有机化学 ›› 2026, Vol. 46 ›› Issue (4): 1739-1749.DOI: 10.6023/cjoc202512005 上一篇 下一篇
研究论文
收稿日期:2025-12-04
修回日期:2026-01-07
发布日期:2026-02-06
通讯作者:
朱晨
基金资助:
Shan Yang, Yasu Chen, Chen Zhu*( )
)
Received:2025-12-04
Revised:2026-01-07
Published:2026-02-06
Contact:
Chen Zhu
Supported by:文章分享
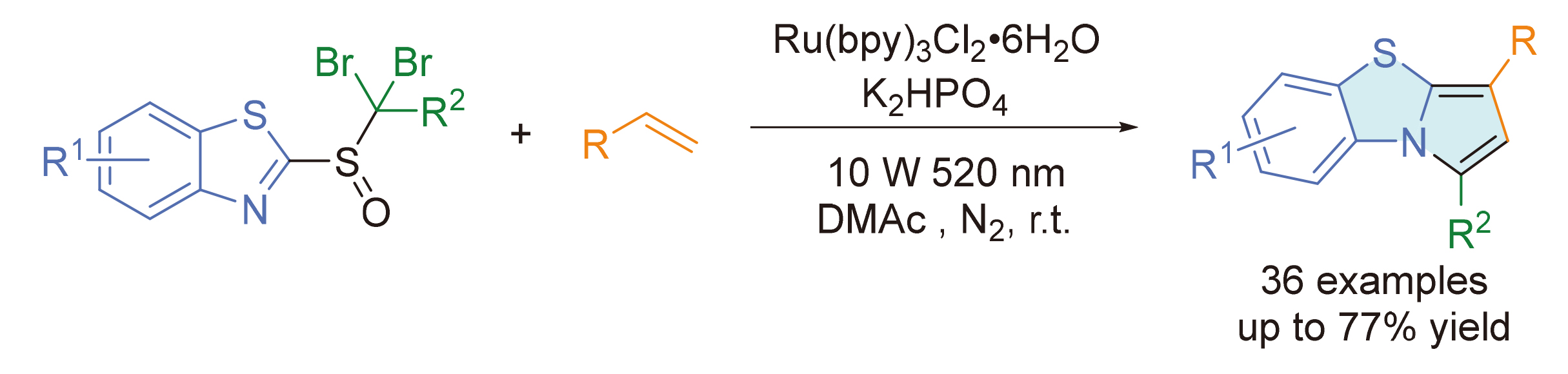
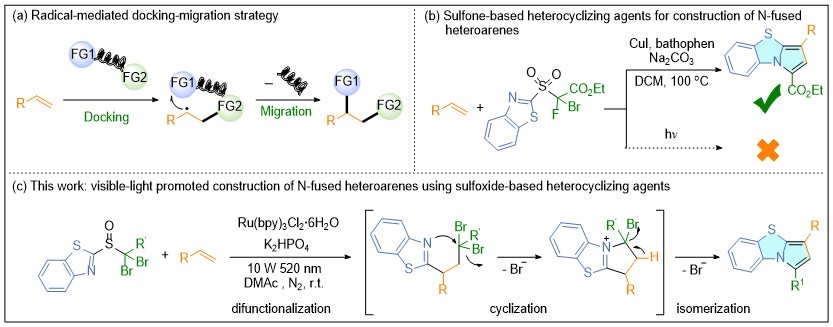
含氮杂环是药物及许多功能分子的核心骨架, 其高效合成始终是有机化学的关键问题. 自由基介导的官能团迁移(FGM)策略为烯烃的双官能团化提供了强大工具. 此文利用官能团迁移-环化串联策略, 发展一种步骤经济且条件温和的含氮稠杂环合成新方法. 基于我们前期对对接-迁移反应研究的积累, 通过对关键连接单元的理性设计, 成功开发了一类新型的基于亚砜的杂环化试剂. 在温和的可见光照射条件下, 该试剂能与多种烯烃发生高效的自由基对接-迁移反应, 并引发分子内的串联环化过程, 一步实现多种含氮稠杂芳烃的模块化构建. 反应可在温和的条件下进行, 底物普适性广、官能团兼容性优异, 为复杂药物活性分子的后期修饰提供了绿色、高效的合成工具.
