 | 0.84 | 1.82 | 0.31 | 0.47 | [ |
| DSOCT-(TFIC)6 | PM6 | -5.67/-4.27 | 0.78 | 4.75 | 0.48 | 1.39 | [ |
| GTs | PM6 | -5.78/-3.95 | 0.935 | 24.66 | 78.3 | 18.05 | [ |
| TYT-S | D18 | -5.66/-4.24 | 0.964 | 25.18 | 77.0 | 18.61 | [ |
| T-Qx-15Cl | PM6 | -5.56/-3.86 | 0.935 | 26.90 | 79.9 | 20.10 | [ |
| T-Qx-15F | PM6 | -5.54/-3.84 | 0.946 | 24.20 | 75.8 | 17.30 | [ |
| T-Qx-12Cl3F | PM6 | -5.57/-3.86 | 0.932 | 26.30 | 78.3 | 19.20 | [ |
| 3BY | PM6 | -5.65/-3.80 | 0.969 | 23.92 | 76.6 | 17.75 | [ |
| 3QY | PM6 | -5.67/-3.83 | 0.951 | 26.36 | 76.9 | 19.27 | [ |
| SP1-Ph | PM6 | -5.74/-3.89 | 0.951 | 21.13 | 59.7 | 11.99 | [ |
| SP2-Ph | PM6 | -5.78/-3.89 | 0.945 | 21.47 | 56.7 | 11.51 | [ |
| SP3-Ph | PM6 | -5.73/-3.89 | 0.937 | 23.95 | 71.6 | 16.07 | [ |
| SP4-Ph | PM6 | -5.74/-3.91 | 0.926 | 23.90 | 72.3 | 16.10 | [ |
| SP6-Ph | PM6 | -5.71/-3.89 | 0.913 | 18.95 | 51.3 | 8.87 | [ |
| TDY-α | PM6 | -5.69/-3.92 | 0.864 | 26.90 | 78.0 | 18.10 | [ |
| TDY-β | PM6 | -5.76/-3.96 | 0.849 | 26.10 | 76.6 | 17.00 | [ |
| [1] |
doi: 10.1021/acsaem.1c01737 |
| [2] |
doi: 10.1038/s42254-024-00719-y |
| [3] |
doi: 10.1016/j.cej.2024.154944 |
| [4] |
doi: 10.1126/science.258.5087.1474 pmid: 17755110 |
| [5] |
doi: 10.1126/science.270.5243.1789 |
| [6] |
doi: 10.1016/j.solmat.2016.11.024 |
| [7] |
doi: 10.1002/adfm.v24.1 |
| [63] |
|
| [64] |
doi: 10.31635/ccschem.023.202202575 |
| [65] |
doi: 10.1038/s41467-023-43846-3 |
| [66] |
doi: 10.1039/D0TC01707H |
| [8] |
doi: 10.1002/admi.v2.7 |
| [9] |
doi: 10.1002/adfm.v22.19 |
| [10] |
doi: 10.1002/adma.v27.10 |
| [11] |
doi: 10.1002/adma.v25.32 |
| [12] |
doi: 10.1002/adma.v25.40 |
| [13] |
doi: 10.1021/jacs.6b04368 |
| [67] |
doi: 10.1039/D0CC04297H |
| [68] |
doi: 10.1002/adfm.v31.24 |
| [69] |
doi: 10.1007/s10118-022-2730-4 |
| [70] |
|
| [71] |
|
| [14] |
doi: 10.1021/cm504140c |
| [15] |
doi: 10.1021/jacs.5b00305 |
| [16] |
doi: 10.1021/acsami.5b05248 |
| [17] |
doi: 10.1021/jacs.6b03495 |
| [18] |
doi: 10.1002/adma.v27.7 |
| [72] |
doi: 10.1039/D5EE00878F |
| [73] |
|
| [74] |
doi: 10.31635/ccschem.024.202404023 |
| [75] |
doi: 10.1002/cjoc.v35.11 |
| [76] |
doi: 10.1021/acs.joc.1c02848 |
| [19] |
doi: 10.1016/j.joule.2019.01.004 |
| [20] |
doi: 10.1038/s41560-021-00820-x |
| [21] |
|
| [22] |
doi: 10.1038/s41467-025-60214-5 |
| [77] |
doi: 10.1021/acsomega.4c07409 |
| [78] |
doi: 10.1002/adma.v33.12 |
| [79] |
doi: 10.1002/adfm.v34.51 |
| [80] |
doi: 10.1002/aenm.v14.8 |
| [81] |
doi: 10.1039/D4EE04149F |
| [82] |
|
| [83] |
doi: 10.1002/adfm.v35.16 |
| [84] |
doi: 10.1038/s41467-023-38673-5 pmid: 37217503 |
| [85] |
doi: 10.3390/molecules22030464 |
| [86] |
doi: 10.1002/cphc.v18.21 |
| [23] |
doi: 10.1038/s41563-024-02062-0 |
| [24] |
doi: 10.1002/adfm.v33.14 |
| [25] |
doi: 10.1002/sus2.v2.5 |
| [26] |
|
|
(刘欢, 史慧敏, 李海阳, 詹传郎, 化学进展, 2025, 37, 235.)