杨珊, 陈亚苏, 朱晨. 自由基介导的官能团迁移-环化构建含氮稠杂芳烃[J]. 有机化学, 2026, 46(4): 1739-1749.
Shan Yang, Yasu Chen, Chen Zhu. Construction of N-Fused Heteroarenes via Radical-Mediated Functional Group Migration-Cyclization[J]. Chinese Journal of Organic Chemistry, 2026, 46(4): 1739-1749.
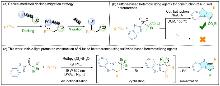
| Entry | Photocatalyst | Base | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | DMF | 47 |
| 2 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | CH3COCH3 | 36 |
| 3 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | CH3CN | 20 |
| 4 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | DCM | 45 |
| 5 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | EtOAc | 49 |
| 6 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | THF | 52 |
| 7 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | DMAc | 56 |
| 8 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | 1,4-Dioxane | 0 |
| 9 | Eosin Y | K2HPO4 | DMAc | 0 |
| 10 | fac-Ir(ppy)₃ | K2HPO4 | DMAc | 47 |
| 11 | Ru(bpy)3Cl2•6H2O | K2HPO4 | DMAc | 64 |
| 12 | Ru(bpy)3Cl2•6H2O | K3PO4 | DMAc | 30 |
| 13 | Ru(bpy)3Cl2•6H2O | Na2HPO4 | DMAc | 52 |
| 14c | Ru(bpy)3Cl2•6H2O | K2HPO4 | DMAc | 73 |
| 15c | None | K2HPO4 | DMAc | 0 |
| 16d | Ru(bpy)3Cl2•6H2O | K2HPO4 | DMAc | 0 |
| Entry | Photocatalyst | Base | Solvent | Yieldb/% |
|---|---|---|---|---|
| 1 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | DMF | 47 |
| 2 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | CH3COCH3 | 36 |
| 3 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | CH3CN | 20 |
| 4 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | DCM | 45 |
| 5 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | EtOAc | 49 |
| 6 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | THF | 52 |
| 7 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | DMAc | 56 |
| 8 | Ir[dF(CF3)ppy]2(dtbbpy)PF6 | K2HPO4 | 1,4-Dioxane | 0 |
| 9 | Eosin Y | K2HPO4 | DMAc | 0 |
| 10 | fac-Ir(ppy)₃ | K2HPO4 | DMAc | 47 |
| 11 | Ru(bpy)3Cl2•6H2O | K2HPO4 | DMAc | 64 |
| 12 | Ru(bpy)3Cl2•6H2O | K3PO4 | DMAc | 30 |
| 13 | Ru(bpy)3Cl2•6H2O | Na2HPO4 | DMAc | 52 |
| 14c | Ru(bpy)3Cl2•6H2O | K2HPO4 | DMAc | 73 |
| 15c | None | K2HPO4 | DMAc | 0 |
| 16d | Ru(bpy)3Cl2•6H2O | K2HPO4 | DMAc | 0 |
| [1] |
For selected reviews: (a) Huang, H.-M.; Bellotti, P.; Glorius, F. Chem. Soc. Rev. 2020, 49, 6186.
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
| [2] |
For selected reviews: (a) Chen, Z.-M.; Zhang, X.-M.; Tu, Y.-Q. Chem. Soc. Rev. 2015, 44, 5220.
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(陈亚苏, 曹柱, 吴鑫鑫, 朱晨, 中国科学:化学, 2023, 53, 289.)
|
|
|
(h)
|
|
|
(i)
|
|
|
(j)
|
|
|
(k)
|
|
|
(贺重隆, 周有康, 段新华, 刘乐, 有机化学, 2025, 45, 1478.)
|
|
| [3] |
For selected examples: (a) Wu, Z.; Ren, R.; Zhu, C. Angew. Chem., Int. Ed. 2016, 55, 10821.
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
|
(i)
|
|
|
(j)
|
|
|
(k)
|
|
|
(l)
|
|
|
(m)
|
|
|
(n)
|
|
|
(o)
|
|
|
(p)
|
|
|
(q)
|
|
|
(r)
|
|
|
(s)
|
|
|
(t)
|
|
|
(u)
|
|
|
(v)
|
|
|
(w)
|
|
|
(x)
|
|
|
(y)
|
|
| [4] |
For selected examples from our group: (a) Yu,J.; Wu, Z.; Zhu, C. Angew. Chem., Int. Ed. 2018, 57, 17156.