doi: 10.7536/PC240416 |
|
| [27] |
|
| [28] |
doi: 10.1039/D0TA10901K |
| [29] |
doi: 10.1039/C6RA12396A |
| [30] |
doi: 10.1002/advs.v7.7 |
| [31] |
doi: 10.1021/jacs.6b02004 |
| [32] |
doi: 10.1039/c9ta01888c |
| [33] |
doi: 10.1016/j.chempr.2024.06.005 |
| [34] |
doi: 10.1002/anie.v59.48 |
| [35] |
|
| [36] |
doi: 10.1021/acsenergylett.4c00043 |
| [37] |
|
| [38] |
doi: 10.1038/s41467-022-30927-y |
| [39] |
doi: 10.1016/j.joule.2019.09.010 |
| [40] |
doi: 10.1002/adma.v30.6 |
| [41] |
doi: 10.1039/C8TA03753A |
| [42] |
doi: 10.1021/jacs.8b12126 |
| [43] |
|
| [44] |
|
| [45] |
doi: 10.1021/acs.chemmater.0c00097 |
| [46] |
doi: 10.1021/jacs.4c00090 |
| [47] |
|
| [48] |
doi: 10.1021/ja303401s pmid: 22679903 |
| [49] |
doi: 10.1021/acs.jpclett.6b01695 |
| [50] |
doi: 10.1038/s41467-019-11001-6 pmid: 31292441 |
| [51] |
doi: 10.1002/anie.v60.32 |
| [52] |
doi: 10.1039/D2TC02289C |
| [53] |
doi: 10.1039/D0TA03683H |
| [54] |
|
| [55] |
doi: 10.1002/cjoc.v41.6 |
| [56] |
doi: 10.1002/adma.v36.4 |
| [57] |
doi: 10.1016/j.giant.2024.100336 |
| [58] |
|
| [59] |
doi: 10.1021/acsami.4c12252 |
| [60] |
|
| [61] |
doi: 10.1002/aenm.v13.34 |
| [62] |
|
| [1] | 崔心怡, 郭丽帆, 马聪璇, 李耘, 梁建华. 抗耐药菌红霉素的结构修饰策略、构效关系及全合成研究进展[J]. 有机化学, 2026, 46(1): 39-73. |
| [2] | 洪赛, 尹发红, 陈明慧, 傅滨, 肖玉梅, 覃兆海. 含吡啶环的1-唑基-1,1-二芳基甲烷类化合物的合成及杀菌活性研究[J]. 有机化学, 2026, 46(1): 106-117. |
| [3] | 赵友学, 李兮若, 孟洛冰, 李春秀, 范贵生, 许建和. 醇脱氢酶/羰基还原酶与多底物分子适配性研究的进展★[J]. 有机化学, 2025, 45(9): 3175-3185. |
| [4] | 郭浩哲, 李玉银, 汤培琛, 樊江莉. 机器学习设计有机荧光诊疗分子的研究进展★[J]. 有机化学, 2025, 45(9): 3203-3212. |
| [5] | 赵瑜, 张紫越, 许书伟, 胡华伟, 吴宏伟, 张琳萍, 陈丰坤. 锯齿形给体-受体齐聚物的设计、合成及光伏性能研究[J]. 有机化学, 2025, 45(11): 4210-4219. |
| [6] | 孙翠萍, 薛雨婷, 陈磅宽. 杂螺烯圆偏振发光材料的研究进展[J]. 有机化学, 2025, 45(11): 4048-4069. |
| [7] | 田海平, 刘东东, 裴鸿艳, 叶家麟, 郑子锐, 高一星, 李昌兴, 田欢, 张静, 张立新. 新型苯基吡唑类衍生物的设计、合成和杀虫活性研究[J]. 有机化学, 2025, 45(1): 227-239. |
| [8] | 秦丽清, 林桂汕, 段文贵, 崔玉成, 杨卯芳, 李芳耀, 李典鹏. 新型长叶烯基萘满并N-酰基吡唑化合物的合成、抗增殖活性、三维定量构效关系及分子对接研究[J]. 有机化学, 2024, 44(6): 1967-1977. |
| [9] | 刘吉永, 吴明慧, 相君成, 庞怀林, 李斌, 吕亮. 新型含(卤代)烷氧基类双酰胺化合物的合成、杀虫活性及构效关系研究[J]. 有机化学, 2024, 44(5): 1584-1591. |
| [10] | 涂开槐, 江雪, 段泰男, 肖泽云. 高性能有机小分子光伏给体材料研究进展[J]. 有机化学, 2024, 44(11): 3282-3298. |
| [11] | 杨家强, 吴学姣, 卢子聪, 陈阳密, 佘慧娴, 刘海军. 含噻唑片段的蛇床子素衍生物的设计、合成及抗菌活性[J]. 有机化学, 2024, 44(11): 3541-3549. |
| [12] | 霍海波, 李桂霞, 王世军, 韩春, 师宝君, 李健. 新型γ-咔啉衍生物的合成及其抑菌活性研究[J]. 有机化学, 2024, 44(1): 204-215. |
| [13] | 左鑫, 许诗诺, 陈忠洋, 鄢剑锋, 袁耀锋. 茂铁类单分子结电子传输性质的研究进展[J]. 有机化学, 2023, 43(7): 2313-2322. |
| [14] | 何金燕, 田富云, 吴青青, 郑月明, 陈玉婷, 许海燕, 金正盛, 詹丽, 程新强, 顾跃玲, 高召兵, 赵桂龙. 基于[3.3.3]螺桨烷的电压门控钙离子通道α2δ亚基配体的合成和生物活性研究[J]. 有机化学, 2023, 43(6): 2226-2238. |
| [15] | 刘兴周, 于明加, 梁建华. 原小檗碱骨架的合成及其抗炎活性研究进展[J]. 有机化学, 2023, 43(4): 1325-1340. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||