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
|
(i)
|
|
|
(j)
|
|
|
(k)
|
|
|
(l)
|
|
| [5] |
For selected examples from other groups: (a) Monos,T. M.; McAtee, R. C.; Stephenson, C. R. J. Science 2018, 361, 1369.
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
|
(g)
|
|
|
(h)
|
|
|
(i)
|
|
|
(j)
|
|
|
(k)
|
|
| [6] |
(a)
|
|
(b)
|
|
|
(c)
|
|
|
(d)
|
|
|
(e)
|
|
|
(f)
|
|
| [7] |
|
| [8] |
|
| [9] |
|
| [1] | 侯馨怡, 史同同, 李泽江, 董庆昊, 孙凯, 王薪. 可见光诱导的磷酰化环化合成磷酰基取代的吲哚并二氮䓬[J]. 有机化学, 2026, 46(4): 1776-1787. |
| [2] | 王齐翔, 陈圣希, 王静, 陈栋, 鲍晓光, 吴新鑫. 无金属参与的多烯选择性氯代反应[J]. 有机化学, 2026, 46(4): 1750-1762. |
| [3] | 郑婧斐, 吕琪妍, 孙凯, 陈晓岚, 屈凌波, 王金泉, 於兵. 自由基介导的6-氮杂尿嘧啶官能团化研究进展[J]. 有机化学, 2026, 46(4): 1603-1620. |
| [4] | 刘世峥, 窦明煜, 崔玉, 窦建民, 魏文廷. 自由基硒磺酰化研究进展[J]. 有机化学, 2026, 46(4): 1621-1634. |
| [5] | 赵雨晴, 朱先进, 石凌宇, 魏鸣宇, 徐紫涵, 石永佳, 李旭锋, 杨道山. 环硫鎓盐开环官能化反应的研究进展[J]. 有机化学, 2026, 46(4): 1572-1602. |
| [6] | 张皓然, 贾均松, 俞琼, 吴宇, 李玉龙, 舒伟. 可见光催化烯烃的氢胺化和氢(磺)酰胺化反应[J]. 有机化学, 2026, 46(4): 1166-1180. |
| [7] | 卢琳玉, 亓雨彤, 张庭珲, 金胜男, 曹中艳, 张洪伟. 自由基介导烯烃的1,2,n-三官能团化反应研究进展[J]. 有机化学, 2026, 46(4): 1540-1559. |
| [8] | 董建洋, 薛东. 自由基途径的[1.1.1]螺桨烷官能化研究进展[J]. 有机化学, 2026, 46(4): 1464-1480. |
| [9] | 孟令旭, 申霖, 罗祥玲, 林玉妹, 龚磊. 具有光活性的铬、钴配合物的设计及其在有机合成中的应用进展[J]. 有机化学, 2026, 46(4): 1513-1528. |
| [10] | 苏雯鑫, 崔浩, 张霄. 单质硫(S8)参与的光化学反应研究进展[J]. 有机化学, 2026, 46(4): 1360-1375. |
| [11] | 刘琼, 杨修龙, 李浩源, 沈佳慧, 杨硕, 孟庆元, 牛效莹. 盐酸-二甲基亚砜体系促进酰胺的需氧光催化N-脱甲基反应[J]. 有机化学, 2026, 46(4): 1713-1721. |
| [12] | 吉伟文, 金健. 配体-金属电荷转移驱动的光促金属催化芳基羧酸脱羧转化[J]. 有机化学, 2026, 46(4): 1320-1329. |
| [13] | 赵丽玮, 张梁, 胡鹏. 氢原子迁移(HAT)介导的官能团迁移反应[J]. 有机化学, 2026, 46(4): 1284-1302. |
| [14] | 余宁, 周于强, 魏晔. 光催化自由基介导的内酯合成研究进展[J]. 有机化学, 2026, 46(4): 1222-1254. |
| [15] | 袁子亮, 李斌栋, 王定海. 可见光催化的烯烃氢膦化反应[J]. 有机化学, 2026, 46(4): 1677-1684. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||